

Background Systemic lupus erythematosus (SLE) is characterised by chronic stimulation of the innate and adaptive immune response. Oxidative stress resulting from Reactive Oxygen Species (ROS) generation is another hallmark of the disease. Mitochondria are a primary source of ROS and play a key role in energy metabolism via Oxidative Phosphorylation (OXPHOS), a vital pathway in maintaining the aberrant immunological response seen in the disease. Currently a number of novel therapeutics focusing on abnormal mitochondrial energy metabolism are under investigation in a variety of autoimmune diseases. Understanding abnormal immune cell mitochondrial function therefore represents an important area for identifying potential future targets in the treatment of SLE.
Objectives To assess for evidence of abnormal mitochondrial function and associated ROS generation in the adaptive immune response in SLE.
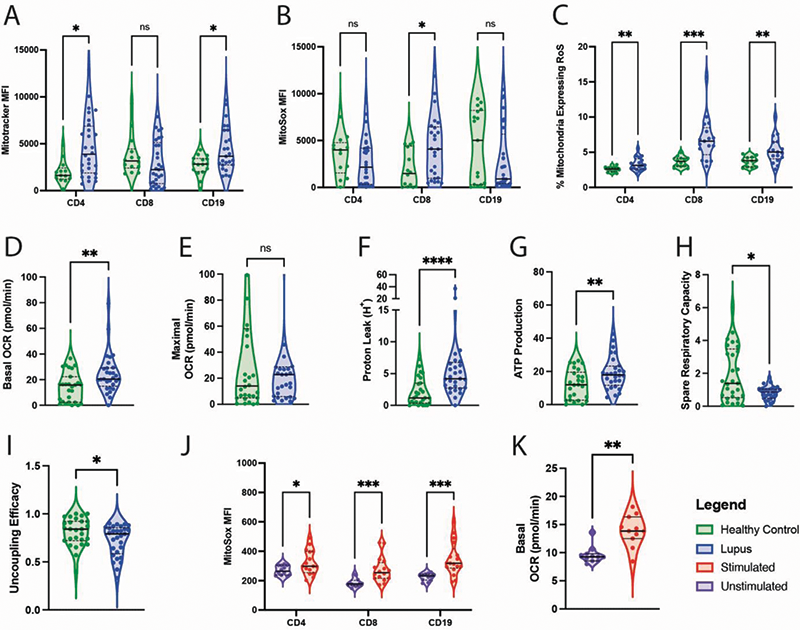
Methods Peripheral blood mononuclear cells (PBMCs) were isolated from whole blood collected from patients with SLE recruited from the UCLH Lupus Clinic, England (n=37) and age/sex matched healthy controls (HC, n=20). Flow cytometry was used to quantify median fluorescence intensity (MFI) of cellular mitochondrial mass (MitoTracker) and mitochondria derived ROS generation (MitoSOX) in CD4+ and CD8+ T cells plus CD19+ B cells. Real-time CD4+ T cell mitochondrial metabolic function was assessed using SeaHorse Respirometry MitoStress Test. To assess for the role of cellular activation in CD4+ T cells, 24-hour anti-CD3/CD28 stimulation culture was conducted.
Results Results are outlined in Figure 1. Briefly, mitochondrial mass was significantly higher in SLE CD4+ and CD19+ cells when compared with HC (A). CD8+ cells derived from patients with SLE showed higher amounts of mitochondrial ROS generation (B). When correcting for mitochondrial mass, individual mitochondria-derived ROS generation was significantly higher in CD4, CD8 and CD19+ cells in SLE when compared with HC (C). SeaHorse Respirometry revealed basal CD4+ respiration was higher in SLE when compared with HC (D), although there was no significant difference in maximal mitochondrial respiratory capacity (E). Proton leak, a marker of mitochondrial damage, was significantly higher in SLE than HC (F). CD4+ T cells from patients with SLE also showed enhanced mitochondrial ATP production when compared with HC (G), although spare respiratory capacity was significantly reduced in SLE (H) suggesting that mitochondria function in the disease is close to maximum with little scope for further upregulation. Mitochondrial uncoupling efficacy is an important mechanism through which higher rates of OXPHOS can be achieved and this appears to be downregulated in SLE (I). Activation of SLE CD4+ T cells appeared to drive higher rates of ROS production in both CD4+ and CD8+ cells (J). Interestingly, CD19+ ROS production was also increased, suggesting that T cell secreted cytokines following activation may also induce B cell ROS generation. Stimulation of SLE CD4+ T cells additionally increased basal mitochondrial respiration when compared with unstimulated SLE CD4+ T cells (K).
Conclusion Increased mitochondrial mass seen in CD4+ and CD19+ SLE cells could be a result of enhanced immune cell senescence, commonly observed in the state of chronic immune activation. At baseline, SLE derived CD4+ T cells were noted to generate ATP through OXPHOS at a higher rate than HC thus suggesting chronic cellulation activation (and is further enhanced following T cell stimulation). CD4+ T cells also demonstrate a number of important pathological abnormalities in SLE, including high levels of proton leak, reduced spare respiratory capacity and lower ability for mitochondrial uncoupling. This may suggest that chronic activation is in turn resulting in mitochondrial damage and less efficient energy metabolism, resulting in ROS generation. Targeting this abnormal immune cell metabolic pathway therefore represents a novel therapeutic strategy.
Figure 1.
Image/graph:
Acknowledgements This research is funded through support from Versus Arthritis and LUPUS UK awarded to CW.
Disclosure of Interests None declared.
Keywords: Systemic lupus erythematosus, Adaptive immunity
DOI: 10.1136/annrheumdis-2023-eular.6118