

Background In our recently published working definition, we proposed that disease modification in systemic lupus erythematosus (SLE) requires ‘minimising disease activity with the fewest treatment-associated toxicities and slowing or preventing organ damage progression’.[1]
Objectives To apply the proposed non-renal disease modification criteria to SLE treatments.
Methods Based on a review of a selection of clinical trial (n=31) and observational (n=42) data publications for 16 SLE medications across different treatment classes and, the authors’ clinical experience, we evaluated their disease modification potential per the non-renal disease activity and organ damage criteria at three time points (Table 1).[1]
Results Nine of the 16 SLE treatments met at least one disease modification criterion across all time points (Table 1). Hydroxychloroquine improved overall survival at >5 years, suggesting long-term disease modification, but with data per specific criteria lacking.[2] Belimumab met two of the three disease modification criteria at the first two time points, but was the only treatment to meet criteria at >5 years based on the current literature. While steroids unambiguously decrease disease activity at early timepoints, at doses higher than 7.5 mg/day the negative impact on damage accrual might hamper their disease modification potential >5 years.
Conclusion This is the first attempt to determine which of the currently used treatments in patients with SLE fulfil the disease modification criteria at different time points. Belimumab and hydroxychloroquine showed evidence of disease modification up to and beyond 5 years; evidence for many other SLE therapies was incomplete, particularly at >5 years of follow up. The use of multiple agents, differences in study designs, patient populations, and definitions of treatment response pose challenges in categorising treatments as disease modifiers. Future studies will evaluate the minimum number of criteria required to designate disease modification at each of the three time points so that disease modification can be considered in the care of patients with SLE and in SLE trial design.
References
Image/graph:
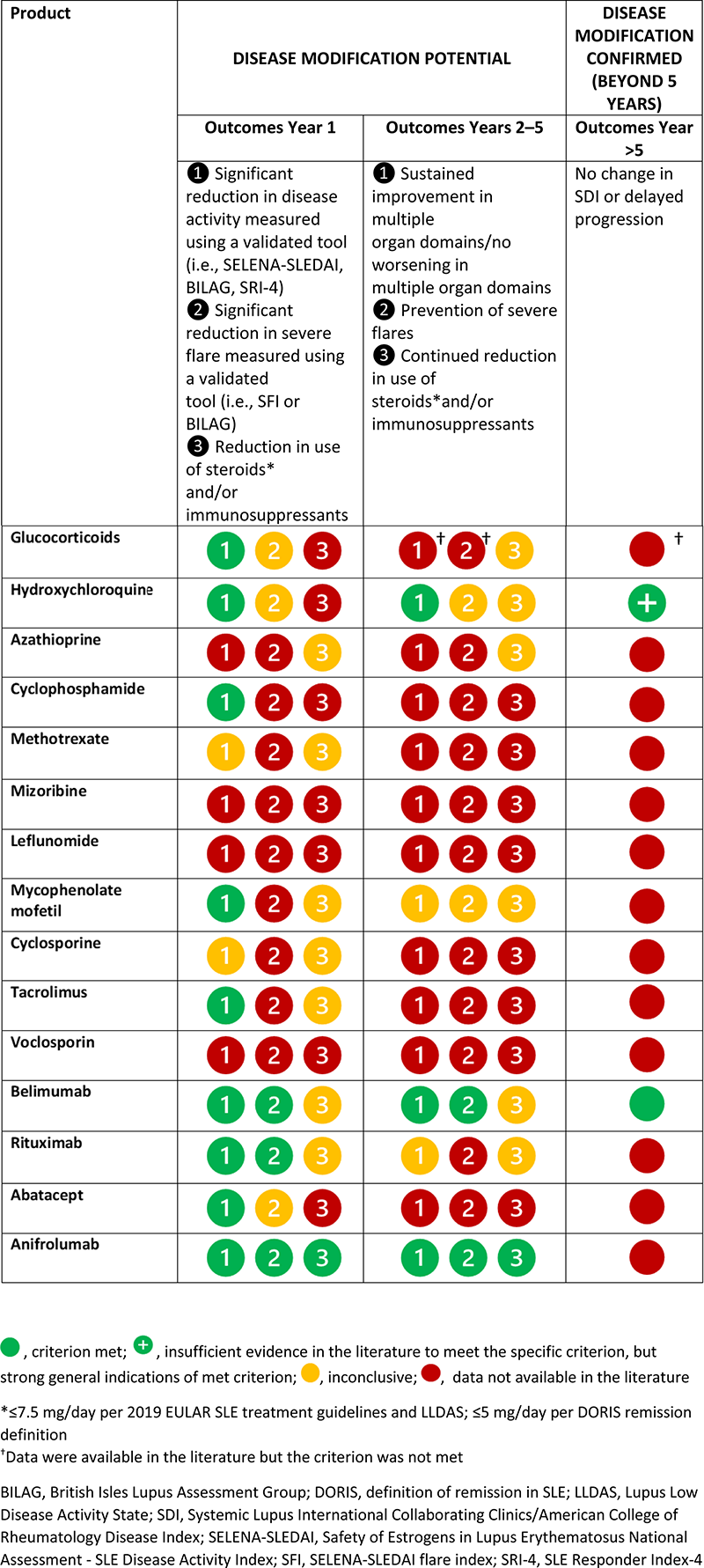
