

Background: Tumor necrosis factor inhibitors (TNFi) are usually used following conventional synthetic disease-modifying antirheumatic drug (csDMARD) failure in Rheumatoid Arthritis (RA), preferably combined with the latter. Patients with inadequate response to first TNFi can cycle to another TNFi or switch to therapies with a different mechanism of action (MOA) such as other biologic disease-modifying anti-rheumatic drugs (nonTNFi bDMARDs: abatacept, rituximab or tocilizumab) or targeted synthetic disease-modifying anti-rheumatic drugs (tsDMARDs: baricitinib, filgotinib, tofacitinib or upadacitinib) 1,2 .
Objectives: This study aimed to identify demographic or clinical differences between patients TNFi cycling or MOA switching due to ineffectiveness of their first TNFi during the first year of therapy.
Methods: This is a retrospective single-center study including adult patients with RA registered in Reuma.pt fulfilling ACR/EULAR 2010 classification criteria who changed to a second targeted therapy after failing a first TNFi during the first year of treatment. Demographic and clinical data were collected. Patients were grouped based on the class of their second targeted therapy either as TNFi cyclers or MOA switchers. Disease parameters were compared at baseline and at the last evaluation before cycling or switching therapy.
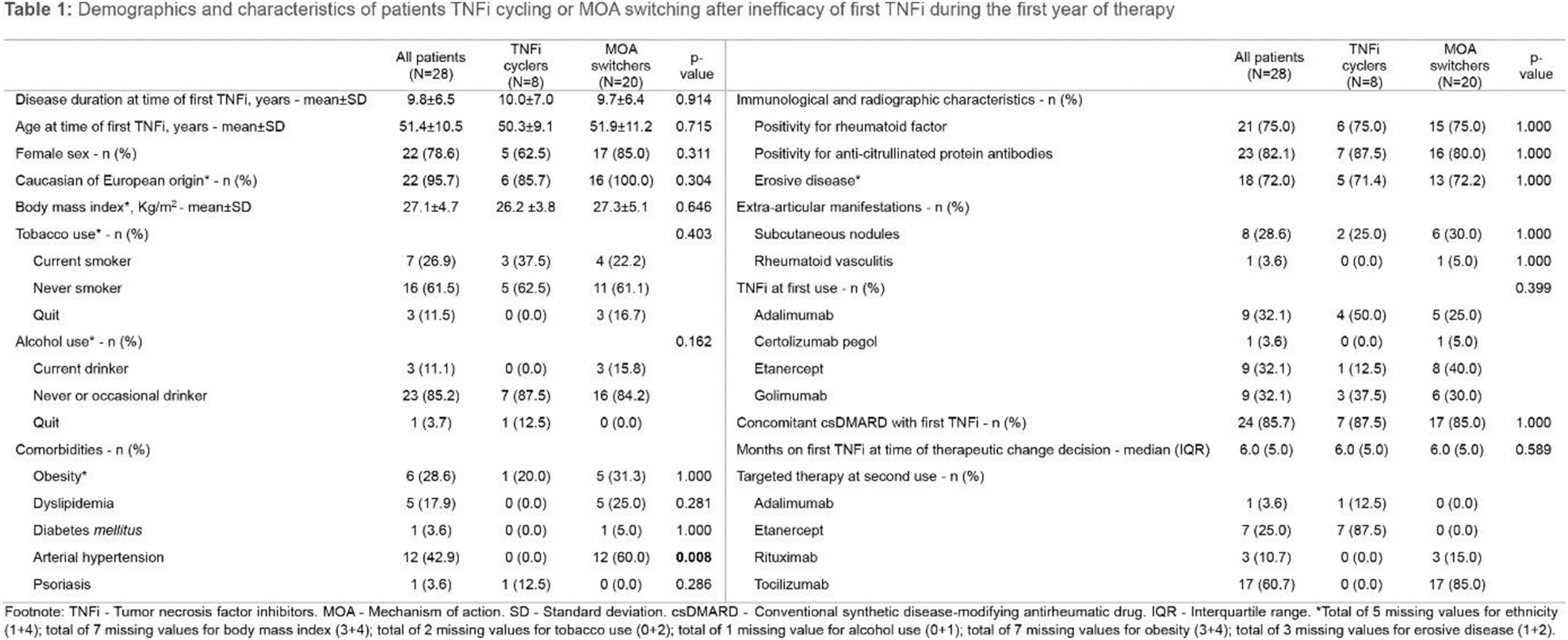
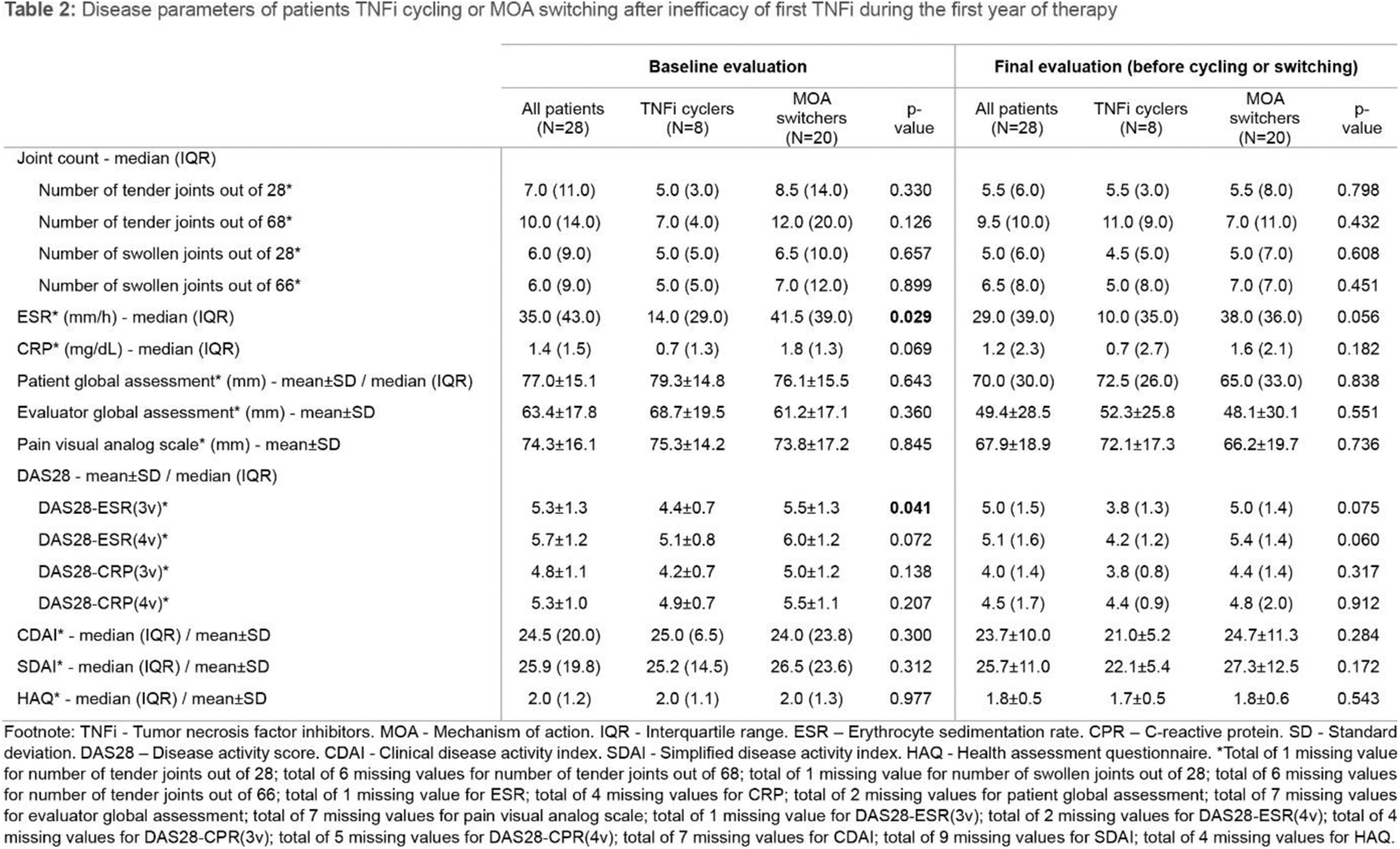
Results: Out of 745 RA patients, 28 met the study inclusion criteria. Eight patients were TNFi cyclers (28.6%) and 20 switched to a second targeted therapy with a different MOA (71.4%). Overall, the mean disease duration was 9.8±6.5 years with mean age of 51.4±10.5 years (Table 1). Most patients were females (78.66%) and Caucasians of European origin (95.7%). Arterial hypertension was significantly more frequent in MOA switchers (p=0.008), but no significant differences were found regarding tobacco use (p=0.403), alcohol use (p=0.162), obesity (p=1.000), dyslipidemia (p=0.281), diabetes mellitus (p= 1.000) or psoriasis (p=0.286). Rates of positivity