

Background Upadacitinib (UPA), an oral JAK inhibitor, was shown to be safe and effective in improving the signs and symptoms of moderate-to-severe RA through 84 weeks (wks) when administered as monotherapy in patients (pts) with a prior inadequate response to MTX in the phase 3 SELECT-MONOTHERAPY trial (NCT02706951).[1]
Objectives To evaluate the efficacy and safety of UPA monotherapy up to wk 260 from the long-term extension (LTE) of SELECT-MONOTHERAPY.
Methods Pts with active RA on stable MTX were randomly assigned to either continue MTX (cMTX) or switch to UPA monotherapy at 15 mg (UPA15) or 30 mg (UPA30) once daily (QD) during the 14-wk randomized, double-blind treatment period. From wk 14, the start of the LTE, pts receiving cMTX were switched to UPA15 or 30 per pre-specified assignment at baseline; pts randomly assigned to UPA15 or 30 continued their initial treatment assignment. After the study protocol was amended (13 December 2019), all pts treated with UPA30 were eventually switched to UPA15 (approved dose). Efficacy outcomes through wk 260 are presented by randomized treatment group and reported as observed and using non-responder imputation. Safety results are presented based on actual treatment received with treatment-emergent adverse events (TEAEs) summarized per 100 pt years (PY) of exposure through a cut-off date of 10 August 2022, when all pts completed wk 260.
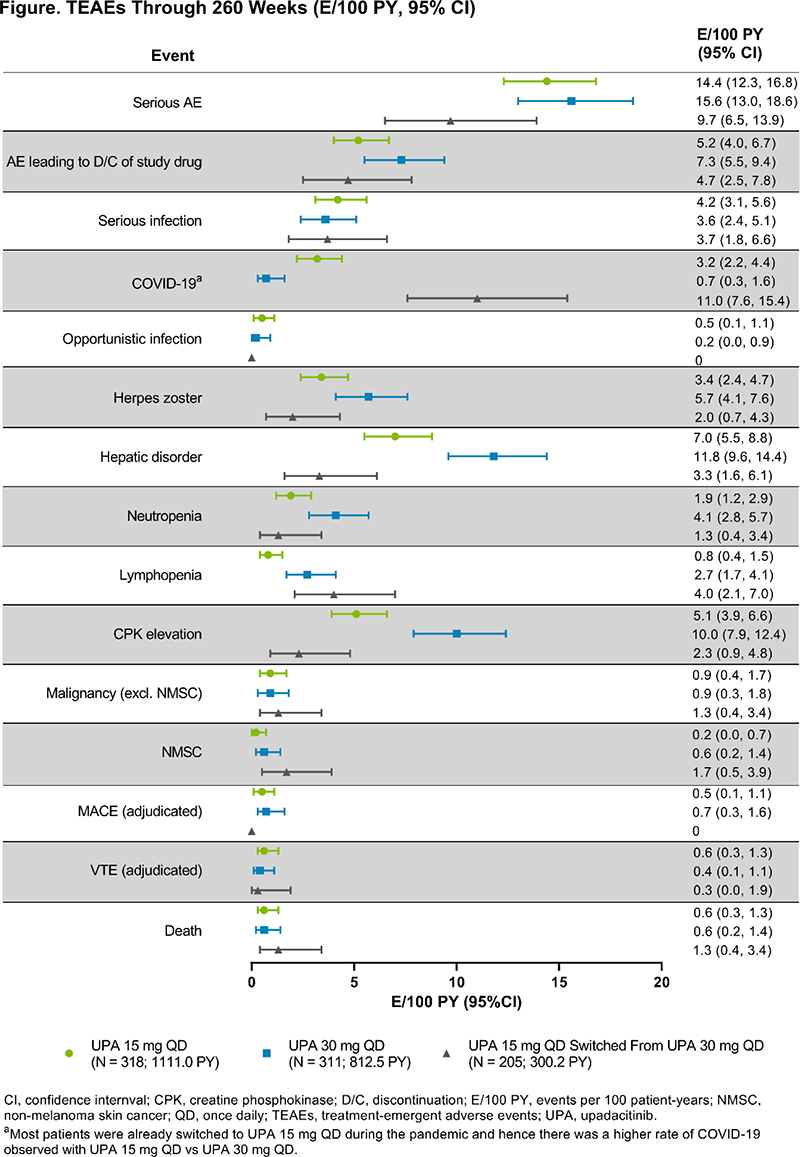
Results Of 648 pts randomized, 299 (46%) discontinued the study drug by wk 260 primarily due to AEs (16%), consent withdrawal (12%), or other reasons (11%). At wk 260, pts on UPA maintained or further demonstrated clinical improvement across various endpoints; similar efficacy outcomes were also observed among pts who switched from cMTX to UPA15 or 30 (Table 1). As observed, over three-quarters of pts achieved low disease activity based on CDAI and DAS28(CRP) at wk 260. Total PY of exposure were 1111.0 for UPA15, 812.5 for UPA30, and 300.2 for UPA15 switched from UPA30. The most frequently reported TEAEs were urinary tract infection, creatine phosphokinase (CPK) elevation, upper respiratory tract