

Background: Treat-to-target strategies for rheumatoid arthritis (RA) aim for remission or low disease activity (LDA) and improve long-term outcomes[1].
Objectives: To report early achievement of remission or LDA (based on CDAI) at 3 months (M) in a real-world population and explore the long-term (2 years) consequences of achieving early remission/LDA.
Methods: In the RA-BE-REAL patients with RA initiated baricitinib (Cohort A) or any biologic (b) or any other targeted synthetic (ts) (Cohort B) disease-modifying antirheumatic drug (DMARD) for the 1st time. [2]. This analysis covers European data. Baseline and effectiveness data at 3M (early remission/LDA) and 24M, are presented descriptively.
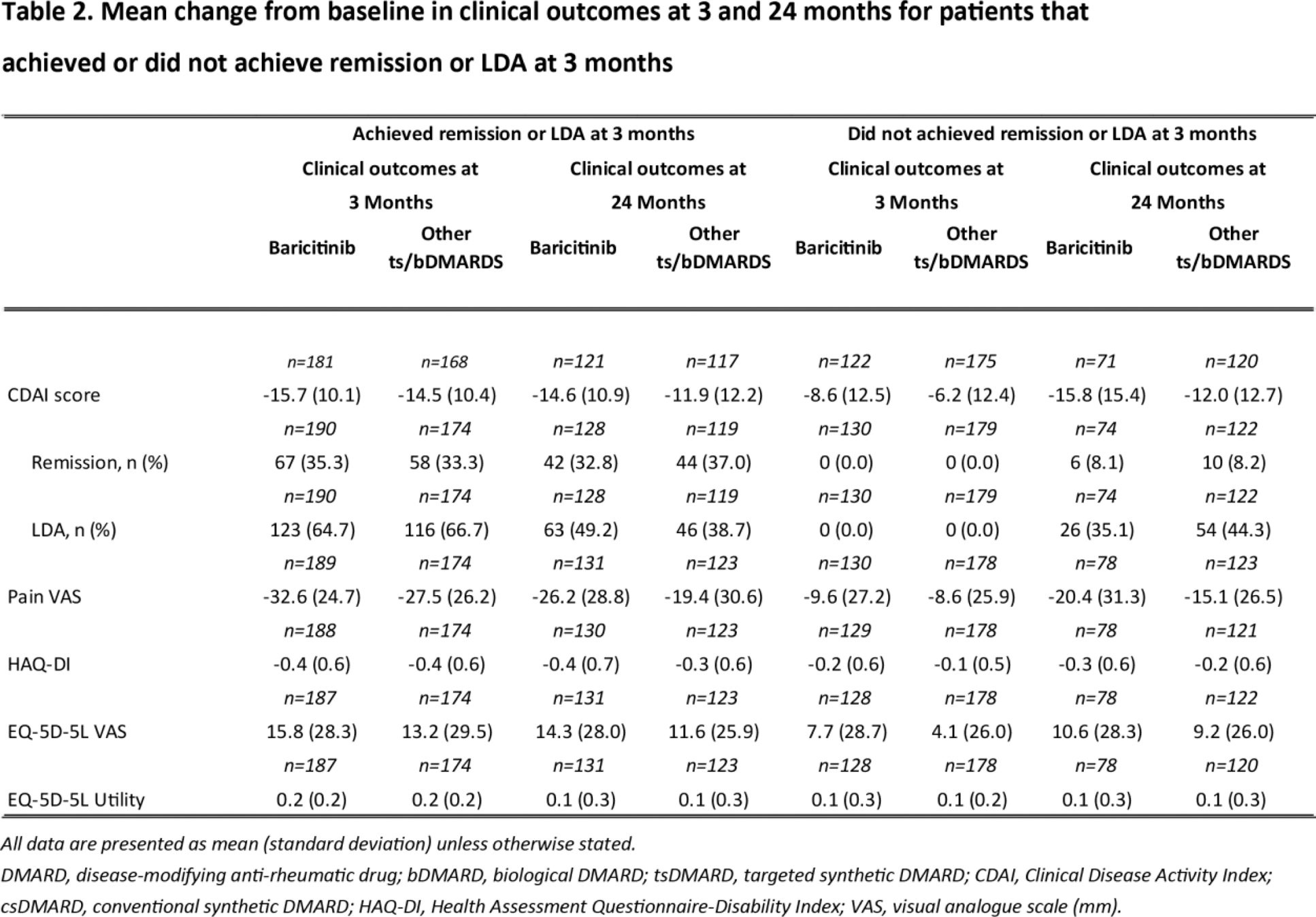
Results: At 3M, high proportions of patients in both cohorts (Cohort A: n= 190, 59.4%; Cohort B: n=174, 49.3%) reached remission or LDA (Table 1). Patients reaching early remission/LDA had greater improvements in short and long-term pain, functioning (HAQ-DI), and quality of life (EQ-5D-5L) than those who didn’t (Table 2). Greater numerical improvements were seen in mean change from baseline on CDAI, pain, HAQ-DI and E5-QD up to 2 years in patients treated with baricitinib compared to those treated with other molecules. Treatment discontinuation between 3-24M in those who did not achieve remission/LDA at 3M (Cohort A: 53.1%, Cohort B: 58.7%) was numerically higher than those who did (Cohort A: 30.0%, Cohort B 41.4%).
