

Background Anti-citrullinated protein antibodies (ACPA) play a role in rheumatoid arthritis (RA) pathogenesis, and their presence is associated with disease severity. Moreover, high serum levels of ACPA in the preclinical phase are one of the major risk factors for RA progression. Consequently, detailed analysis of ACPApos B cells is required to disentangle the role of these cells in RA. Multiple intracellular signalling pathways are involved in functional B cell responses. NF-κB signalling is one of the prime regulators of B cell proliferation, differentiation and (auto)antibody production. Moreover, NF-κB activation leads to pro-inflammatory cytokine and chemokine production. JAK-STAT signalling is induced after activation of the B cell receptor (BCR) and is also involved in B cell proliferation and maturation. Targeting NF-κB or JAK-STAT signalling may advance our understanding of the mechanisms involved in the activation of ACPA-producing B cells. We make use of ACPApos B cell clones, since peripheral ACPApos cells are present at low frequencies in the peripheral blood.
Objectives To identify whether NF-κB or JAK-STAT signalling inhibition using small molecule inhibitors (SMIs) is effective in targeting functional responses of ACPApos B cell clones from RA patients, including survival, proliferation, differentiation, and antibody production.
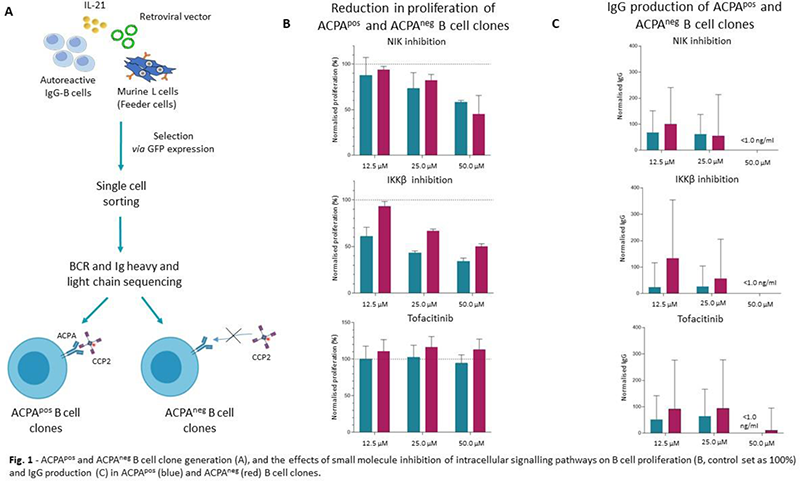
Methods Previously generated ACPApos and ACPAneg B cell clones [1] (see Figure 1A for method) were expanded and cultured with anti-CD40 and IL-21. Canonical and non-canonical NF-κB signalling were targeted by validated SMIs of Inhibitor of κB kinase β (IKKβi, canonical NF-κB signalling) and NF-κB inducing kinase (NIKi, non-canonical NF-κB signalling), respectively. Tofacitinib (a JAK1/JAK3 specific small molecule inhibitor) was used to target JAK-STAT signalling. Cell viability, proliferation and differentiation were evaluated by flow cytometry. Antibody production was measured by ELISA.
Results We observed a dose-dependent reduction in proliferation in ACPA-producing B cell clones treated with either IKKβi (12.5 µM IKKβi: 39.12±9.86% decrease, 25 µM IKKβi: 56.55±1.84% decrease, 50 µM IKKβi: 65.65±3.1% decrease; data normalised on DMSO) or NIKi (12.5 µM NIKi: 12.13±19.42% decrease, 25 µM NIKi: 26.63±17.37% decrease, 50 µM NIKi: 41.69±2.00% decrease; data normalised on DMSO) upon T cell-dependent stimulation with 2.25 µg/ml anti-CD40 and 1.0 ng/ml IL-21 (Figure 1B). Similarly, we observed a dose-dependent reduction in IgG production (Figure 1C). IKKβi treatment seemed to have a stronger effect than NIKi treatment on ACPApos B cell clones, while we did not observe a clear difference between IKKβi and NIKi in ACPAneg B cells (12.5 µM IKKβi: 6.90±5.38% decrease, 25 µM IKKβi: 33.32±2.15% decrease, 50 µM IKKβi: 50.11±3.15% decrease; 12.5 µM NIKi: 6.09±3.61% decrease, 25 µM NIKi: 17.83±6.53% decrease, 50 µM NIKi: 54.67±20.37% decrease; data normalised on DMSO). These effects were not caused by cytotoxic effects as cell viability was not affected by IKKβi or NIKi treatment. In contrast to the effects of IKKβi and NIKi treatment, tofactinib only had limited effects on ACPApos and ACPAneg B cell proliferation and IgG production. At present these results are being corroborated in freshly isolated ACPApos B lineage cells of RA patients.
Conclusion Our data point towards a critical role of the NF-κB signalling pathways in the functional responses of ACPA-producing B cells, whereas a limited role of JAK-STAT signalling was observed. Consequently, targeting NF-κB signalling may have beneficial effects in limiting (autoreactive) B cell responses in RA.
Reference [1] Germar K, et al. Generation and Characterization of Anti-Citrullinated Protein Antibody-Producing B Cell Clones From Rheumatoid Arthritis Patients. Arthritis Rheumatol. 2019 Mar;71(3):340-350.
Image/graph:
Acknowledgements: NIL.
Disclosure of Interests None Declared.
Keywords: Rheumatoid arthritis, Cell biology
DOI: 10.1136/annrheumdis-2023-eular.3542