infection, nasopharyngitis, bronchitis, and RA worsening/flare. COVID-19 pneumonia was the most common serious AE. Pts on UPA30 had higher rates of herpes zoster (HZ), hepatic disorder, neutropenia, lymphopenia, and CPK elevation than pts on UPA15; rates of serious infection and malignancy excluding non-melanoma skin cancer (NMSC) were similar between UPA doses (Figure 1). Most HZ events affected 1–2 dermatomes with 2 ophthalmic (UPA15; 0.2 E/100 PY) and 1 disseminated (UPA30; 0.1 E/100 PY) cases reported. Eleven adjudicated MACE (UPA15: 0.5 E/100 PY; UPA30: 0.7 E/100 PY) and 10 adjudicated VTE (UPA15: 0.6 E/100 PY; UPA30: 0.4 E/100 PY; UPA15 switched from UPA30: 0.3 E/100 PY) all occurred in pts with ≥ 1 cardiovascular risk factor. Through wk 260, 16 deaths were reported (10 treatment-emergent) with 5 related to COVID-19 infection.
Conclusion UPA monotherapy continued to be effective in treating RA signs and symptoms through wk 260. No new safety signals were identified with longer-term exposure to UPA, consistent with prior findings[1–3] and the established safety profile of UPA across indications.
References
Image/graph:
| Response, % | cMTX -> UPA 15 mg | cMTX ->UPA 30 mg | UPA 15 mg | UPA 30 mg | ||||
|---|---|---|---|---|---|---|---|---|
| AO a N = 57 – 65 | NRI N = 108 | AO a N = 54 – 56 | NRI N = 108 | AO a N = 107 – 114 | NRI N = 217 | AO a N = 117 – 123 | NRI N = 215 | |
| ACR20/50/70 | 89/82/61 | 48/43/32 | 86/75/57 | 43/39/30 | 90/76/59 | 50/42/32 | 93/80/63 | 50/44/34 |
| DAS28(CRP) |
86/72 | 43/36 | 84/67 | 41/33 | 89/71 | 42/34 | 84/76 | 44/40 |
| CDAI |
88/49 | 48/28 | 82/40 | 40/20 | 86/42 | 43/21 | 79/54 | 42/29 |
| Boolean |
39 | 21 | 32 | 17 | 32 | 16 | 42 | 23 |
AO, as observed; cMTX, continue MTX; NRI, non-responder imputation; UPA, upadacitinib.
aAO response rate was calculated from patients with observed records only.
Acknowledgements AbbVie funded this study and participated in the study design, research, analysis, data collection, interpretation of data, review, and approval of the abstract. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship. Medical writing support was provided by Julia Zolotarjova, MSc, MWC, of AbbVie.
