

Background The efficacy of Janus Kinase inhibitors (JAKis) in Rheumatoid Arthritis (RA) has been established in the last years through numerous randomized clinical trials. Recently, the safety profile of this drug class has come under the spotlight, questioning the actual use in clinical practice and making real-life data even more crucial.
Objectives The purpose of this study is to evaluate the long-term retention rate of JAKis and to estimate the influence of baseline population characteristics on treatment persistence.
Methods Data of all RA patients who started a JAKi were prospectively collected in the Italian multicentric GISEA registry. The 3-year retention rate of JAKis was calculated by the Kaplan-Meier method and compared by a log-rank test after stratification according to different baseline population characteristics. A descriptive analysis of reasons for discontinuation was performed.
Results The study population included 1361 patients (females 83.5%, mean age [±SD] 57.3 [±12.3] years, over 65 years 30%, mean disease duration 12.7 [±9.7] years, mean baseline SDAI 20.6 [±11.7], ACPA positive 68%, RF positive 66%, current smokers 19%, past smokers 13%) who received a JAKi (baricitinib=763, tofacitinib=321, upadacitinib=151, filgotinib=126) as first (n=491) or subsequent line (n=870) targeted drug. The overall 3-year retention rate was 46%. Drug survival was significantly higher in ACPA positive compared with negative patients (51.6 vs 41.8%, p=0.0051) (Figure 1A) and in first-line versus second or further lines of therapy (50.6 vs 43.3%, p=0.0013) (Figure 1B). No difference was found according to age (< or ≥65 years), concomitant methotrexate, smoke, history of diabetes, or at least one cardiovascular comorbidity (hypertension, stroke, or heart failure). Therapy was discontinued in a total of 523 patients because of ineffectiveness (67.5%), adverse events (25.8%), or compliance/other reasons (0.3%). Of relevance, adverse events included cancer (0.7%), major adverse cardiovascular events (MACE) (0.4%), deep vein thrombosis (DVT) (0.6%), and herpes zoster virus (HZV) infections (1.5%).
Conclusion Our data confirmed in a real-life setting a favorable 3-year retention rate of JAKis in RA. ACPA positivity and use as first-line targeted drug significantly improved persistence of JAKis. Discontinuations of JAKis because of adverse events (including MACE, DVT, HZV, and malignancy) were very uncommon, suggesting an overall favorable safety profile.
Image/graph:
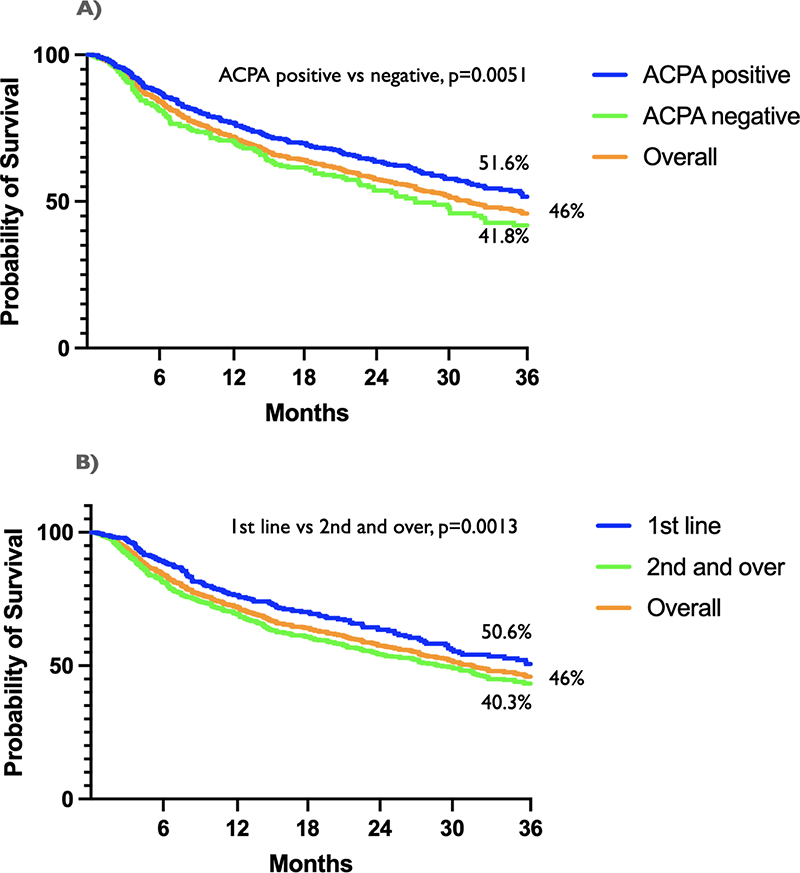
Figure 1. A) Retention rate in ACPA positive versus ACPA negative patients. B) Retention rate in first-line versus second or further lines of therapy.
Acknowledgements We thank the physicians and patients who participated in these studies.
Disclosure of Interests Martina Biggioggero Speakers bureau: Galapagos, Elisa Gremese: None declared, Serena Bugatti: None declared, Andreina Manfredi: None declared, Simone Parisi: None declared, Chiara Bazzani: None declared, Antonio Carletto: None declared, Carlo Garaffoni: None declared, Angelo Semeraro: None declared, Addolarata Corrado: None declared, Rosario Foti: None declared, Alberto Cauli: None declared, Francesca Romana Spinelli Speakers bureau: Amgen, AbbVie, Eli Lilly, Galapagos, Consultant of: Amgen, AbbVie, Eli Lilly, Galapagos, Florenzo Iannone: None declared, Ennio Giulio Favalli Speakers bureau: AbbVie, BMS, Celltrion, Galapagos, Janssen, Lilly, MSD, Novartis, Pfizer, UCB, Consultant of: AbbVie, BMS, Celltrion, Galapagos, Janssen, Lilly, MSD, Novartis, Pfizer, UCB.
Keywords: Registries, Targeted synthetic drugs, Rheumatoid arthritis
DOI: 10.1136/annrheumdis-2023-eular.4685