for rheumatoid factor and for anti-citrullinated protein antibodies and erosive disease were similar for TNFi cyclers and MOA switchers (p=1.000). No extra-articular manifestations prevailed between groups (p=1.000). Half of TNFi cyclers received adalimumab as first TNFi and 40% of MOA switchers started with etanercept (p=0.399). Overall, 85.7% of patients were prescribed a csDMARD concomitantly with their first bDMARD. Median time on first TNFi did not differ between groups (p=0.589). Most patients cycled to etanercept (87.5%) or switched to tocilizumab (85.0%). There were no switchers to tsDMARDs. Joint count was similar both at baseline and at the final evaluation before cycling or switching (Table 2). Erythrocyte sedimentation rate (ESR) was higher in MOA switchers, reaching statistical significance at baseline [median 41.5 (39.0) vs. 14.0 (29.0) mm/h, p=0.029]. Disease activity score (DAS28) tended to be higher in the MOA switching group, showing statistically significant difference at baseline evaluation with DAS28-ESR three variables (mean 5.5±1.3 vs. 4.4±0.7, p=0.041). There were no significant differences regarding C-reactive protein, patient or evaluator global assessments, pain visual analog scale, clinical disease activity index, simplified disease activity index or health assessment questionnaire.
Conclusion: Most patients received a first TNFi combined with a csDMARD, in accordance with current guidelines 1,2 . MOA switchers showed greater prevalence of arterial hypertension as well as higher titles of ESR and higher disease activity at baseline according to DAS28-ESR three variables. These findings may influence therapeutic decisions, but further investigation is needed.
REFERENCES: [1] Smolen JS, Landewé RBM, Bergstra SA, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2022 update. Ann Rheum Dis. 2023;82(1):3-18.
[2] Fraenkel L, Bathon JM, England BR, et al. 2021 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis. Arthritis Care Res (Hoboken). 2021;73(7):924-939.
Acknowledgements: NIL.
Disclosure of Interests: None declared.