

Background Belimumab (BLM) is a recombinant human IgG-1λ monoclonal antibody that inhibits B-cell activating factor. It is approved for the treatment of systemic lupus erythematosus (SLE). It is effective in reducing disease activity, flares, damage prevention and also as a steroid-sparing agent. A treat to target (T2T) approach in the care of SLE patients is important in terms of improving short and long-term outcomes.
Objectives To evaluate belimumab (BLM) effectiveness in SLE patients from a Spanish multicenter registry.
Methods A longitudinal retrospective multicenter cohort including SLE patients treated with belimumab from 18 Spanish rheumatology departments. Demographic, clinical and serological data were collected at baseline, 6, 12 and in the last visit available. Changes in SLEDAI-2K; LLDAS and DORIS 2021 states and global response according to physician criteria were compared between visits, as well as changes in damage and glucocorticoids used. T-test was used for numerical variables and the Fisher’s test for categorical variables.
Results 324 patients were included: 295 (91%) females with a mean (±SD) age of 42.4 (±12.9) years. Mean follow-up was 3,8 (±2.7) years and mean time with BLM was 2.7 (±2.4) years. At baseline, mean SLEDAI-2K was 10.4 (±5.25), 68.2% had elevated anti-double-stranded DNA (anti-dsDNA) antibodies and 69.8% had complement consumption. BLM was initiated concomitant to other DMARD in 67.9% (n=220) of patients.
Mean reduction in SLEDAI-2K score was 5.0 (± 5.1), 6.1 (± 5.5) and 7.13 (± 5.3) points at 6, 12 months and in the last visit, respectively (p<0.05 for all comparisons). Rates of achievement of LLDAS, DORIS and clinical response according to physician criteria, significantly increased from baseline to 6, 12 months, and to the last visit (Table 1). Anti-dsDNA antibodies and inflammatory markers (ESR, CRP), significantly decreased from baseline to 6, 12 months and in the last visit. Complements increased over the follow up but without statistical significance.
A total of 107 (45,9%) patients discontinued GC. At 6 months, 58.9% (n=155) of patients reduced the dose of GC with respect to baseline and 72.8% (n=131) of patients did it at the last visit.
Mean (±SD) prednisone dose was significantly reduce over the visits: 12.3 (±12.16); 7.42 (±5.36); 5.8 (±4.42) and 4.7 (±3.7) mg/day at baseline, 6 and 12 months and in the last visit, respectively. Median (IQR) SDI score at the end of the observation period did not change from baseline visit: 0 (0-1) and 0 (0-1), respectively (p=0.97). Neither were changes observed in the percentage of patients with damage between the beginning and the end of the observation period: at baseline 47.5% (n=152) patients presented damage and, in the last visit, 45.6% (n=99).
| Disease activity Mean (±SD) or number (%); n=324 | Baseline | 6 months | 12 months | Last visit |
|---|---|---|---|---|
| SLEDAI reduction; mean (±SD) | 10 (± 5.25) | 5.0 (± 5.1) * | 6.1 (± 5.5) * | 7.13 (± 5.3) * |
| LLDAS | 6 (1.9%) | 137(45.8%) * | 145 (62%) * | 177 (73.1%) * |
| DORIS | 6 (1.9%) | 72 (24%) * | 85 (36.3%) * | 127 (52.5%) * |
| Response according to physician | 212 (65.4%) * | 185 (57.1%) * | 165 (50.9%) * | |
| Number of swollen joints; mean (±SD) | 3.3 (±3.6) | 1.2 (±2.8) * | 0.69 (±1.95) * | 0.55 (±1.82) * |
| Prednisone dose (mg/day); mean (±SD) | 12.3 (± 12.16) * | 7.4 (± 5,36) * | 5.8 (± 4,42) * | 4.75 (±3.74) * |
SLEDAI: Systemic Lupus Erythematosus Disease Activity Index. cSLEDAI: clinical SLEDAI; LLDAS: Lupus Low Disease Activity State. DORIS: Definition of remission in SLE. *p<0.05
Image/graph:
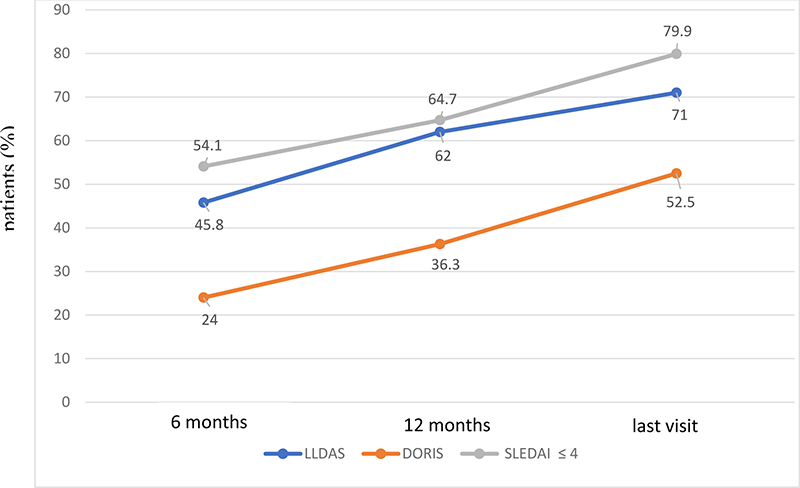
Figure 1. Rates of therapeutic targets attained by patients in treatment with Belimumab.
| 6 months | 12 months | Last visit | |
|---|---|---|---|
| Discontinued | 45 | 74 | 130 |
| Ongoing | 279 | 250 | 194 |
Conclusion Real-world data of SLE patients confirm belimumab efficacy in real world, reducing clinical and serological activity in the short and medium-term. Add-on therapy with BLM leads to high rates of LLDAS and DORIS at 6 months, that continue increasing over time. BLM has an important GC sparing effect and prevents organ damage accrual. All these data shows that BLM is useful to achieve the therapeutic goals of a T2T strategy.
REFERENCES:
NIL.
Acknowledgements: NIL.
Disclosure of Interests None Declared.
Keywords: Systemic lupus erythematosus, bDMARD, Real-world evidence
DOI: 10.1136/annrheumdis-2023-eular.3482