Disclosure of Interests Josef S. Smolen Speakers bureau: AbbVie, Amgen, AstraZeneca, Astro, BMS, Celgene, Celltrion, Chugai, Eli Lilly, Gilead, ILTOO, Janssen, Merck Sharp & Dohme, Novartis, Novartis-Sandoz, Pfizer, Roche, Samsung, and UCB, Consultant of: AbbVie, Amgen, AstraZeneca, Astro, BMS, Celgene, Celltrion, Chugai, Eli Lilly, Gilead, ILTOO, Janssen, Merck Sharp & Dohme, Novartis, Novartis-Sandoz, Pfizer, Roche, Samsung, and UCB, Grant/research support from: AbbVie, Amgen, AstraZeneca, Astro, BMS, Celgene, Celltrion, Chugai, Eli Lilly, Gilead, ILTOO, Janssen, Merck Sharp & Dohme, Novartis, Novartis-Sandoz, Pfizer, Roche, Samsung, and UCB, Paul Emery Consultant of: AbbVie, BMS, Eli Lilly, Gilead, Merck Sharp & Dohme, Novartis, Pfizer, Roche, and Samsung, Grant/research support from: AbbVie, BMS, Eli Lilly, Gilead, Merck Sharp & Dohme, Novartis, Pfizer, Roche, and Samsung, William Rigby Consultant of: AbbVie, BMS, Genentech, and Pfizer, Yoshiya Tanaka Speakers bureau: AbbVie, Amgen, Asahi-Kasei, Astellas, AstraZeneca, Boehringer-Ingelheim, BMS, Chugai, Corrona, Daiichi-Sankyo, Eisai, Eli Lilly, Gilead, Kowa, Mitsubishi-Tanabe, Takeda, YL Biologics, and Takeda, Consultant of: AbbVie, Amgen, Asahi-Kasei, Astellas, AstraZeneca, Boehringer-Ingelheim, BMS, Chugai, Corrona, Daiichi-Sankyo, Eisai, Eli Lilly, Gilead, Kowa, Mitsubishi-Tanabe, Takeda, YL Biologics, and Takeda, Grant/research support from: AbbVie, Amgen, Asahi-Kasei, Astellas, AstraZeneca, Boehringer-Ingelheim, BMS, Chugai, Corrona, Daiichi-Sankyo, Eisai, Eli Lilly, Gilead, Kowa, Mitsubishi-Tanabe, Takeda, YL Biologics, and Takeda, Juan Ignacio Vargas Consultant of: AbbVie, Nemanja Damjanov Consultant of: AbbVie, Gedeon Richter, Merck, Novartis, Pfizer, and Roche., Grant/research support from: AbbVie, Gedeon Richter, Merck, Novartis, Pfizer, and Roche., Manish Jain Speakers bureau: AbbVie, Amgen, Celgene, Eli Lilly, Medac, Novartis, Pfizer, and Takeda, Consultant of: AbbVie, Amgen, Celgene, Eli Lilly, Medac, Novartis, Pfizer, and Takeda, Grant/research support from: AbbVie, Amgen, Celgene, Eli Lilly, Medac, Novartis, Pfizer, and Takeda, Koji Kato Shareholder of: Employee of AbbVie and may own stock or options, Employee of: AbbVie, Kyle Carter Shareholder of: Employee of AbbVie and may own stock or options, Employee of: AbbVie, Nasser Khan Shareholder of: Employee of AbbVie and may own stock or options, Employee of: AbbVie, Heidi Camp Shareholder of: Employee of AbbVie and may own stock or options, Employee of: AbbVie, Stanley B. Cohen Consultant of: AbbVie, Amgen, Boehringer Ingelheim, Eli Lilly, Gilead, Pfizer, Roche, and Sandoz, Grant/research support from: AbbVie, Amgen, Boehringer Ingelheim, Eli Lilly, Gilead, Pfizer, Roche, and Sandoz.
Keywords: Rheumatoid arthritis, Targeted synthetic drugs, Safety
DOI: 10.1136/annrheumdis-2023-eular.2423