

Background Complete renal response (CRR) defined by ACR/EULAR guidelines is a desirable clinical outcome associated with long-term preservation of kidney function in patients with lupus nephritis (LN) as demonstrated in clinical trials when achieved after 1-2 years. Urinary CD163 (uCD163), a biomarker that correlates with LN histologic inflammation, has been observed to predict clinical response to therapy[1]. Results previously reported on MISSION, a Phase 2 open-label study evaluating the safety and tolerability of zetomipzomib in active, proliferative LN, demonstrated clinically meaningful renal responses along with reductions in uCD163 and strong correlation with UPCR[2]. Decrease in uCD163 at early timepoints and its prediction in proteinuria response may increase probability of achieving a meaningful treatment effect.
Objectives Data from the MISSION Phase 2 study of zetomipzomib was used to evaluate uCD163 as a potential predictor of complete renal response (CRR) for patients with LN.
Methods In the MISSON Phase 2 study, patients with active proliferative LN (Class III or IV ± V) received 60 mg of zetomipzomib subcutaneously once weekly (first dose 30 mg) in addition to stable background therapy for 24 weeks. End-of-treatment (EOT) was at Week (W) 25, and end-of-study (EOS) occurred at W37. uCD163 data at W13 and W25 were used to model a predictive association with CRR* at W25 & W37, respectively. A logistic regression model and the Youden Index were used to generate Receiver Operating Characteristics (ROC) curves to help evaluate accuracy and identify optimal cut-off points to differentiate responders and non-responders.
*Complete Renal Response: UPCR ≤0.5, eGFR ≥60 mL/min/1.73m2 or no worsening of eGFR from baseline of ≥25%, prednisone (or equivalent) ≤10 mg and no use of prohibited medication.
Results Of the 21 patients enrolled in MISSION, 17 reached EOT and EOS. CRR was achieved in 35% and 41% of patients at W25 and W37. Anti-inflammatory potential of zetomipzomib was demonstrated by reduction of uCD163, which was strongly correlated with UPCR improvement at W13, W25 and W37 (Figure 1). For the thirteen patients who consented to urine biomarker analysis, baseline 24-hour UPCR was: mean=2.8 mg/mg, median=1.8 mg/mg, SD=3.3, range 0.93-13.4; uCD163: mean=1.7 mg/mmol, median=0.97 mg/mmol, SD=2.3, range 0.28, 8.9. ROC analysis results from the MISSION study suggests that LN patients with uCD163 values of ≤0.13 at W13 and ≤0.09 at W25 are more likely to achieve a CRR at W25 and W37 following treatment with zetomipzomib (Table 1).
Conclusion This post-hoc analysis of zetomipzomib data generated from the MISSION Ph2 study suggests that in patients with proliferative LN, uCD163 levels might predict CRR achieved up to 3 months later and could therefore potentially help to guide optimal therapy. Further evidence is needed from larger randomized LN trials with zetomipzomib to confirm the utility and limits of uCD163 as a predictive biomarker.
Reference [1]Mejia Vilet et al, JASN 2020; 2Parikh SV et al, ASN 2022.
Image/graph:
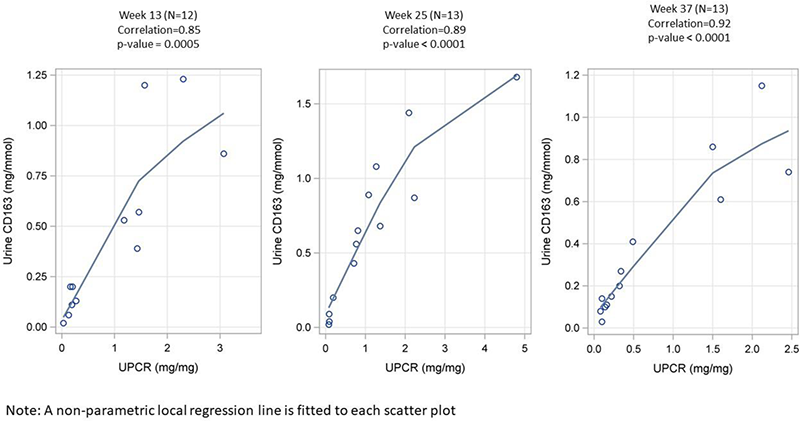
Figure 1. uCD163 and UPCR Correlation Plots
| Predictor | Outcome | N used |
Youden |
ROC AUC |
Sensitivity |
Specificity |
Predictive |
Predictive |
|---|---|---|---|---|---|---|---|---|
| uCD163 at Week 13 |
CRR at Week 25 |
12 |
uCD163 |
1.0 | 1.0 | 1.0 | 100% | 100% |
| uCD163 at Week 25 |
CRR at Week 37 |
13 | uCD163 |
0.8 | 0.6 | 1.0 | 100% | 80% |
| uCD163 |
0.8 | 0.75 | 67% | 86% |
*based on the criterion of smallest Δ=Sensitivity – Specificity.
Acknowledgements: NIL.
Disclosure of Interests Joan T Merrill Consultant of: AbbVie, Alexion, Alumis, Amgen, Astra Zeneca, Aurinia, Bristol Myers Squibb, EMD Serono, Genentech, Gilead, GlaxoSmithKline, Lilly, Merck, Pfizer, Provention, Remegen, Sanofi, UCB, and Zenas, Grant/research support from: Astra Zeneca, Bristol Myers Squibb, and GlaxoSmithKline, Richard Leff Shareholder of: Kezar Life Sciences, Consultant of: Kezar Life Sciences, Andrea Fan Shareholder of: Kezar Life Sciences, Employee of: Full time employee of Kezar Life Sciences, Kiruthi Palaniswamy Shareholder of: Kezar Life Sciences, Employee of: Full time employee of Kezar Life Sciences, Noreen Henig Shareholder of: Kezar Life Sciences, Employee of: Chief Medical Officer at Kezar Life Sciences, Steven Hua Shareholder of: Kezar Life Sciences, Employee of: Full time employee of Kezar Life Sciences.
Keywords: Biomarkers, Systemic lupus erythematosus, Clinical trials
DOI: 10.1136/annrheumdis-2023-eular.2695