

Background: Since 2022 clinicians have been consistently aware by the European Medical Agency (EMA) of prescribing Jak-inhibitors (JAKi) after assessing a risk/benefit ratio, especially in patients with rheumatoid arthritis (RA) 1 . Nevertheless, during the last years, uptake of selective JAKis has consistently increased, although limited data regarding differences between selective and unselective JAKis are available to date.
Objectives: To assess differences between selective and unselective JAKIs in terms of discontinuation’s rate.
Methods: In this retrospective study, all patients with RA treated with JAKis were prospectively enrolled from 22 Italian centres since 2017. Selective JAKi included filgotinib and upadactinib, whereas unselective JAKIs included tofacitinib and baricitinib. Reasons of discontinuation rates were: primary and secondary inefficacy, remission, adverse events [deaths, infections, onset of major cardiovascular events-(MACEs)-, deep vein thrombosis-(DVT)-, pulmonary embolism-PE, cancers].
The following variables were collected at JAKi’s first prescription: sex, age, disease duration (years), smoking, BMI, comorbidities (diabetes, hypertension, dyslipidaemia, cancer, major cardiovascular events-MACEs), positive RF/ACPA, associated cDMARDs, prednisone and prednisone dosage (mg/day), previous use of JAKi, time to discontinuation (days), reasons of discontinuation, discontinuation rates for each JAKi, line of treatment, DAS28-ESR, b/tsDMARDS naïve patients.
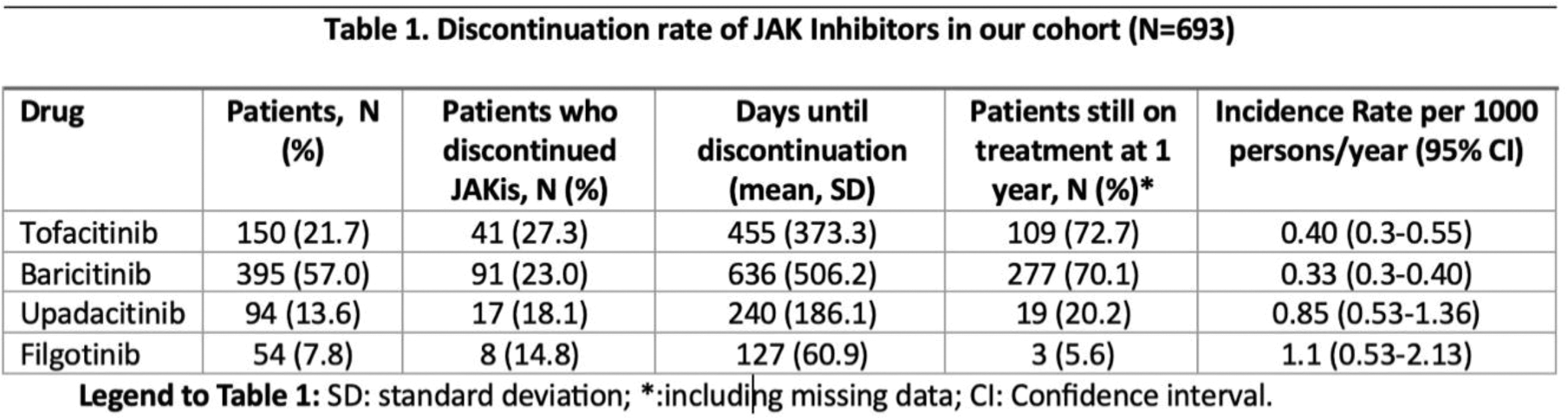
Results: 693 patients were included in study. 395 (57%) patients received baricitinib, 150 (21.7%) tofacitinib, 94 (13.6%) upadacitinib and 54 (7.8%) filgotinib. The majority of patients were treated with unselective JAKi (78.6% vs 21.4% of selective JAKi). 157 (22.7%) patients discontinued JAKI after a median time of 334 days (IQR 152-869). Details of discontinuation rates for each drug are reported in Table 1.
Reasons of discontinuation were primary inefficacy (n=55, 7.9%), secondary inefficacy (n=29, 4.2%), infections (n=9, 1.3%), PE/DVT (n=6, 0.9%), cancer (n=6, 0.9%), deaths (n=2, 0.3%), and other causes (n=7, 3.5%) including remission status in 1 patient. No MACEs were observed in this cohort during the follow up period, although 348 patients (50.2%) had at least one comorbidity at baseline. Notably, both VZV infection (n=3) and DVT/PE (N=6) occurred in patients on treatment with baricitinib.
