

Background The efficacy and safety of upadacitinib (UPA) in patients (pts) with psoriatic arthritis (PsA) and inadequate response to ≥1 biologic disease-modifying antirheumatic drug (bDMARD-IR) have been demonstrated up to 56 and 104 weeks of treatment [1,2].
Objectives To assess the long-term safety and efficacy of UPA in bDMARD-IR pts with PsA through 152 weeks of treatment in the SELECT-PsA 2 study.
Methods Pts were randomized to receive UPA 15 or 30 mg once daily (QD), or placebo (PBO) for 24 weeks, followed by blinded switch to UPA 15 or 30 mg QD for pts initially randomized to PBO. After 56 weeks, pts continued their assigned dose up to 152 weeks in an open-label extension (OLE). Following approval of the 15 mg QD dose, the protocol was amended and all pts on UPA 30 mg QD were switched to the approved dose (earliest switch at week 116). Endpoints assessed at 152 weeks included: % of pts achieving 20/50/70% improvement in American College of Rheumatology (ACR) criteria, minimal disease activity (MDA), and 75/90/100% improvement in Psoriasis Area and Severity Index, as well as patient-reported outcomes. Efficacy data were evaluated using non-responder imputation (NRI) and as-observed (AO) data (binary endpoints), and mixed effect model for repeated measures (continuous endpoints). Treatment-emergent adverse events (TEAEs) through 3 years (cut-off date, 05 May 2022) are also reported.
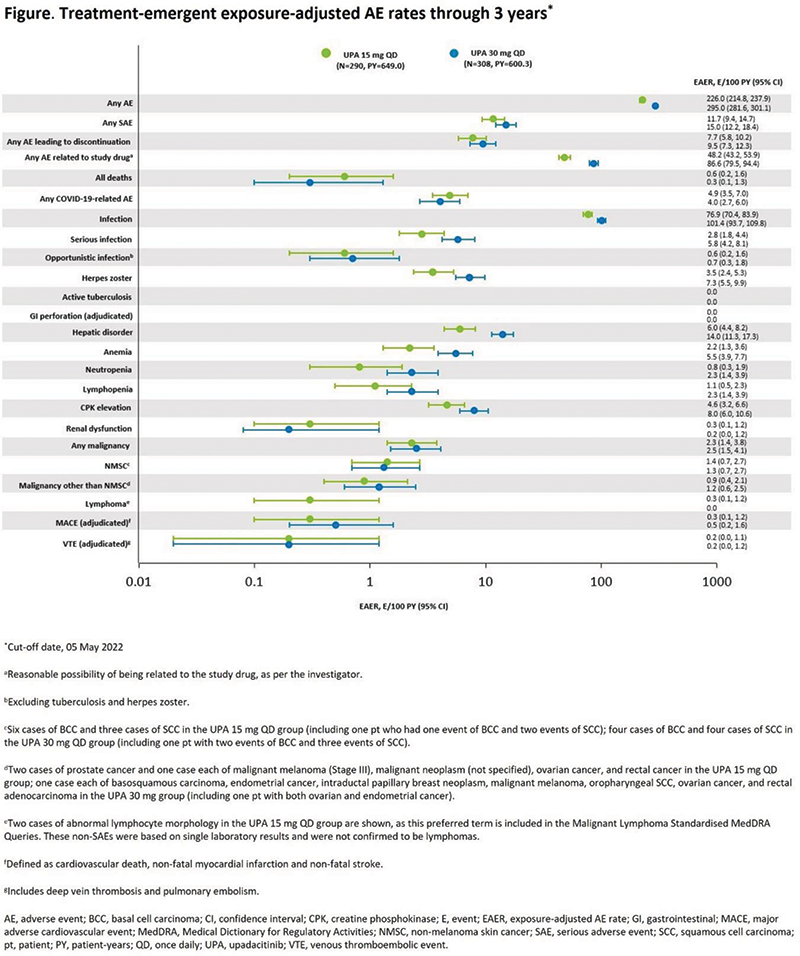
Results Of the 641 pts randomized to receive UPA 15 or 30 mg QD, or PBO followed by UPA 15 or 30 mg QD (n=211, 218, 106, and 106, respectively), 478 entered the OLE. Improvements in efficacy outcomes with UPA observed at 561 and 1042 weeks were maintained through the OLE, with ACR20 achieved by 43–51% (NRI) and 79–87% (AO) of pts across treatment arms at week 152. The highest % of MDA responders at week 152 (31% [NRI] and 44% [AO]) were in the continuous UPA 15 mg arm (Table 1). UPA was well tolerated through 152 weeks, with no new safety signals identified. The overall exposure-adjusted event rates for any TEAE were higher for UPA 30 vs 15 mg (295 vs 226 events/100 PY, respectively [Figure]).
Conclusion Observed improvements in the signs and symptoms of PsA with UPA were maintained through 3 years of treatment in bDMARD-IR pts. The long-term safety profile of UPA in this treatment-refractory population was consistent with its known safety profile across indications.
