

Background: JAK inhibitors (JAKi)[1] offer alternatives for the treatment of chronic inflammatory diseases, allowing improve outcomes of patients who have failed other previous treatments, reducing the dose of corticosteroids, with an adequate profile of safety.
Objectives: To describe clinical-epidemiological characteristics of patients who have required treatment with JAKi and to analyze the complications derived from their use.
Methods: We performed a descriptive observational study of all patients treated with JAKi in our center until December 31, 2023. The following variables are described: diagnosis, sex, average age at diagnosis and at the start of treatment, time delay between both dates, cardiovascular risk factors (CVRF) and toxic habits, adverse events (AE) due to JAKi (infectious/thrombotic/neoplastic), prescribed medications and reason for discontinuation and remission of the disease at last consultation.
Results: 71 patients were included (62% women). Tofactinib (38%), Upadactinib (34%) and Baricitinib (28%) were used; filgotinib is not authorized by the pharmacy of our center. The diagnoses of the patients were: Rheumatoid arthritis (46,5%), psoriasic arthritis (19,7%), axial spondyloarthritis (12,7%), seronegative arthritis (9,9%), enteropathic spondyloarthritis (2,8%), systemic lupus erithematosus (2,8%), inflammatory myophaty (2,8%), aortitis (1,4%) and juvenile idiophatic arthritis (1,4%). Mean age at the start of treatment was 54.34±8.52 years with a delay between the diagnosis of the disease and the start of JAKi of 22.9±19.3 months. 86% received vaccine against herpes zoster (HZ), 5.6% had previously suffered from HZ. 5% had serious infections prior to JAKi that had required hospitalization (3 pneumonias - one of them due to tuberculosis - and 1 episode of septic thrombophlebitis of the cavernous sinus). 78.9% had CVRF (dyslipidemia in 54.9% and high blood preassure in 33.8%). 42.3% had been smokers. 9.9% had established cardiovascular disease (atherosclerosis 85.7%) and 8.5% had presented a previous thrombotic episode (4 heart attacks, 2 pulmonary thromboembolisms and 1 ischemic colitis).
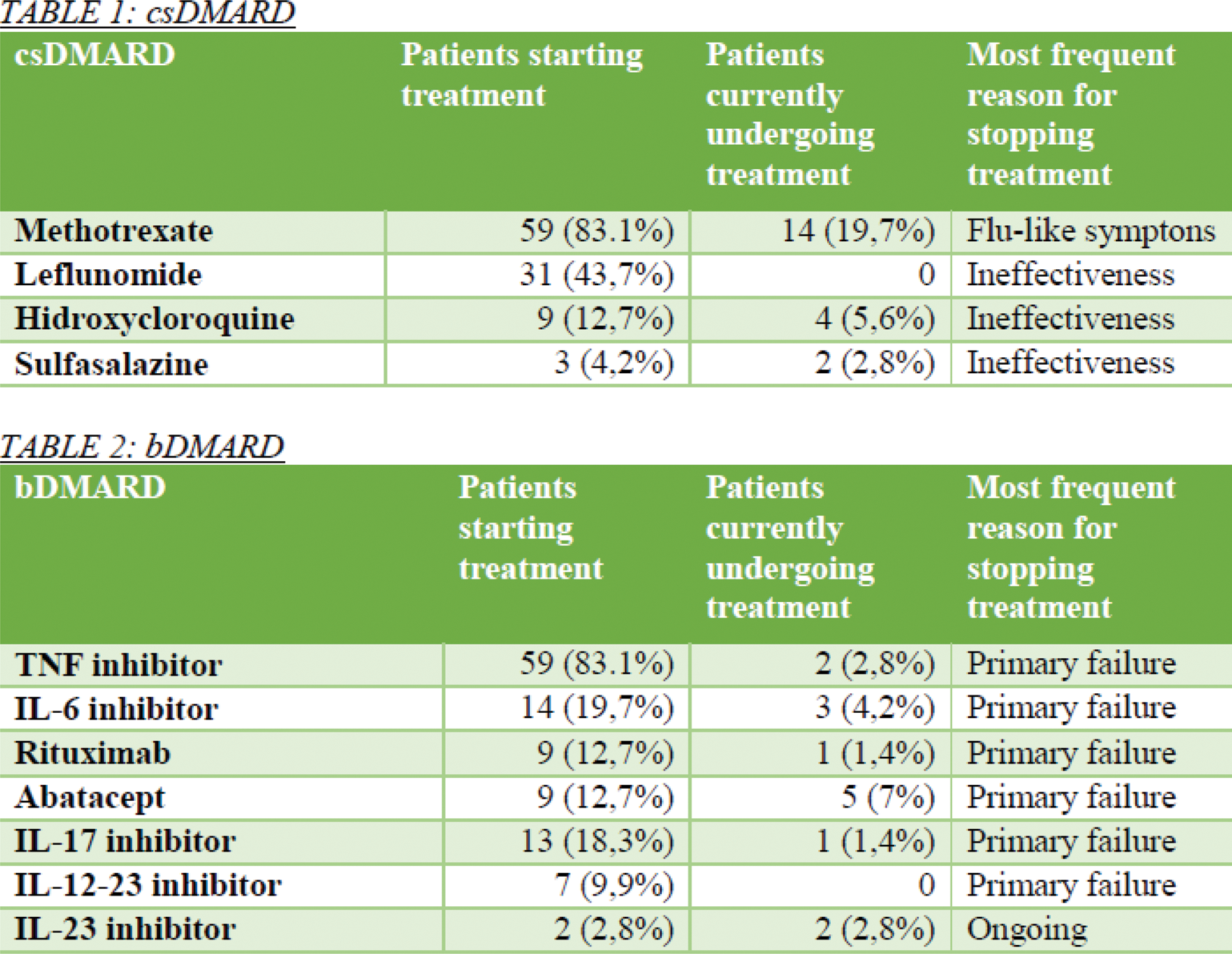
The following AE were noticed: infections , only 1 case of HZ and 2 serious infections that required hospitalization were documented (specifically due to Sars-Cov2), 3 cases of neoplasms (2 basal cell carcinomas and 1 dermatofibroma) and only 1 thrombotic episode (pulmonary thromboembolism). These events appeared in patients treated with tofacitinib (10 mg/day), except for basal cell carcinomas that occurred with Baricitinib (4 mg/day). No adverse events were documented for Upadacitinib. No statistically significant differences were observed by medication as the subgroups had a low sample size. At the last consultation, clinical remission was achieved in 74.6% of the patients. Of the 71 patients, 18 (25.4%) had to discontinue JAKi: The most frequent causes were primary and secondary failure (55%-10% respectively), only 1 due to thrombotic event and none due to infectious or oncological complication.
Conclusion: In our experience, JAKi prove to be a useful therapeutic option in the management of different inflammatory pathologies and, furthermore, with an adequate safety profile with low risk of infection, oncological and/or thrombotic.
REFERENCES: [1] Benucci M, Bernardini P, Coccia C, De Luca R, Levani J, Economou A, Damiani A, Russo E, Amedei A, Guiducci S, Bartoloni E, Manfredi M, Grossi V, Infantino M, Perricone C. JAK inhibitors and autoimmune rheumatic diseases. Autoimmun Rev. 2023 Apr;22(4):103276. doi: 10.1016/j.autrev.2023.103276. Epub 2023 Jan 14. PMID: 36649877.
Acknowledgements: NIL.
Disclosure of Interests: None declared.