

Background Upadacitinib (UPA) 15 mg QD is efficacious in patients with active ankylosing spondylitis (AS) exhibiting inadequate responses to nonsteroidal anti-inflammatory drugs (NSAIDs-IR).[1]
Objectives We determined the impact of UPA on the global metabolome in these patients.
Methods Baseline (BL), weeks 4, and 14 serum samples were obtained from a subset of patients enrolled in SELECT-AXIS1 (PBO, n = 59 out of 94; UPA, n = 59 out of 93). Patients met the modified New York AS criteria and had active disease at BL. Samples were processed using dansyl-12C-labeling kits or DmPA-12C-labeling kits for each chemical-group channel and analyzed by LC-MS–based metabolomics. Data analysis was performed using a commercial software and metabolite identification was done using a commercial reference database.[2] Tier 1 and 2 metabolites matched single compounds from a labeled metabolite library and a linked identity library, respectively. Tier 3 metabolites matched more than 1 compound. Differentially modulated metabolites (DMM) were identified using a mixed linear model measuring change from BL for each treatment group (selection criteria, absolute Log10 fold change ≥ 0.079, and false discovery rate ≤ 0.1). Pathway enrichment analyses were conducted using a dedicated software.[3] Relationships between metabolite level changes, and clinical changes were assessed by Pearson’s correlation.
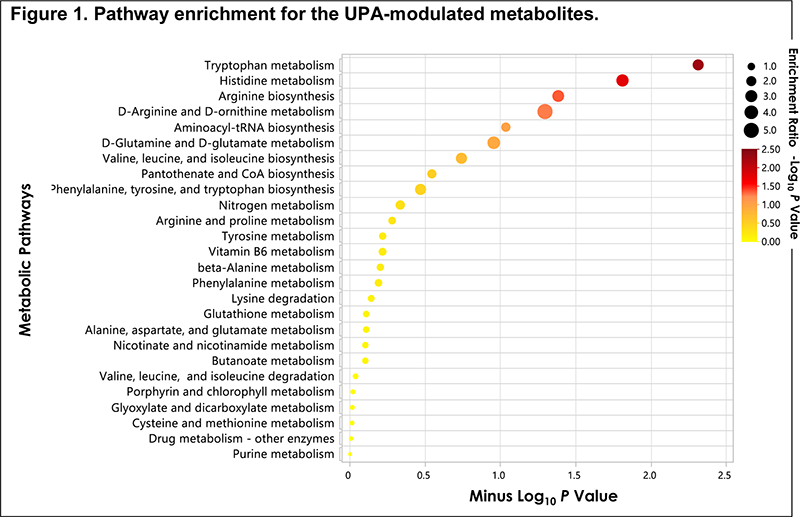
Results We detected a total of 8020 distinct metabolites (ie, ≥ 80% detectability across timepoints within each treatment group) across the global metabolome. From these 8020 metabolites, we identified 289 and 221 DMM at weeks 4 and 14 in the UPA group vs 32 and 89 in the PBO group, respectively. Most DMM in the UPA group (187 and 144 at weeks 4 and 14, respectively) belonged to the Tier 3 identification category, and 51 and 40 at weeks 4 and 14, respectively, belonged to the Tier 1 and 2 categories. Pathway enrichment suggests UPA affects the histidine and tryptophan biochemical pathways (Figure 1), consistent with increases in metabolites observed in the UPA group. Increases in histidine and tryptophan levels were significantly associated with an improvement in CRP levels and Ankylosing Spondylitis Disease Activity Score-CRP scores (P ≤.05). Increases in histidine levels were also associated with the improvement in MRI Spondyloarthritis Research Consortium of Canada Spine indices.
Conclusion This is the first comprehensive metabolome analysis of serum samples collected from patients with AS treated with UPA. Data highlight the promise of advanced metabolomics to elucidate therapeutic agent’s mode of action in AS. The putative effect of UPA on the tryptophan biochemical pathway is of particular interest because the tryptophan/kynurenine pathway is associated with chronic pain[4] and is perturbed toward the degradation of tryptophan in patients with AS.[5] We hypothesize that the increase in tryptophan and histidine may be related to the mode of action of UPA in NSAID-IR patients with AS.
References
Image/graph:
Acknowledgements Medical writing assistance, funded by AbbVie, was provided by Steve Page, PhD, and Lamara D. Shrode, PhD, CMPP™, of JB Ashtin.
Disclosure of Interests Thierry Sornasse Shareholder of: AbbVie, Employee of: AbbVie, Liang Li Shareholder of: Founder of Nova Medical Testing Inc. (NovaMT)., Consultant of: Meliomics Inc., Grant/research support from: Matching fund from NovaMT as part of a Mitacs PDF support and receives grant royalty payment from Bruker on a licensed QTOF Metabolomics Technology., Employee of: founder of Nova Medical Testing Inc. (NovaMT)., Shuang Zhao: None declared, Xiaohang Wang: None declared, Fang Cai Shareholder of: AbbVie, Employee of: AbbVie, Yingtao Bi Shareholder of: AbbVie, Employee of: AbbVie, In-Ho Song Shareholder of: AbbVie, Employee of: AbbVie, Stephanie Wichuk: None declared, Robert Lambert: None declared, Walter P Maksymowych Consultant of: AbbVie, Bristol Myers Squibb, Boehringer, Celgene, Eli Lilly, Galapagos, Janssen, Novartis, Pfizer, and UCB, Grant/research support from: AbbVie, Novartis, Pfizer, and UCB.
Keywords: -omics, Spondyloarthritis, Clinical trials
DOI: 10.1136/annrheumdis-2023-eular.2813