

Background Upadacitinib (UPA) is an oral reversible Janus kinase inhibitor. In patients (pts) with RA, UPA 15 mg once daily (QD) demonstrated better clinical responses at 12 weeks (wks) vs adalimumab (ADA) 40 mg every other week (EOW) in the phase 3 SELECT-COMPARE study; these were maintained through 3 years (yrs) in the ongoing long-term extension (LTE), along with an acceptable safety profile.[1,2]
Objectives To assess the safety and efficacy of UPA vs ADA through 5 yrs in the SELECT-COMPARE LTE.
Methods Pts receiving background methotrexate were randomized 2:2:1 to UPA 15 mg QD, placebo (PBO), or ADA 40 mg EOW. Rescue (PBO to UPA, UPA to ADA, or ADA to UPA) was mandated for lack of response (<20% improvement in tender or swollen joint counts at wks 14, 18, or 22), or failure to achieve Clinical Disease Activity Index (CDAI) low disease activity (LDA) at wk 26. All remaining PBO pts switched to UPA at wk 26. Pts who completed the 48-wk double-blind period could continue to receive open-label UPA or ADA in the LTE for up to 10 yrs total. Rates of treatment-emergent adverse events (TEAEs) and AEs of special interest were calculated per 100 pt-yrs through 5 yrs for all pts receiving UPA or ADA. Efficacy assessments at 5 yrs were performed by original randomized group for CDAI LDA (≤10) and remission (≤2.8), and disease activity score for 28-joints C-reactive protein (DAS28[CRP]) ≤3.2 and <2.6. Radiographic progression (change from baseline in modified total Sharp score [mTSS]) and proportion of pts with no radiographic progression (change from baseline in mTSS ≤0) were assessed at 192 wks (latest available timepoint; data collected at wks 96/192/520 only) by treatment sequence. No formal statistical comparisons were performed.
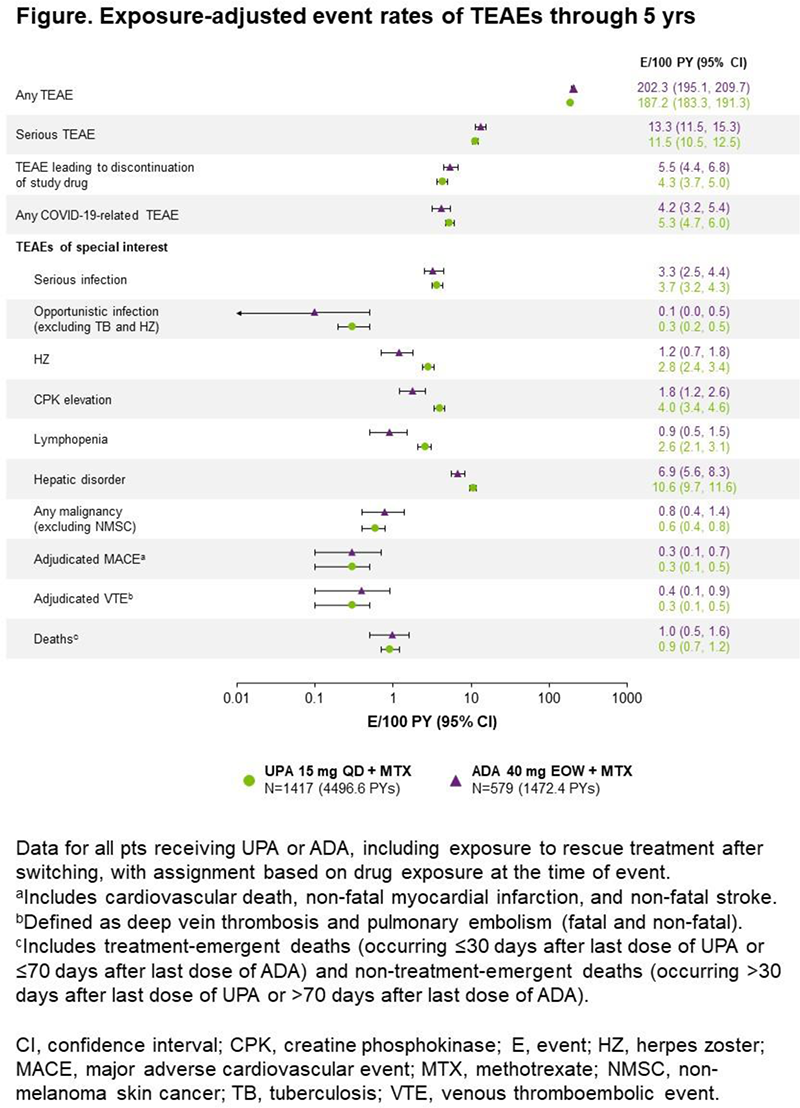
Results Through 5 yrs, 1417 pts were exposed to UPA (4497 pt-yrs) and 579 to ADA (1472 pt-yrs). UPA was generally well tolerated, with similar rates of TEAEs, serious TEAEs, TEAEs leading to discontinuation of study drug, and COVID-related TEAEs vs ADA (Figure 1). Rates of most AEs of special interest with UPA were similar vs ADA, except for numerically higher rates of herpes