Conclusion: In a real-life setting, 59.4% of patients treated with baricitinib and 49.3% of patients on other b/tsDMARDs achieve LDA or remission at 3 months. Achieving LDA or remission was associated with long-term outcomes up to 2 years. Improvements on major outcomes was greater up to 2 years in Baricitinib treated patients than for those receiving other ts/bDMARDs.
REFERENCES: [1] Smolen JS. Treat-to-target as an approach in inflammatory arthritis. Curr Opin Rheumatol. 2016 May;28(3):297-302.
[2] Alten R, Burmester GR, Matucci-Cerinic M, et al. Comparative Effectiveness, Time to Discontinuation, and Patient-Reported Outcomes with Baricitinib in Rheumatoid Arthritis: 2-Year Data from the Multinational, Prospective Observational RA-BE-REAL Study in European Patients. Rheumatol Ther. 2023 Dec;10(6):1575-1595.
Baseline demographics, treatment history, and clinical characteristics for patients who achieved or did not achieve CDAI Remission or LDA at 3 months.
| Achieved Remission or LDA | Did not achieve Remission or LDA | |||
|---|---|---|---|---|
|
Baricitinib
|
Other ts/bDMARDs
|
Baricitinib
|
Other ts/bDMARDs
|
|
| Age in years, mean (SD) | 58.8 (13.4) | 56.1 (14.4) | 59.6 (13.0) | 57.4 (13.0) |
| Female (Yes) | 143 (75.3) | 124 (71.3) | 105 (80.8) | 146 (81.6) |
| BMI (kg/m2), mean (SD) | 26.1 (5.1) | 26.2 (5.6) | 27.1 (5.3) | 27.6 (6.4) |
| Current/former smoker | 75 (39.5%) | 75 (43.1%) | 58 (44.6%) | 87 (48.6%) |
| Family history of RA | 30 (15.8) | 23 (13.2) | 20 (15.4) | 40 (22.3) |
| Duration of RA from diagnosis to enrolment, years, mean (SD) | 10.0 (9.1) | 7.4 (8.3) | 10.5 (9.5) | 9.8 (10.4) |
| Naïve to b/tsDMARD | 109 (57.4) | 111 (63.8) | 39 (30.0) | 99 (55.3) |
| 1 b/tsDMARD prior to enrolment | 24 (12.6) | 19 (10.9) | 18 (13.8) | 15 (8.4) |
| 2 b/tsDMARDs prior to enrolment | 34 (17.9) | 23 (13.2) | 39 (30.0) | 31 (17.3) |
| >2 b/tsDMARDs prior to enrolment | 23 (12.1) | 21 (12.1) | 34 (26.2) | 34 (19.0) |
| Monotherapy at time of enrolment | 85 (44.7) | 48 (27.6) | 63 (48.5) | 66 (36.9) |
| CDAI score, mean (SD) | 20.1 (10.1) | 19.2 (10.3) | 28.5 (12.1) | 27.2 (12.2) |
| Pain VAS, mean (SD) | 52.5 (23.1) | 49.0 (23.1) | 65.4 (21.4) | 61.9 (22.7) |
| HAQ-DI, mean (SD) | 1.1 (0.7) | 1.0 (0.7) | 1.7 (0.6) | 1.6 (0.6) |
| EQ-5D-5L VAS, mean (SD) | 56.1 (21.3) | 56.6 (22.0) | 43.2 (23.1) | 48.6 (22.4) |
| EQ-5D-5L Utility, mean (SD) | 0.6 (0.2) | 0.6 (0.2) | 0.4 (0.2) | 0.5 (0.3) |
All data are presented as n (%) unless otherwise stated.
