

Background ABBV-599 is a novel combination of upadacitinib (UPA), a reversible JAK inhibitor approved for the treatment of several autoimmune diseases, and elsubrutinib (ELS), a covalent BTK inhibitor being developed for the treatment of immune-mediated inflammatory diseases. The efficacy and safety profile of the monotherapies (UPA and ELS) and combination (ABBV-599) were evaluated in systemic lupus erythematous (SLE) in the global Phase 2 Study SLEek (NCT#03978520).
Objectives The analyses presented here characterize the pharmacokinetics of UPA and ELS as well as exposure-response relationships for efficacy at Week 24 and Week 48 and safety endpoints through Week 48.
Methods Phase 2 Study SLEek evaluated UPA 30 mg once-daily (QD), ELS 60 mg QD, ABBV-599 high dose (ELS 60 mg + UPA 30 mg) QD and ABBV-599 low dose (ELS 60 mg + UPA 15 mg) QD in a placebo-controlled manner. Pharmacokinetic, efficacy, and safety data from subjects with moderately to severely active SLE enrolled in the study were used to conduct the analyses (N = 341). UPA pharmacokinetics were described using a previously published population pharmacokinetic model[1] and ELS pharmacokinetics were characterized using Bayesian population pharmacokinetic analyses based on data from the healthy subjects, RA patients, and SLE patients. Exposure-response analyses for efficacy were conducted using logistic regression models to characterize the relationships for the monotherapies and combination average plasma concentration (Cavg) and the percentage of patients achieving SLE Responder Index-4 (SRI-4), SRI-4 and steroid dose ≤ 10 mg QD, and British Isles Lupus Assessment Group (BILAG)-Based Combined Lupus Assessment (BICLA) response at Week 24 and Week 48. Exposure-response analyses for safety were conducted using quartile plots and logistic regression modeling at or through Week 48.
Results The previously developed population pharmacokinetic model for UPA showed that UPA pharmacokinetics were comparable in subjects with SLE and RA. A one-compartment model with a proportional error model best described the pharmacokinetics of ELS.
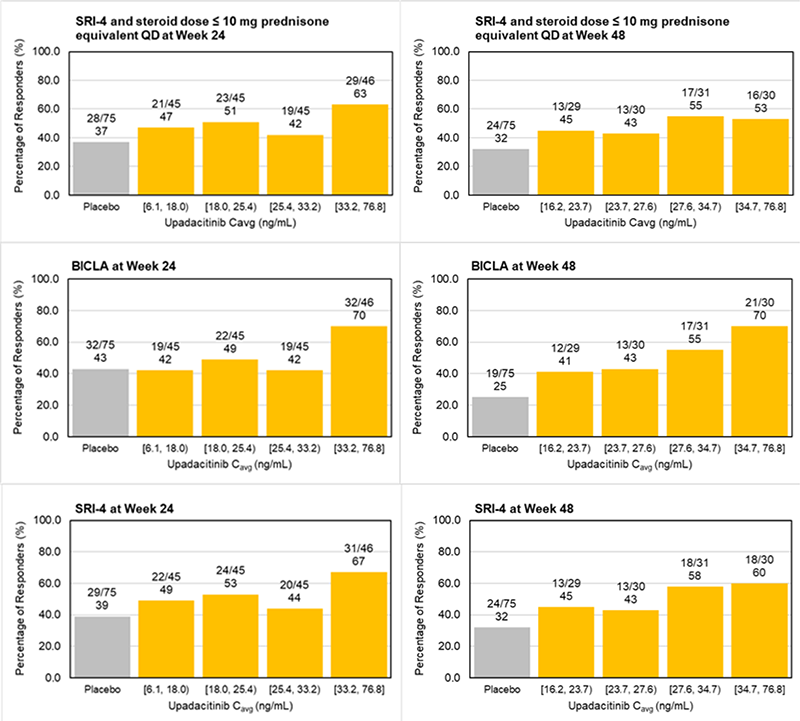
Exposure-response analyses for efficacy based on logistic regression modeling demonstrated that at Week 24 and Week 48, statistically significant relationships were observed between UPA Cavg and SRI-4, SRI-4 and steroid dose ≤ 10 mg QD, and BICLA (Figure 1). No exposure-response trends were observed for ELS Cavg and the evaluated efficacy endpoints during the Week 24 analysis. Inclusion of ELS Cavg in the logistic regression models did not improve the model performance. Simulations based on the logistic regression models showed that UPA 30 mg QD is predicted to provide 8% to 17% higher response rates across the evaluated endpoints compared to UPA 15 mg QD.
Exposure-response analyses for safety showed no clear trend for serious infections, lymphopenia, or neutropenia through Week 48. A shallow trend was observed for an increase in the percentage of subjects experiencing herpes zoster and a > 2 g/dL decrease in hemoglobin from Baseline at or through Week 48 with increasing upadacitinib Cavg.
Conclusion Population pharmacokinetic and exposure-response analyses demonstrated that plasma exposures associated with UPA 30 mg QD were efficacious in subjects with SLE, while maintaining an acceptable safety profile. Addition of ELS, as part of ABBV-599, was not estimated to provide any additional efficacy benefit over UPA alone.
Reference [1]Klunder et al. Population Pharmacokinetics of Upadacitinib Using the Immediate-Release and Extended-Release Formulations in Healthy Subjects and Subjects with Rheumatoid Arthritis: Analyses of Phase I–III Clinical Trials. Clinical Pharmacokinetics. 58, 1045–1058 (2019).
Figure 1. Graphical Assessment for Exposure-Response Relationships Between Upadacitinib Average Plasma Concentration and Evaluated Efficacy Endpoints at Week 24 (left) and Week 48 (right)
Cavg: average plasma concentration; QD: once-daily
Image/graph:
Acknowledgements: NIL.
Disclosure of Interests Sumit Bhatnagar Shareholder of: AbbVie, Employee of: AbbVie, Lucia Siovitz Shareholder of: AbbVie, Employee of: AbbVie, Alan Friedman Shareholder of: AbbVie, Employee of: AbbVie, Wei Liu Shareholder of: AbbVie, Employee of: AbbVie, Mohamed-Eslam Mohamed Shareholder of: AbbVie, Employee of: AbbVie.
Keywords: Systemic lupus erythematosus
DOI: 10.1136/annrheumdis-2023-eular.1312