Table 1. Application of the proposed matrix for non-renal immunoinflammatory and organ damage disease modification criteria
Acknowledgements This review was funded by GSK. Medical writing support was provided by Casmira Brazaitis, PhD, and Cristina Teles, PhD, Fishawack Indicia Ltd, UK, part of Fishawack Health, and Hannah Jedrey, PhD, and Meer Basharat, PhD, of TVF Communications Ltd, UK, and was funded by GSK.
Disclosure of Interests Anca Askanase Consultant of: AbbVie, Amgen, AstraZeneca, Aurinia, BMS, Celgene, Eli Lilly, Idorsia, Janssen, Genentech, GSK, Mallinckrodt, Pfizer and UCB, Richard Furie Speakers bureau: AstraZeneca, Genentech/Roche, Consultant of: GSK, Aurinia, AstraZeneca, BMS, Genentech/Roche, Grant/research support from: GSK, Aurinia, AstraZeneca, BMS, Genentech/Roche, Maria Dall’Era Consultant of: GSK, AstraZeneca, Aurinia, Pfizer, Janssen, Andrew Bomback Consultant of: Alexion, Principio, Calliditas, Aurinia, Catalyst, Travere, GSK, Visterra, Silence, Novo Nordisk, Otsuka, ChemoCentryx, Novartis, Andreas Schwarting Speakers bureau: GSK, AstraZeneca, Pfizer, Novartis, Roche, Janssen, Boehringer Ingelheim, Grant/research support from: GSK, Pfizer, AbbVie, Roche, Novartis, AstraZeneca, Ming-hui Zhao Consultant of: GSK, AstraZeneca, Novartis, Kira, Ian N. Bruce Speakers bureau: GSK, AstraZeneca, UCB, Consultant of: GSK, UCB, Eli Lilly, BMS, Merck Serono, Aurinia, IL-TOO, Grant/research support from: National Institute for Health Research (NIHR), GSK, Genzyme/Sanofi, MUNTHER KHAMASHTA Shareholder of: GSK, Employee of: GSK, Bernard Rubin Shareholder of: GSK, Employee of: GSK, Angela Carroll Shareholder of: GSK, Employee of: GSK, Mark Daniels Shareholder of: GSK, Employee of: GSK, Roger Levy Shareholder of: GSK, Employee of: GSK, Ronald van Vollenhoven Speakers bureau: AbbVie, Galapagos, GSK, Janssen, Pfizer, UCB, Roche, Consultant of: AbbVie, AstraZeneca, Biogen, Biotest, BMS, Galapagos, Gilead, Janssen, Pfizer, Sanofi, Servier, UCB, Vielabio, Grant/research support from: Pfizer, Roche, Murray B Urowitz Speakers bureau: GSK, AstraZeneca, Consultant of: GSK, AstraZeneca, Grant/research support from: GSK.
Keywords: Organ damage, Kidneys, Outcome measures
DOI: 10.1136/annrheumdis-2023-eular.3052