

Background: The efficacy of Janus Kinases inhibitors (JAKi) in patients with rheumatoid arthritis (RA) has been univocally shown. However, the available information about comparative drug retention rates (DRR) of different JAKi is heterogeneous.
Objectives: The scope of the present study was to compute and compare DRR of different JAKi in a large cohort of RA patients.
Methods: Patients with RA followed-up at our Center treated with at least one JAKi were retrospectively identified. DRR of each JAKi were computed at 24 months. The association of baseline features with drug persistence was tested. Variations of DAS28-CRP and CDAI scores between baseline and 12 months were analyzed.
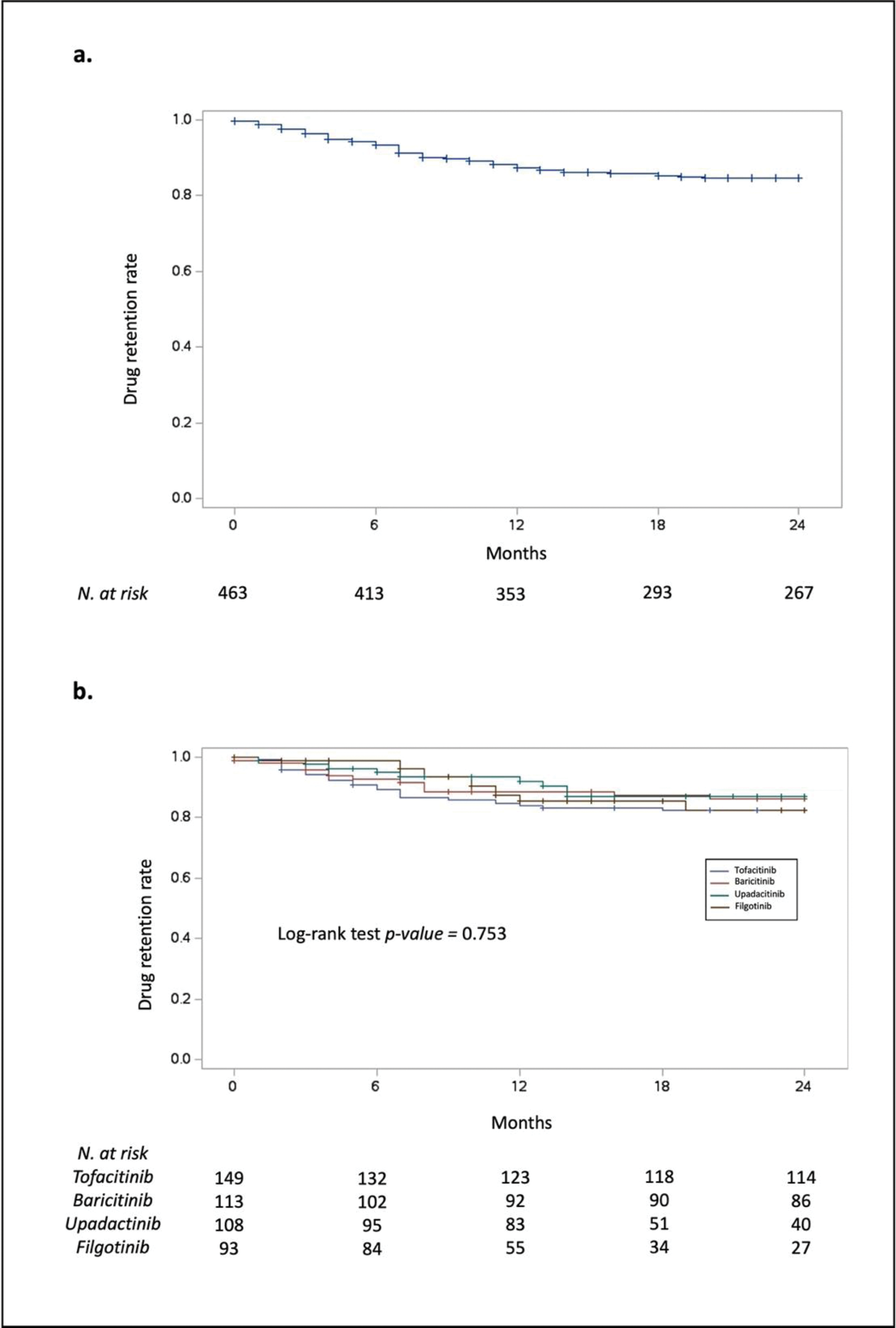
Results: We identified 365 patients that received one or more treatment with a JAKi, accounting for a total of 463 therapy courses. Demographic, disease, and treatment features of the study population are shown in Table 1. JAKi were started after a previous biologic DMARD in 264 (57%) cases. Tofacitinib was the most prescribed JAKi (n=149, 32%), followed by baricitinib (n=113, 25%), upadacitinib (n=108, 23%), and filgotinib (n=93, 20%). A total of 81 and 17 patients were treated with two and three different JAKi, respectively. The mean duration of treatment was 24 ± 17 months, up to a maximum of 70 months. At 24 months, the overall DRR (Figure 1a) was 86%. In this timeframe, inefficacy was the most common reason for JAKi discontinuation (72%), followed by safety concerns (cardiovascular risk factors in 12% of cases, cancer risk factors 5%). Less common causes were infections (5%), gastrointestinal intolerance (5%), and neutropenia (1%). DRRs were not significantly different across different JAKi when tested with the log-rank test (Figure 1b). Among the baseline features reported in Table 1, the only predictor of early treatment discontinuation was a previous treatment with a biologic DMARD (OR 1.96, 95% CI 1.32-2.92; p-value < 0.001). There was a significant reduction in both DAS28-CRP (4.5 ± 1.2 vs 2.5 ± 1.2; p-value < 0.001) and CDAI (20 ±9 vs 6 ± 7; p-value < 0.001) when comparing baseline values to those recorded at month 12 of treatment.
