

Background Interstitial lung disease (ILD) is a severe extra-articular manifestation of rheumatoid arthritis (RA). Abatacept and rituximab are the recommended drugs [1-2]. JAK inhibitors (JAKi) have demonstrated efficacy in RA. However, in clinical trials patients with active ILD were usually excluded. Moreover, a warning on ILD toxicity is included in SmPC (Summary of Product Characteristics) with tofacitinib (TOFA). Nonetheless, evidence on efficacy of JAKi in RA-ILD is growing [3].
Objectives to assess a) the effectiveness and b) the safety of JAKi in AR-ILD patients.
Methods National multicenter study of 57 RA-ILD patients on treatment with JAKi. We analyzed from baseline the following outcomes: a) forced vital capacity (FVC), b) diffusing capacity of the lungs for carbon monoxide (DLCO), c) chest high resolution computed tomography (HRCT), d) dyspnea (modified Medical Research Council scale), e) arthritis activity (DAS28-ESR or clinical records), and f) sparing corticosteroids effect.
Results We studied 57 patients (37 women/ 20 men; mean age 66 ±10 years) from clinical practice on treatment with JAKi [baricitinib (BARI)= 42 (74%), TOFA= 6 (11%), upadacitinib (UPA)= 8 (14%), filgotinib (FILGO)= 1 (2%)]. Baseline demographic and clinical characteristics are shown in Table 1. All patients had received disease-modifying antirheumatic drugs (DMARDs) before JAKi [Methotrexate (49,86%), Leflunomide (37, 65%), Sulfasalazine (14, 25%), Hydroxychloroquine (13, 23%), Abatacept (32, 56%), Tocilizumab (14, 25%) and Rituximab (10, 18%)].
Since most patients were on BARI we focused on this group (n=42). Median [IQR] ILD duration up to BARI initiation was of 42 [13-62] months. Mean baseline values of FVC and DLCO (% predicted) were 87±22 and 71±21, respectively. Patients were followed-up for a mean of 37 ±37 months. The evolution of FVC and DLCO remained stable during the first 12 months (Figure 1). At the end of the follow-up, available chest HRCT images improved/ stabilized in 82% of patients. Stabilization or improvement of dyspnea was found in 87% of patients. Most patients showed articular remission or low activity. BARI was withdrawn in 12 (29%) patients due to articular inefficacy (n=11) and development of hypersensitivity pneumonitis (n=1).
Conclusion JAKi, especially BARI, may be useful and safe in controlling the course of both pulmonary and joint disease in RA-ILD patients, even in refractory cases.
References
Image/graph:
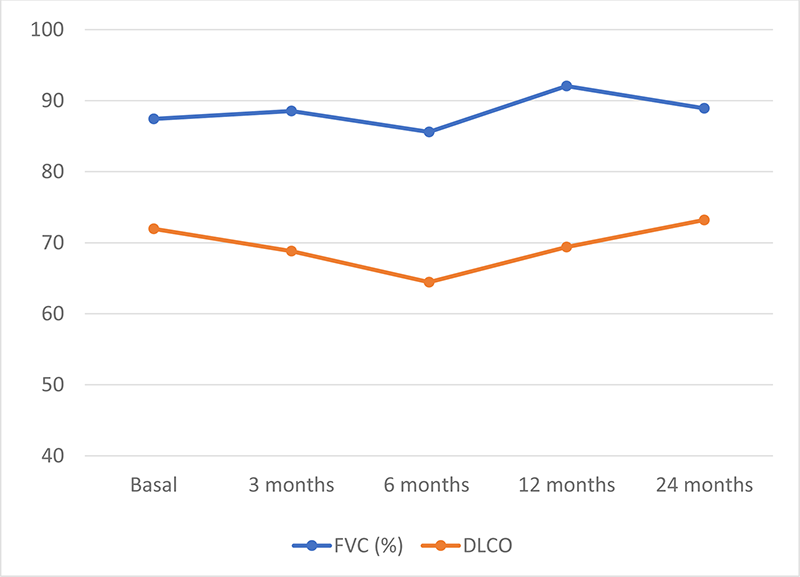
Figure 1. Evolution of pulmonary function tests (mean % of the predicted FVC and DLCO) in RA-ILD patients with BARI therapy at baseline and 24 months.
| RA-ILD with JAKi (n=57) | |
|---|---|
| Age, years mean±SD | 66 ± 10 |
| Women, n (%) | 37 (64,9) |
| Smoker ever, n (%) | 36 (63) |
| Time since ILD diagnosis, months, median [IQR] | 49 [14-63] |
| RF// ACPA, n (%) | 56 (98) |
| FVC (% of the predicted), mean±SD | 90 ±22 |
| DLCO (% of the predicted), mean±SD | 71 ±19 |
| UIP-like fibrotic pattern on HRCT, n (%) | 30 (55) |
| Joint activity n (%) | 41 (89) |
| Type of JAKi, n (%) | |
| Baricitinib (BARI) | 42 (74) |
| Tofacitinib (TOFA) | 6 (11) |
| Upadacitinib (UPA) | 8 (14) |
| Filgotinib (FILGO) | 1 (2) |
| Previous immunosuppressive therapy, n (%) | |
| Conventional/ biologic DMARD | 57 (100)/ 47 (82) |
| Concomitant immunosuppresive therapy, n (%) | |
| Conventional/ biologic DMARD | 19 (33)/ 0 (0) |
| Concomitant antifibrotic therapy, n (%) | 5 (9) |
ACPA, anti-citrullinated protein antibodies; DLCO, diffusing capacity of the lung for carbon monoxide; DMARD, disease-modifying antirheumatic drug; FVC, forced vital capacity; HRCT, high-resolution computed tomography; ILD, interstitial lung disease; JAKi, JAK inhibitor; RA, rheumatoid arthritis; RF, rheumatoid factor; UIP, usual interstitial pneumonia.
Acknowledgements: NIL.
Disclosure of Interests Ana Serrano-Combarro: None declared, Belén Atienza-Mateo: None declared, Jesús Alejandro Valero Jaimes: None declared, MARTA PASTOR MENA: None declared, Rafael Melero: None declared, David Castro-Corredor: None declared, MARIA MARTIN LOPEZ: None declared, Santos Castañeda: None declared, Jesús Loarce-Martos: None declared, Natalia Mena-Vázquez: None declared, Maria del Carmen Carrasco Cubero: None declared, Carolina Díez: None declared, Andrea García-Valle: None declared, Gemma Bonilla: None declared, J M Blanco: None declared, N. Del-Val: None declared, Nuria Vegas-Revenga: None declared, Lorena Pérez Albaladejo: None declared, Rafaela Ortega Castro: None declared, DESEADA PALMA SANCHEZ: None declared, ANA MARIA FERNANDEZ ORTIZ: None declared, Patricia López Viejo: None declared, María América López Lasanta: None declared, Marta Garijo Bufort: None declared, Ivette Casafont-Solé: None declared, Olga Maiz: None declared, Juan Moreno Morales: None declared, ANA URRUTICOECHEA-ARANA: None declared, Carolina Pérez: None declared, Jose Rosas: None declared, Diego Ferrer: None declared, Ricardo Blanco Speakers bureau: Abbvie, Pfizer, Roche, lilly, Bristol-Myers, Janssen, Galapagos and MSD., Consultant of: Abbvie, Pfizer, Roche, lilly, Bristol-Myers, Janssen and MSD., Grant/research support from: Abbvie, MSD, novartis and Roche.
Keywords: Rheumatoid arthritis, Lungs, Disease-modifying drugs (DMARDs)
DOI: 10.1136/annrheumdis-2023-eular.5209