

Background DARWIN 3 (NCT02065700) is a long-term extension (LTE) study assessing the safety and efficacy of filgotinib (FIL) in patients with rheumatoid arthritis (RA) and an inadequate response to methotrexate (MTX).[1] In the DARWIN 1 (NCT01888874) and DARWIN 2 (NCT01894516) parent studies, patients received FIL in combination with MTX or FIL monotherapy, respectively.
Objectives To provide an update on the safety and efficacy of FIL 200 mg (FIL200) in patients with RA, with or without MTX, with a maximum of 8.2 years of exposure.
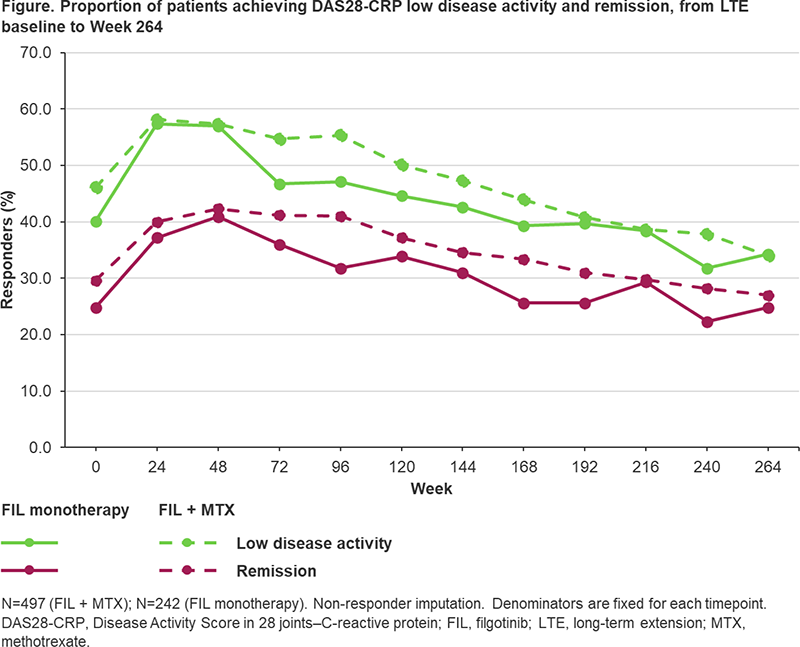
Methods Patients completing the DARWIN 1 (FIL + MTX) and DARWIN 2 (FIL monotherapy) phase 2 studies could enter DARWIN 3, receiving FIL200. The proportion of patients experiencing treatment-emergent adverse events (TEAEs) were reported using the safety analysis set, comprising data from both the parent and LTE studies. Efficacy was assessed from LTE baseline using the American College of Rheumatology (ACR) 20/50/70 improvement criteria and Disease Activity Score in 28 joints–C-reactive protein (DAS28-CRP), up to 264 weeks. Low disease activity and remission were defined as DAS28-CRP ≤3.2 and <2.6, respectively.
Results In total, 739 patients were enrolled in DARWIN 3. Mean (standard deviation; SD) FIL exposure was 4.89 (2.72) years in the FIL + MTX group and 4.78 (2.79) years in the FIL monotherapy group. In the FIL + MTX vs FIL monotherapy groups, TEAEs were reported for 90.9% and 92.1% of patients, respectively (Table 1). The most common TEAE was infection. In both treatment groups, 8 patients had a TEAE leading to death (1.6% and 3.3%, respectively). Exposure-adjusted incidence rates, censored at time of first event for major adverse cardiovascular event, venous thromboembolism, herpes zoster, infections, serious infections, non-melanoma skin cancer (NMSC), malignancies excluding NMSC, gastrointestinal perforations and TEAEs leading to death, will be reported. Through 5 years, ACR20/50/70 responses were maintained in 86.3%/66.7%/50.7% of the FIL + MTX group and 90.8%/74.8%/51.4% of the FIL monotherapy group, respectively (observed data). DAS28-CRP low disease activity and remission rates (non-responder imputation) at DARWIN 3 baseline were 46.1%/40.1% (FIL + MTX) and 29.6%/24.8% (FIL monotherapy) (Figure 1). At Week 264, the proportion of patients achieving low disease activity and remission were 34.0%/34.3% (FIL + MTX) and 27.0%/24.8% (FIL monotherapy).
Conclusion With a maximum of 8.2 years of exposure in patients with RA, the FIL safety profile is similar between the background MTX and monotherapy treatment arms. Both arms show sustained efficacy over time.
Reference [1]Kavanaugh A, et al. J Rheumatol 2021;48:1230–8.