Conclusion: In our large, monocentric cohort, 24-month drug retention rate for JAKi was overall greater than 80%. No significant differences in retention were found among the different molecules. Persistence was lower in patients who had previously been treated with other biologic disease modifying anti-rheumatic drugs.
Demographic, disease, and treatment features of the study population according to each Janus Kinases inhibitor treatment course.
Table 1.
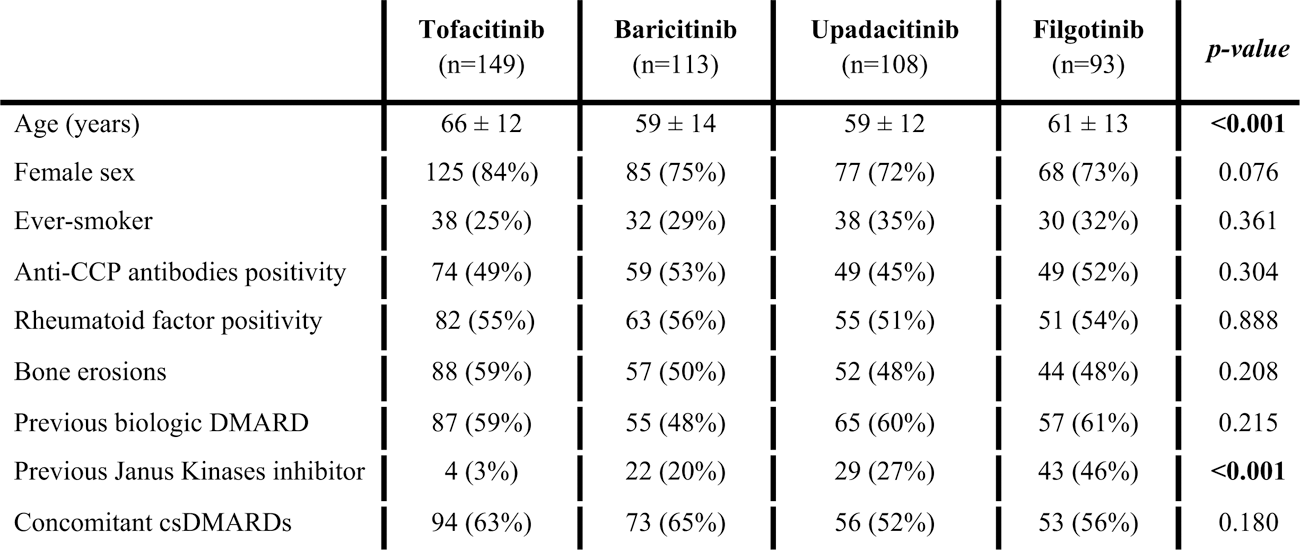
Continuous and categorial variable are expressed as mean ± standard deviation and absolute number (%), respectively. ANOVA and Chi Square tests were used to compare continuous and categorial variable, respectively.
CCP, citrullinated cyclic peptide; cs, conventional synthetic; DMARD, conventional synthetic disease modifying antirheumatic drugs.
Drug retention rates of Janus Kinases inhibitors in our cohort, both as a class (panel a) and grouped according to the specific drug (panel b).
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Nicola Farina: None declared, Alessandro Tomelleri: None declared, nicola boffini Abbvie, Eli Lilly, Galapagos, Pfizer., Adriana Cariddi: None declared, Stefania Laura Calvisi: None declared, Naomi Viapiana: None declared, Elena Baldissera Abbvie, Eli Lilly, Galapagos, Pfizer., Marco Matucci-Cerinic Abbvie, Eli Lilly, Galapagos, Pfizer., Lorenzo Dagna Abbvie, Eli Lilly, Galapagos, Pfizer.