References
Image/graph:
| Endpoint | UPA 15 mg QD (n=211) | UPA 30 mg QDa (n=218) | PBO/UPA 15 mg QD (n=106) | PBO/UPA 30 mg QDa (n=106) | ||||
|---|---|---|---|---|---|---|---|---|
| Proportion of patients (%) | NRI | AO | NRI | AO | NRI | AO | NRI | AO |
| ACR20/50/70 | 51/39/24 | 81/60/36 | 49/35/22 | 79/56/34 | 43/29/20 | 87/60/40 | 43/28/18 | 79/53/36 |
| MDA | 31 | 44 | 25 | 41 | 16 | 41 | 19 | 37 |
| PASI75/90/100 | 43/31/20 | 66/46/30 | 41/36/29 | 72/61/48 | 45/34/23 | 79/61/42 | 41/35/30 | 75/63/55 |
| Resolution of enthesitis (LEI=0)b | 38 | 69 | 36 | 68 | 24 | 76 | 25 | 62 |
| Resolution of dactylitis (LDI=0)c | 49 | 94 | 50 | 100 | 34 | 100 | 28 | 100 |
| Change from BL | Mixed effect model for repeated measuresmean (95% confidence interval) | |||||||
| Health Assessment Questionnaire – Disability Index | -0.4 |
-0.5 |
-0.5 |
-0.3 |
||||
| Bath Ankylosing Spondylitis Disease Activity Indexd | -2.6 |
-2.0 |
-2.3 |
-2.5 |
||||
| Ankylosing Spondylitis Disease Activity Scored | -1.4 |
-1.1 |
-1.0 |
-1.2 |
||||
aFollowing a protocol amendment, all pts on UPA 30 mg QD switched to UPA 15 mg QD (earliest switch at week 116).
bBL LEI >0.
cBL LDI >0.
dAmong pts with psoriatic spondylitis at BL, as determined by the investigator.
ACR20/50/70, 20/50/70% improvement in American College of Rheumatology Criteria; AO, as observed; BL, baseline; LDI, Leeds Dactylitis Index; LEI, Leeds Enthesitis Index; MDA, minimal disease activity; NRI, non-responder imputation; PASI75/90/100, 75/90/100% improvement in Psoriasis Area and Severity Index; PBO, placebo; pt, patient; QD, once daily; UPA, upadacitinib.
Acknowledgements AbbVie funded this study; contributed to its design; participated in data collection, analysis, and interpretation; and participated in the writing, review, and approval of the abstract. AbbVie and the authors thank all study investigators for their contributions and the patients who participated in this study. No honoraria or payments were made for authorship. Medical writing support was provided by Katerina Betsista, MD, of 2 the Nth (Cheshire, UK), and was funded by AbbVie.
Disclosure of Interests Philip J Mease Grant/research support from: AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squib, Celgene, Galapagos, Genentech, Gilead, GlaxoSmithKline, Janssen, Lilly, Novartis, Pfizer, Sun Pharma, and UCB, Arathi Setty Employee of: AbbVie, and may own stock or options, Kim Papp Speakers bureau: AbbVie, Akros, Allergan, Almirall, Amgen, Bausch Health, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Dermavant, Dermira, Eli Lilly and Company, Galderma, Genentech/Roche, Janssen, Kyowa Kirin, LEO, Meiji, Merck Sharp & Dohme, Novartis, Pfizer, Regeneron, Sanofi Genzyme, Sienna Pharmaceuticals, Sun Pharma, Takeda, UCB, and Valeant, Consultant of: AbbVie, Akros, Allergan, Almirall, Amgen, Arcutis, Avillion, Bausch Health, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Dermavant, Dermira, Eli Lilly and Company, Galderma, Genentech/Roche, GlaxoSmithKline, Janssen, Kyowa Kirin, LEO, Meiji, Merck Sharp & Dohme, Novartis, Pfizer, Regeneron, Sanofi Genzyme, Sienna Pharmaceuticals, Sun Pharma, Takeda, UCB, and Valeant, Filip van den Bosch Speakers bureau: AbbVie, Bristol Myers Squibb, Celgene, Eli Lilly, Galapagos, Janssen, Merck, Novartis, Pfizer, Sanofi, and UCB, Consultant of: AbbVie, Bristol Myers Squibb, Celgene, Eli Lilly, Galapagos, Janssen, Merck, Novartis, Pfizer, Sanofi, and UCB, Shigeyoshi Tsuji Speakers bureau: AbbVie, Eli Lilly, Janssen, Novartis, and UCB, Consultant of: AbbVie, Eli Lilly, Janssen, Novartis, and UCB, Grant/research support from: AbbVie, Eli Lilly, Janssen, Novartis, and UCB, MAURO KEISERMAN Speakers bureau: AbbVie, Bristol Myers Squibb, Celgene, Eli Lilly, Roche, Novartis, Pfizer, Janssen, and UCB, Grant/research support from: AbbVie, Bristol Myers Squibb, Celgene, Eli Lilly, Roche, Novartis, Pfizer, Janssen, and UCB, Kyle Carter Employee of: AbbVie, and may own stock or options, Reva McCaskill Employee of: AbbVie, and may own stock or options, Erin McDearmon-Blondell Employee of: AbbVie, and may own stock or options, Peter Wung Employee of: AbbVie, and may own stock or options, William Tillett Speakers bureau: AbbVie, Amgen, Celgene, Eli Lilly, Janssen, Novartis, Pfizer, and UCB, Consultant of: AbbVie, Amgen, Celgene, Eli Lilly, MSD, Novartis, Ono Pharma, Pfizer, and UCB, Grant/research support from: AbbVie, Celgene, Janssen, Eli Lilly, Pfizer, and UCB.
Keywords: Psoriatic arthritis, Targeted synthetic drugs, Safety
DOI: 10.1136/annrheumdis-2023-eular.2513