zoster, creatine phosphokinase elevation, lymphopenia, and hepatic disorder (mainly transaminase elevations) with UPA. In the 651 and 327 pts originally randomized to UPA and ADA, respectively, greater proportions of pts achieved CDAI LDA and remission, and DAS28(CRP) scores ≤3.2 and <2.6, with UPA vs ADA (Table 1). Through 192 wks, similar proportions of pts treated with UPA vs ADA had no radiographic progression; mean changes from baseline in mTSS were similar, except for a numerically smaller change with continuous UPA (Table 1).
Conclusion The safety profile of UPA over 5 yrs was consistent with the 3-yr results and the integrated phase 3 safety analysis.[1,2] Consistent with the 3-yr analyses,[2] UPA continued to show numerically better clinical responses than ADA at 5 yrs. Radiographic progression remained similarly low through 192 wks with UPA and ADA.
References
Image/graph:
| At 5 yrs, by original randomized group (non-responder imputation) | UPA N=651a,b | ADA N=327a,c | |||
|---|---|---|---|---|---|
| CDAI ≤10 | 36.4 (32.7, 40.1) | 26.9 (22.1, 31.7) | |||
| CDAI ≤2.8 | 24.6 (21.3, 27.9) | 18.7 (14.4, 22.9) | |||
| DAS28(CRP) ≤3.2 | 34.7 (31.1, 38.4) | 24.8 (20.1, 29.4) | |||
| DAS28(CRP) <2.6 | 31.8 (28.2, 35.4) | 23.2 (18.7, 27.8) | |||
| At 192 wks, by treatment sequence (as observed) | PBO to UPA N=442 | UPA N=288 | UPA to ADA N=150 | ADA N=109 | ADA to UPA N=111 |
| Radiographic progression (change from baseline in mTSS, mean [95% CI]) | 1.3 (0.8, 1.7) | 0.5 (0.2, 0.9) | 1.7 (0.7, 2.8) | 1.2 (0.5, 1.9) | 0.9 (0.2, 1.5) |
| No radiographic progression (mTSS change from baseline ≤0) | 77.1 |
80.9 |
74.0 |
78.0 |
77.5 |
Data are % of pts (95% confidence interval) unless otherwise stated.
aPts rescued at or before wk 26 were considered non-responders. b252 rescued. c159 rescued.
Acknowledgements AbbVie funded this trial and participated in the trial design, research, analysis, data collection, interpretation of data, and the review and approval of the publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship. Medical writing support was provided by Laura Chalmers, PhD, of 2 the Nth (Cheshire, UK), and was funded by AbbVie.
