

Background In the age of targeted-synthetic disease-modifying antirheumatic drugs (tsDMARDs), filgotinib represents the last JAK inhibitor available in Europe for rheumatoid arthritis (RA). Filgotinib is characterized by predominantly inhibition of JAK1 and its efficacy and safety have been highlighted by phase 2/3 studies, but no real-life data in RA are currently available.
Objectives The aim of this study was to evaluate the effectiveness and safety profile of filgotinib in real-life setting in RA patients included in Italian GISEA (Group for the Study of Early Arthritis) registry.
Methods For this study, data from RA patients treated with filgotinib recorded in Italian GISEA registry were analysed. Disease activity scores and patients reported outcomes (PROs) were compared at baseline and six months follow-up using paired t-tests. The retention rate was estimated by the Kaplan-Meier method, while a cox regression model was used to search for possible factors influencing drug survival.
Results One hundred and seventy-nine patients (female 89.4%, age 57.8±12 years, FR/ACPA+ 64.3%, current/former smoker 31.8%) included in GISEA registry started filgotinib for active RA. Most patients were taking filgotinib as second (23.5%) or further (43%) b/tsDMARDs line of treatment. Filgotinib was used in monotherapy in 66.5% of patients, while 52% were not on treatment with glucocorticoids (GCs) at baseline. All demographic and clinical data are reported in Table 1. A follow-up visit was available for 122 patients (mean time of first follow-up visit: 4±2 months). As shown in table 1, we observed a decrease of all disease activity scores and PROs. At first follow-up visit, 67.8% of patients were in remission/low disease activity according to CDAI and 65.4% according to SDAI. Kaplan-Meyer analysis highlighted that drug persistence was similar either in monotherapy or combination therapy (Figure 1a), and irrespective of GCs at baseline (Figure 1b). However, a better persistence was observed in RA patients on first line treatment with filgotinib (Figure 1c). Thirty-five patients stopped filgotinib during follow-up, 10 for lack of efficacy, 4 for loss of efficacy, 4 for adverse events, while for the remaining cases the cause of drug discontinuation was unknown. No major cardiovascular events were reported. Finally, univariate Cox-regression model showed that b/tsDMARD naïve patients had a lower risk of drug discontinuation (naïve vs other lines: HR 0.37, 95%CI 0.20-0.86).
Conclusion In Italian real-life setting, filgotinib confirms a good effectiveness and safety profile.
| Variables | baseline (n. 179) | T1 (n. 122) | |
|---|---|---|---|
| Age | mean (SD), years | 57.8 (12.7) | 58 (12.6) |
| Gender | n. (%), female |
160 (89.4) |
110 (90.2) |
| BMI | n. (%), underweight |
4 (3.8) |
4 (5.3) |
| Smokers | n. (%), currently smoker |
13 (13.8) |
8 (13.5) |
| IgG RF/ACPA + | n. (%) | 74 (64.3) | 54 (71.1) |
| VAS pain | mean (SD) | 63.1 (29.1) | 35 (30.7)*** |
| VAS PtGA | mean (SD) | 60 (26.7) | 35 (29)*** |
| VAS PhGA | mean (SD) | 44 (25.4) | 20.9 (23)*** |
| TJC28 | mean (SD) | 5.4 (5) | 2.7 (3.8)** |
| SJC28 | mean (SD) | 3.3 (3.5) | 1.3 (2.2)*** |
| DAS28-ESR | mean (SD) | 4.6 (1.3) | 3.3 (1.4)** |
| CDAI | mean (SD) | 19.1 (11.3) | 9.6 (9.5)** |
| SDAI | mean (SD) | 20.5 (12) | 10.3 (10)** |
| HAQ-DI | mean (SD) | 1.3 (0.7) | 0.9 (0.7)*** |
| glucocorticoid | n. (%) | 86 (48) | 47 (38.5)** |
| Prednisone (equivalent), mg/die | mean (SD) | 5.8 (3.7) | 3.4 (2.6)* |
| csDMARD (in corso) | n. (%) | 60 (33.5) | 31 (25.4) |
| b/tsDMARD line | n. (%), 1^ line |
60 (33.5) |
/ |
Mean time of first follow-up visit: 4±2 months
*p<0.05, **p<0.01, ***p<0.001
Image/graph:
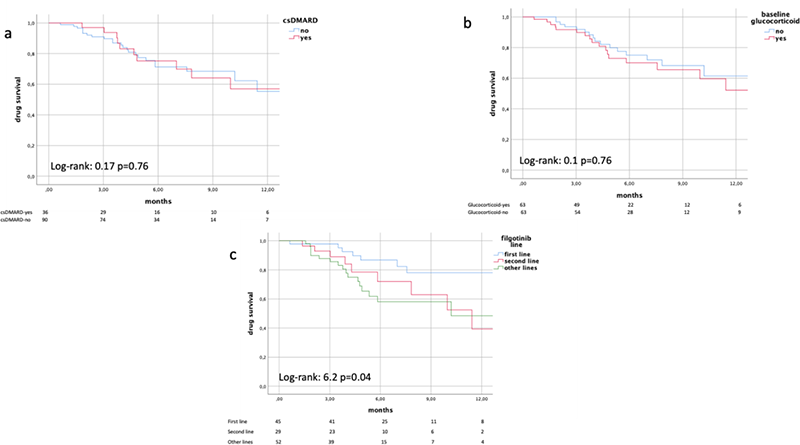
Figure 1. Survival analysis in RA patients treated with Filgotinib.
REFERENCES:
NIL.
Acknowledgements: NIL.
Disclosure of Interests None Declared.
Keywords: Targeted synthetic drugs, Rheumatoid arthritis, Registries
DOI: 10.1136/annrheumdis-2023-eular.3488