

Background Systemic lupus erythematosus (SLE) is a multi-system autoimmune disease characterized by aberrant expansion of B cells. The B cell-stimulating molecules, B cell activating factor/a proliferation-inducing ligand (BAFF/APRIL), are critical factors in promoting the proliferation and differentiation of B cells thus play a prominent role in the pathogenesis of SLE. There are two types of BAFF/APRIL-targeted biological agents being approved as an add-on therapy for patients with active SLE in China, including the BAFF/APRIL dual inhibitor, telitacicept and the BAFF inhibitor, belimumab. However, the differences of efficacy and safety between these two agents in real clinical practice has not been clarified.
Objectives To compare the differences of efficacy and safety between the two types BAFF/APRIL inhibitors telitacicept and belimumab in the treatment of active patients with SLE.
Methods Clinical features, laboratory data, SLE Disease Activity Index 2000 (SLEDAI-2K), treatment history, and adverse events (AEs) during the six months follow-up were collected. Propensity score based inverse probability of treatment weighting (IPTW) was used to reduce the selection bias.
Results Active SLE patients who received telitacicept (N=52) and belimumab (N=77) from 2019 to 2022 at our rheumatology department were retrospectively reviewed. No significant differences were observed in patient characteristics between the two groups after adjustment by IPTW. Patients who received telitacicept have a significantly higher decrease of SLEDAI-2K score and Physician’s Global Assessment (PGA) score at 24 weeks (p<0.05). Consistently, there was a higher elevation of complement (C) 3 and C4 levels at 4 and 12 weeks in telitacicept group (p<0.05). The telitacicept group acquire a higher frequency of primary efficacy renal response (PR) than belimumab at 12 weeks and no obvious differences were observed in complete renal response (CR) at 12 and 24 weeks according to the Belimumab International Lupus Nephritis Study (BLISS-LN) criteria. In addition, the improvement of anemia was better in the telitacicept group (p<0.05). Importantly, the telitacicept group had a lower incidence of AEs, including upper respiratory tract infections, diarrhea and leukopenia, than the belimumab group after IPTW (p<0.05).
Conclusion Telitacicept showed a better efficacy and safety than belimumab for active SLE patients in a single center clinical practice. Further investigations in a larger cohort are required to testify these findings.
References
Image/graph:
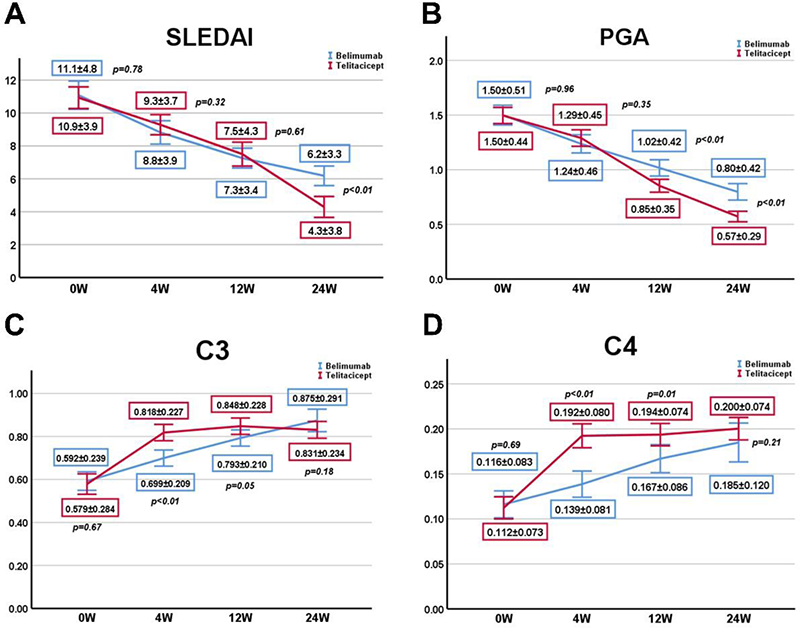
Figure 1. (A,B) The improvement of SLEDAI-2K and PGA scores after belimumab and telitacicept treatment at 4w, 12w, 24w by IPTW. (C,D)The elevation of complement (C) 3 and C4 levels after belimumab and telitacicept treatment at 4w, 12w, 24w by IPTW
Acknowledgements: NIL.
Disclosure of Interests None Declared.
Keywords: Treat to target, Real-world evidence, Systemic lupus erythematosus
DOI: 10.1136/annrheumdis-2023-eular.4616