Disclosure of Interests Roy M. Fleischmann Consultant of: AbbVie, Amgen, Boehringer-Ingelheim, Bristol-Myers Squibb, Eli Lilly, Galapagos, Galvani, Gilead, GSK, Janssen, Novartis, Pfizer, and UCB, Grant/research support from: AbbVie, Amgen, Biosplice, Bristol-Myers Squibb, Flexion, Gilead, Horizon, Eli Lilly, Galvani, Janssen, Novartis, Pfizer, Sanofi-Aventis, Selecta, Teva, UCB, Viela, and Vorso, Jerzy Swierkot Speakers bureau: AbbVie, Accord, BMS, Janssen, MSD, Pfizer, Roche, Sandoz, and UCB, Consultant of: AbbVie, Accord, BMS, Janssen, MSD, Pfizer, Roche, Sandoz, and UCB, Grant/research support from: AbbVie, Accord, BMS, Janssen, MSD, Pfizer, Roche, Sandoz, and UCB, Sara Penn Employee of: AbbVie, and may hold stock or options, Patrick Durez Speakers bureau: AbbVie, Galapagos, Lilly, Nordimed, and Thermofischer, Louis Bessette Speakers bureau: AbbVie, Amgen, Bristol-Meyers Squibb, Celgene, Eli Lilly, Fresenius Kabi, Gilead, Janssen, Merck, Novartis, Pfizer, Roche, Sanofi-Aventis, Teva, and UCB, Consultant of: AbbVie, Amgen, Bristol-Meyers Squibb, Celgene, Eli Lilly, Fresenius Kabi, Gilead, Janssen, Merck, Novartis, Pfizer, Roche, Sanofi-Aventis, Teva, and UCB, Grant/research support from: AbbVie, Amgen, Bristol-Meyers Squibb, Celgene, Eli Lilly, Fresenius Kabi, Gilead, Janssen, Merck, Novartis, Pfizer, Roche, Sanofi-Aventis, Teva, and UCB, Xianwei Bu Employee of: AbbVie, and may hold stock or options, Nasser Khan Employee of: AbbVie, and may hold stock or options, Yihan Li Employee of: AbbVie, and may hold stock or options, Charles Peterfy Shareholder of: Spire Sciences, Inc, Consultant of: Daiichi Sankyo, Eli Lilly, Five Prime, Genentech, Gilead, GlaxoSmithKline, Istesso, Labcorp, Paradigm, SetPoint, Sorrento, and UCB, Employee of: Spire Sciences, Inc, Yoshiya Tanaka Speakers bureau: AbbVie, Asahi Kasei, Astellas, BMS, Chugai, Daiichi-Sankyo, Eisai, GSK, Janssen, Lilly, Mitsubishi Tanabe, MSD, Novartis, Ono, Pfizer, Sanofi, Taisho Toyama, Takeda, UCB, and YL Biologics, Grant/research support from: AbbVie, Asahi Kasei, Astellas, BMS, Chugai, Daiichi-Sankyo, Eisai, GSK, Janssen, Lilly, Mitsubishi Tanabe, MSD, Novartis, Ono, Pfizer, Sanofi, Taisho Toyama, Takeda, UCB, and YL Biologics, Eduardo Mysler Speakers bureau: AbbVie, Amgen, AZ, BMS, Janssen, Lilly, Novartis, Pfizer, Roche, Sandoz, and Sanofi, Paid instructor for: AbbVie, Amgen, AZ, BMS, Janssen, Lilly, Novartis, Pfizer, Roche, Sandoz, and Sanofi, Grant/research support from: AbbVie, Amgen, AZ, BMS, Janssen, Lilly, Novartis, Pfizer, Roche, Sandoz, and Sanofi.
Keywords: Safety, Rheumatoid arthritis, Targeted synthetic drugs
DOI: 10.1136/annrheumdis-2023-eular.2615