DMARD, disease-modifying anti-rheumatic drug; bDMARD, biological DMARD; BMI, body mass index; CDAI, Clinical Disease Activity Index; csDMARD, conventional synthetic DMARD; HAQ-DI, Health Assessment Questionnaire-Disability Index; RA, rheumatoid arthritis; TNFi, tumour necrosis factor inhibitor; tsDMARD, targeted synthetic DMARD; VAS, visual analogue scale (mm).
Acknowledgements: Medical writing support for this disclosure was provided by Mr. Alan Ó Céilleachair, an employee of Eli Lilly and Company Limited.
Disclosure of Interests: Rieke Alten Rieke Alten has been paid as a speaker by Abbvie, Amgen, BMS, Celltrion, Chugai, Eli Lilly and Company Ltd., Galapagos, Gilead, Janssen, Mylan/Viatris, Novartis, Pfizer, Roche, and UCB., Rieke Alten has received consulting fees from Abbvie, Amgen, BMS, Celltrion, Chugai, Eli Lilly and Company Ltd., Galapagos, Gilead, Janssen, Mylan/Viatris, Novartis, Pfizer, Roche, and UCB., Rieke Alten has received support for travel from Abbvie, Amgen, BMS, Celltrion, Chugai, Eli Lilly and Company Ltd., Galapagos, Gilead, Janssen, Mylan/Viatris, Novartis, Pfizer, Roche, and UCB., Andrew Östör Andrew Östör has provided consulting support to AbbVie, BMS, Roche, Janssen, Lilly, Novartis, Pfizer, UCB, Gilead, and Paradigm, Christopher John Edwards Christopher John Edwards has been paid as a speaker by AbbVie, BMS, Galapagos, Gilead, Eli Lilly and Company Ltd., Fresenius, Pfizer, and Roche., Christopher John Edwards has received consulting fees from AbbVie, Eli Lilly and Company Ltd., Galapagos, and Gilead, Christopher John Edwards has received support for travel from AbbVie and Eli Lilly and Company Ltd., Josef S. Smolen Joseph S. Smolen has received consultancy fees from: AbbVie, Amgen, AstraZeneca, Astro, BMS, Celgene, Celltrion, Chugai, Eli Lilly and Company, Gilead, ILTOO, Janssen, Medimmune, MSD, Novartis, Pfizer, Roche, Samsung, Sanofi, and UCB., Joseph S. Smolen has received grant/research support from: AbbVie, Eli Lilly and Company, Janssen, MSD, Novartis, Pfizer, and Roche., Ewa Haladyj Ewa Haladyj is a minor shareholder in Eli Lilly and Company Ltd., Ewa Haladyj is an employee of Eli Lilly and Company Ltd., Khai Jing Ng Khai Jing Ng is a minor shareholder in Eli Lilly and Company Ltd., Khai Jing Ng is an employee of Eli Lilly and Company Ltd., Samuel Ogwu Samuel Ogwu is a minor shareholder in Eli Lilly and Company Ltd., Samuel Ogwu is an employee of Eli Lilly and Company Ltd., Yuko Kaneko Yuko Kaneko has been paid as a speaker by Pfizer, Gilead, Eli Lilly Japan K.K. Eisai, Chugai, Tanabe-Mitsubishi, Asahi Kasei Pharma, Bristol Myres Squib, Astellas, UCB, AbbVie, and Taisho, Yuko Kaneko has received grants from Pfizer, Gilead, Eli Lilly Japan K.K., Eisai, Chugai, Tanabe-Mitsubishi and Asahi kasei Pharma, Lars Erik Kristensen Lars-Erik Kristensen has been paid as a speaker by Pfizer, AbbVie, Amgen, UCB, Celgene, BMS, Sanofi, Biogen, Forward Pharma, MSD, Novartis, Eli Lilly and Company Ltd., and Janssen., Lars-Erik Kristensen is a shareholder in Novo Nordisk, Eli Lilly, Merck, Novartis, and UCB, Lars-Erik Kristensen has received research grants from Novartis, Janssen; UCB, and Pfizer.