

Background Systemic Lupus Erythematosus (SLE) is characterized by great clinical heterogeneity. Its complex pathogenesis has not been fully understood and factors influencing the development of different clinical phenotypes are still to unveil. A better understanding of these aspects could help in identifying new potential biomarkers and possible therapeutic targets to manage different disease subsets. Interleukin-10 (IL-10) has two opposite effects: it inhibits pro-inflammatory cytokine production but can also exert stimulatory effects by promoting B cell growth and differentiation [1-3]. Consequently, follicular and extrafollicular IL-10 producing B Helper T cells could play a pathogenic role. In SLE patients an increased frequency of circulating extrafollicular CCR6+ B Helper T cells have been reported especially in patients with active disease [1,4]. CRR6+ B helper T cells produce IL-10 and spontaneously induce IgG in SLE patients, including the pathogenic anti-dsDNA autoantibody [4].
Objectives Our aim was to evaluate the role of IL-10 -secreting cells in the pathogenesis of SLE and to highlight differences in the expression of these cells in different disease stages and clinical phenotypes.
Methods We analyzed the percentages of IL-10-producing lymphocytes either ex vivo (a standard negative control) or after brief stimulation with PMA and ionomycin, which activates all T-cells to produce cytokines. Surprisingly, 4 patients had very high levels of IL-10 production ex vivo, while the remaining patients produced some IL-10 only upon stimulation, similarly to healthy controls. We then divided our population into 2 groups according to disease activity: patients with SLEDAI ≥ 4 displayed significantly higher IL-10 production than inactive patients. To assess which cells were responsible for spontaneous IL-10 production, we analyzed the phenotype of IL-10+ cells. Spontaneously IL-10 secreting lymphocytes were mainly CD4+ helper T-cells that expressed high levels of CCR6, CXCR5 and IL-7R.
Results We looked at the percentage frequency of IL-10 -producing cells out of total lymphocytes under two conditions: ex vivo and after stimulation with PMA and ionomycin. Surprisingly, 4 patients had high levels of IL-10 production ex vivo, while the remaining patients produced IL-10 only following stimulation as expected. We then divided the test population into two groups according to disease activity: patients with SLEDAI ≥ 4 displayed significantly higher IL-10 levels than inactive patients. To assess which cells were responsible for IL-10 production, we analyzed the percentage of CD4 and CD8 cells in two populations of lymphocytes, those that expressed IL-10 and those that did not (Figure 1). Spontaneosly IL-10 secreting cells were mainly CD4+ helper T-cells that expressed high levels of CCR6, CXCR5 and IL-7R.
Conclusion IL-10 was reported to be spontaneously produced by B-cells and monocytes, but this is to our knowledge the first evidence that also T-cells spontaneously produce Il.-10 in SLE. IL-10 levels were higher in active disease and produced mainly by B helper T-cells, suggesting that it stimulates autoreactive B cells to produce pathogenic autoantibodies: the ex vivo production of IL-10 by B helper T cells in selected patients suggest that they are continuously activated by autoantigens and contribute to the aberrant production of autoantibodies. However, further analysis are needed to better characterize the spontaneously IL-10 producing lymphocytes in these patients.
References
Image/graph:
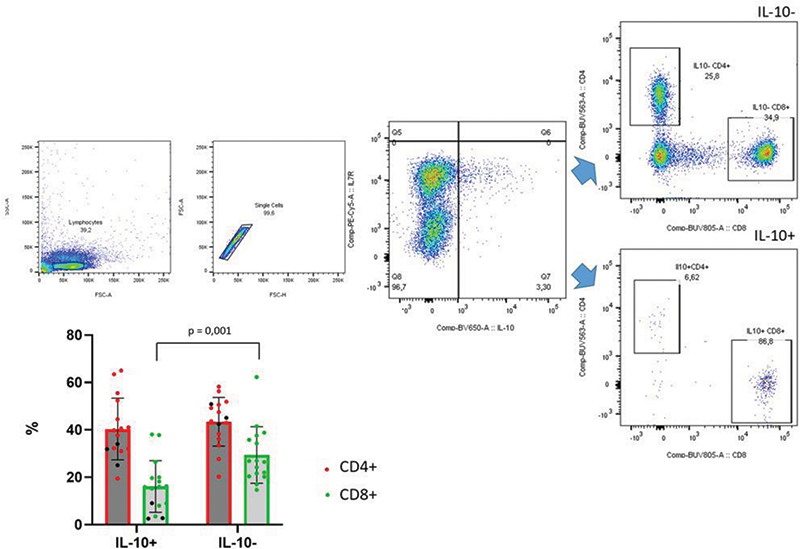
Figure 1. CD4 and CD8 cells in IL10+ and IL10- T cells
Acknowledgements: NIL.
Disclosure of Interests None Declared.
Keywords: Cell biology, Adaptive immunity, Systemic lupus erythematosus
DOI: 10.1136/annrheumdis-2023-eular.1646