

Background An international initiative is underway to develop a revised systemic lupus erythematosus (SLE) organ damage index (SDI) using a multi-phase process that is supported by the SLE International Collaborating Clinics (SLICC), American College of Rheumatology (ACR) and Lupus Foundation of America (LFA). The first two phases (evaluating the construct of damage and item generation through literature review and an international Delphi exercise) were previously reported [1,2], and 220 candidate items were generated.
Objectives To report the initial reduction phase results of a five-phase process for developing a revised SDI.
Methods An international two-part Delphi exercise asked SLE experts and patients to rate 220 items for how appropriate they were for inclusion in a revised SDI on a 9-point Likert scale (where 1 = not at all appropriate and 9 = completely appropriate) considering the new construct of damage in SLE [1]. The definitions of items in plain English were provided for the patient representatives. In the first part of the item reduction Delphi round, participants were asked to suggest any additional items. In the subsequent Delphi round, participants were presented their own rating, median scores and 25th-75th percentiles for each item. They were given the opportunity to revise their rating. Items with ratings less than or equal to 4.0/ 9.0 were considered for removal.
Results A total of 143 out of 146 participants who completed the first round Delphi for the item generation were invited for the item reduction rounds. Completion rates for the second and third Delphi rounds for the item reduction were 95% and 91.7% respectively. Appropriateness rating scores included the full range of scoring possibilities from 1 to 9. Participants suggested 53 potentially new items, however only 6 were retained, as others were duplicates or invalid.
226 items were presented to re-rate for appropriateness. There was stability in terms of summary median scores and 25th-75th percentiles for each item. Thirty-six items had a median score of less than or equal to 4. Figure 1 shows the number of candidate items identified at different stages of the project. All original SDI items were retained with group median appropriateness scores of 7, 8 and 9; the exceptions were upper gastrointestinal tract surgery (median appropriateness score 5) and osteomyelitis (median appropriateness score 5).
Image/graph:
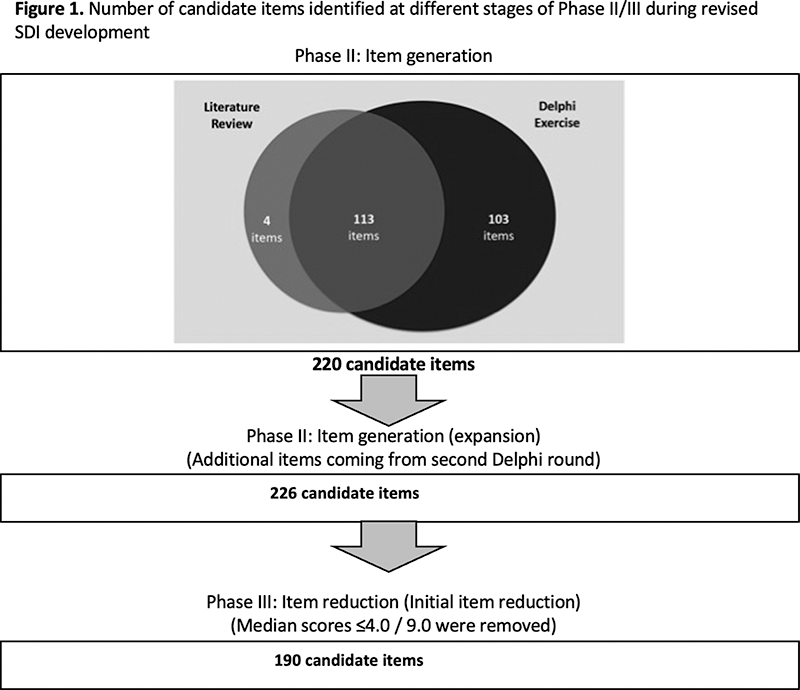
Conclusion The item reduction phase using Delphi methods reduced the candidate items for consideration. All original 1996 SDI [3] items were retained in this step. Further steps are now underway to refine and rationalize this candidate list.
References
Acknowledgements We thank the revised SDI collaborators for their valuable input. The project is supported by LFA, SLICC and ACR.
Disclosure of Interests Burak Kundakci: None declared, Megan Barber Consultant of: AbbVie, AstraZeneca, GSK, Janssen, Sanofi-Genzyme, Ann E Clarke Speakers bureau: AstraZeneca, GSK, Consultant of: BMS, Grant/research support from: GSK, Sindhu Johnson: None declared, Ian N. Bruce Speakers bureau: GSK, Astra Zeneca, Janssen.
Consultant of: GSK, Astra Zeneca, Aurinia, Lilly.
Grant/research support from: GSK, Astra Zeneca, Janssen.
Keywords: Organ damage, Systemic lupus erythematosus
DOI: 10.1136/annrheumdis-2023-eular.803