

Background The EULAR guidelines for RA recommend that treatment should be aimed at reaching a predefined disease activity target (T2T). If this treatment target is not achieved with csDMARD, adding a TNFi or a JAK-inhibitor are advised options in patients with poor prognostic factor, obviously considering contraindications. While randomized clinical trials have provided relevant data on the relative efficacy and safety of TNFi and JAKi under ideal conditions, the extent to which such results can be generalized to real-life clinical practice conditions remains unclear.
Objectives To demonstrate non-inferiority (NI) and, in case NI could be shown, subsequent superiority of a T2T strategy in which csDMARDs refractory RA patients are subsequently treated with baricitinib versus TNFi.
Methods Biologic or targeted synthetic DMARD-naïve RA patients failing to respond to csDMARDs were eligible if they were pretreated according to T2T principles, had a disease duration <5 years and no contraindications to b/tsDMARD. All included patients were treated open label at the discretion of their attending rheumatologist with either TNFi (any type) or baricitinib. Patients were seen at baseline and 12-weekly until final follow-up (48 weeks). Full clinical assessment was performed at each visit. Self-report questionnaires, including PROMs, were collected as well. The primary endpoint was defined as NI of baricitinib versus TNFi in the proportion of patients achieving ACR50 response at week 12, with subsequent superiority testing in case non-inferiority was shown. For the primary efficacy analysis, the proportion of patients achieving ACR50 response were compared, using 95% confidence intervals calculated using the Wilson score method. The non-inferiority margin for baricitinib was set at -12%.
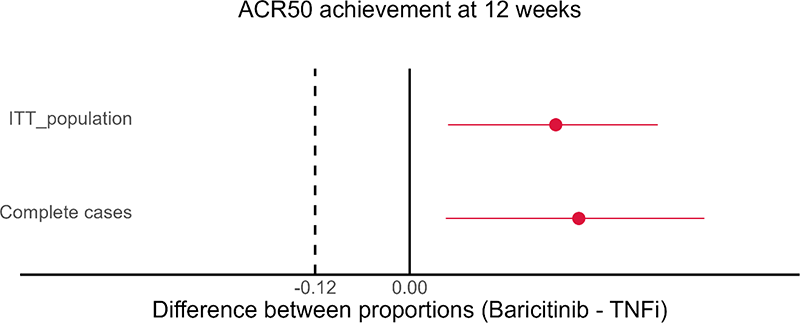
Results 199 patients who received a first dose of either TNFi (102) or baricitinib (97) were included. Baseline characteristics were comparable between both groups (see Table 1). At 12 weeks, the lower bound of the 95% confidence interval for the difference in proportions of patients meeting the ACR50 response is to the right of zero in both the per-protocol and intention-to-treat analysis (see Figure 1). Hence, baricitinib was found to be non-inferior and statistically superior to TNFi in the analysis of the primary endpoint. Moreover, DAS28 remission (DAS28-CRP <0.6) was achieved in 74% of baricitinib patients compared with 46% of TNFi patients (p<0.001) at 12 weeks.
Conclusion Baricitinib was found to be non-inferior and superior to TNFi in terms of ACR50 response at 12 weeks in real-world csDMARD refractory RA patients. Analysis of secondary endpoints, disease activity across other measurement points, PROMs, radiology, safety and costs is currently ongoing.
| TNFi | Baricitinib | p | |
|---|---|---|---|
| N=102 | N=97 | ||
| Age | 55.2 (13.4) | 54.8 (12.0) | 0.833 |
| Gender | 0.796 | ||
| F | 68 (66.7%) | 62 (63.9%) | |
| M | 34 (33.3%) | 35 (36.1%) | |
| Smoking | 0.963 | ||
| never | 38 (37.3%) | 37 (38.9%) | |
| Stopped | 39 (38.2%) | 36 (37.9%) | |
| Yes | 25 (24.5%) | 22 (23.2%) | |
| BMI | 27.5 (4.91) | 26.5 (5.03) | 0.178 |
| Disease duration (years) | 2.00 [1.00;3.00] | 2.00 [1.00;3.00] | 0.766 |
| Erosions | 0.537 | ||
| No | 71 (69.6%) | 74 (76.3%) | |
| Unknown | 12 (11.8%) | 10 (10.3%) | |
| Yes | 19 (18.6%) | 13 (13.4%) | |
| CV | 0.432 | ||
| No | 74 (72.5%) | 76 (78.4%) | |
| Yes | 28 (27.5%) | 21 (21.6%) | |
| RF | 0.576 | ||
| Neg | 33 (32.4%) | 27 (27.8%) | |
| Pos | 67 (65.7%) | 66 (68.0%) | |
| Unknown | 2 (1.96%) | 4 (4.12%) | |
| ACCP | 0.262 | ||
| Neg | 37 (36.3%) | 27 (27.8%) | |
| Pos | 65 (63.7%) | 70 (72.2%) | |
| MTX | 1 | ||
| No | 31 (30.4%) | 30 (30.9%) | |
| Yes | 71 (69.6%) | 67 (69.1%) | |
| DAS28ESR | 4.41 (1.06) | 4.43 (1.14) | 0.919 |
| DAS28CRP | 4.17 (1.02) | 4.12 (1.07) | 0.741 |
| TJC28 | 4.00 [2.00;7.00] | 4.00 [2.00;7.00] | 0.662 |
| SJC28 | 3.00 [1.00;5.00] | 3.00 [2.00;4.00] | 0.805 |
| BSE | 24.0 (19.7) | 25.0 (22.1) | 0.738 |
| CRP | 13.7 (19.1) | 12.8 (17.6) | 0.728 |
| HAQ | 12.2 (5.66) | 10.9 (6.35) | 0.13 |
| SDAI | 21.2 (8.97) | 21.1 (9.00) | 0.933 |
Figure 1.
Image/graph:
REFERENCES:
NIL.
Acknowledgements: NIL.
Disclosure of Interests Martjin Oude Voshaar: None declared, Peter ten Klooster: None declared, Danyta Tedjo: None declared, Celine van de Laar: None declared, Mart van de Laar Speakers bureau: Eli Lilly and Company, Grant/research support from: Eli Lilly and Company.
Keywords: Randomized control trial, Targeted synthetic drugs, Real-world evidence
DOI: 10.1136/annrheumdis-2023-eular.4317