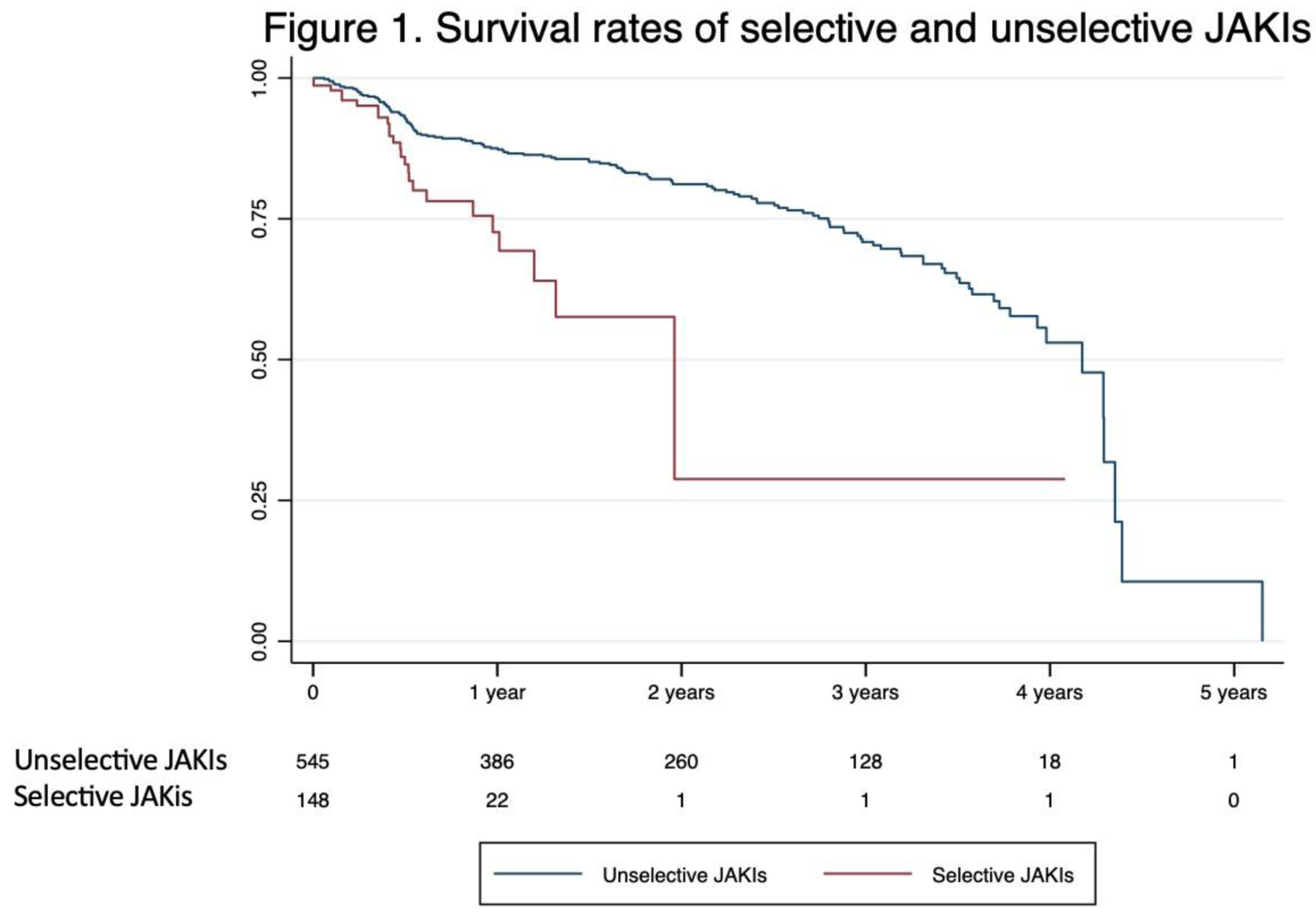
Among 157 patients who discontinued JAKi, a statistical difference between selective and unselective JAKIs was found (15.9% on selective JAKi vs. 84.1% on unselective JAKI, p value <0.001 at log-rank test, Figure 1). Finally selective JAKi were withdrawn before unselective JAKi (median 173 days, IQR 129-22 vs median 401.5 days, IQR 173.5-981).
Conclusion: In this real-world cohort, only a minority of patients discontinued JAKi (22.7%). Primary and secondary inefficacy were the most common reasons of JAKi’s withdrawal. All TVP/PE and VZV infections occurred in the baricitinib group, probably due to different overall time of drug’s exposition. Finally, selective JAKis were discontinued earlier than unselective JAKi.
REFERENCES: [1] Smolen JS, et al. Ann Rheum Dis 2023.
Acknowledgements: NIL.
Disclosure of Interests: Maddalena Larosa UCB, ABBVIE, AMGEN, Dario Camellino AStrazeneca, Boerhinger Ingelheim GSK, and Janssen, Andrea Becciolini: None declared, Eleonora Di Donato: None declared, Giuditta Adorni: None declared, Gianluca Lucchini: None declared, Daniele Santilli: None declared, Eugenio Arrigoni: None declared, Elena Bravi: None declared, Ilaria Platè: None declared, Alessandra Bezzi: None declared, Alessandra Bezzi: None declared, Maria Cristina Focherini: None declared, Fabio Mascella: None declared, Vincenzo Bruzzese: None declared, Palma Scolieri: None declared, Simone Parisi: None declared, Maria Chiara Ditto: None declared, Enrico Fusaro: None declared, Viviana Ravagnani: None declared, Guido Rovera: None declared, Alessia Fiorenza: None declared, Rosetta Vitetta: None declared, Antonio Marchetta: None declared, Alessandro Volpe: None declared, BERND RAFFEINER: None declared, Eleonora Celletti: None declared, Myriam Di Penta: None declared, Emanuela Sabatini: None declared, Francesco Cipollone: None declared, Francesca Ometto: None declared, Cecilia Giampietro: None declared, Valeria Nucera: None declared, Aurora Ianniello: None declared, Francesca Serale: None declared, Marta Priora: None declared, Dilia Giuggioli: None declared, Carlo Salvarani: None declared, Gilda Sandri: None declared, Federica Lumetti: None declared, FRANCESCO MOLICA COLELLA: None declared, Veronica Franchina: None declared, Aldo Molica Colella: None declared, Patrizia Del Medico: None declared, Rosalba Caccavale: None declared, Marino Paroli: None declared, Giulio Ferrero: None declared, Natalia Mansueto: None declared, Romina Andracco: None declared, Matteo Colina: None declared, Simone Bernardi: None declared, Francesco Girelli: None declared, Antonella Farina: None declared, Roberta Foti: None declared, Francesco De Lucia: None declared, Giorgio Amato: None declared, Rosario Foti: None declared, Alberto Lo Gullo: None declared, Mirco Magnani: None declared, Olga Addimanda: None declared, Massimo Reta: None declared, Alarico Ariani AMGEN, JANSSEN.