Image/graph:
| FIL + MTX N=497 | FIL monotherapy N=242 | |
|---|---|---|
| Any TEAE | 452 (90.9) | 223 (92.1) |
| MACE | 4 (0.8) | 3 (1.2) |
| VTE | 3 (0.6) | 0 |
| Herpes zoster | 30 (6.0) | 14 (5.8) |
| Infections | 317 (63.8) | 140 (57.9) |
| Serious infections | 28 (5.6) | 19 (7.9) |
| NMSC | 6 (1.2) | 2 (0.8) |
| Malignancies excluding NMSC | 12 (2.4) | 8 (3.3) |
| GI perforations | 1 (0.2) | 0 |
| TEAEs leading to death | 8 (1.6) | 8 (3.3) |
Data are n (%).
FIL, filgotinib; GI, gastrointestinal; MACE, major adverse cardiovascular event; MTX, methotrexate; NMSC, non-melanoma skin cancer; TEAE, treatment-emergent adverse event; VTE, venous thromboembolism.
Acknowledgements Funding: DARWIN 3 was co-funded by Gilead Sciences Inc. (Foster City, CA, USA) and Galapagos NV (Mechelen, Belgium). Publication coordination was provided by Fabien Debailleul, PhD, of Galapagos NV. Medical writing support was provided by Iain Haslam, PhD, CMPP (Aspire Scientific Ltd, Bollington, UK), and funded by Galapagos NV.
Disclosure of Interests Rene Westhovens Speakers bureau: Celltrion, Galapagos, and Gilead, Consultant of: Celltrion, Galapagos, and Gilead, Rieke Alten Consultant of: AbbVie, Amgen, Biogen, BMS, Celltrion, Gilead, Janssen, Lilly, Medac, MSD, Mylan, Novartis, Pfizer, Roche, Sandoz, Sanofi-Genzyme, UCB, and VIATRIS, Lorenzo Dagna Consultant of: AbbVie, Amgen, AstraZeneca, Biogen, Boehringer-Ingelheim, BMS, Celltrion, Eli Lilly & Company, Galapagos, GlaxoSmithKline, Janssen, Kiniksa Pharmaceuticals, Novartis, Pfizer, Roche, Sanofi-Genzyme, SOBI, and Takeda, Grant/research support from: AbbVie, BMS, Celgene, GlaxoSmithKline, Janssen, Kiniksa, Merck Sharp & Dohme, Mundipharma Pharmaceuticals, Novartis, Pfizer, Roche, Sanofi-Genzyme, and SOBI, Arthur Kavanaugh Consultant of: Amgen, AbbVie, BMS, Pfizer, UCB, Janssen, Novartis, and Pfizer, Kevin Winthrop Consultant of: AbbVie, AstraZeneca, BMS, Eli Lilly & Company, Galapagos, Gilead, GlaxoSmithKline, Novartis, Pfizer, Regeneron, Roche, Sanofi, and UCB, Grant/research support from: BMS and Pfizer, Jane Barry Shareholder of: Galapagos, Employee of: Galapagos, Robin Besuyen Shareholder of: Galapagos, Employee of: Galapagos, Claudio Corallo Shareholder of: Galapagos, Employee of: Galapagos, Dick de Vries Shareholder of: Galapagos, Employee of: Galapagos, Nicolas Martin Consultant of: Galapagos, Chris Watson Shareholder of: Galapagos, Employee of: Galapagos, Mark Genovese Shareholder of: Gilead Sciences, Inc., Employee of: Gilead Sciences, Inc., Alberto Spindler: None declared, Mykola Stanislavchuk Grant/research support from: As a Principal Investigator for: Amgen, AstraZeneca, Celgene, Eli Lilly, Galapagos, Gilead, Human Genome, Janssen, MSD, Nichi-Iko, Pfizer, and Roche., Maria Greenwald Grant/research support from: AbbVie, Aclaris, Galapagos, Janssen, Lilly, and Nimbus, Paul Emery Speakers bureau: AbbVie, AstraZeneca, BMS, Boehringer-Ingelheim, Galapagos, Gilead, Lilly, Novartis, Pfizer, and Samsung, Consultant of: AbbVie, AstraZeneca, BMS, Boehringer-Ingelheim, Galapagos, Gilead, Lilly, Novartis, Pfizer, Roche, and Samsung, Grant/research support from: AbbVie, BMS, Lilly, Novartis, Pfizer, Roche, and Samsung.
Keywords: Rheumatoid arthritis, Safety
DOI: 10.1136/annrheumdis-2023-eular.3508