

Background: Janus kinase inhibitors (JAKi) represent a novel class of oral targeted disease-modifying drugs, heralding a recent revolution in the therapeutic landscape for rheumatoid arthritis (RA) and other immune-mediated conditions. These inhibitors, which can complement or even replace conventional and biological drugs, operate through a distinctive mechanism involving intracellular disruption of the JAK-STAT pathway—a pivotal player in the immune response. The Hill coefficient (H) serves to describe the sigmoidicity of a drug’s concentration-response curve. When H > 2.0, a “time-dependent” effect is observed, where the critical factor is not the peak drug concentration but the crossing of a threshold beneath which the effect becomes negligible. The prolonged serum concentration above this threshold correlates with a more pronounced effect. Conversely, when H < 2.0, a “concentration-dependent” effect emerges, with the peak concentration holding significance [1]. In the report by Chimalakonda A et al [2], the H for the inhibitory effect of JAK1/3 in tofacitinib, baricitinib, and upadacitinib ranged from 1.0-1.3. For JAK1 inhibition in filgotinib (FIL), the H was nearly 2 [3]. Intriguingly, in a phase II study, FIL at 100 mg BID demonstrated a tendency to be more effective than FIL at 200 mg QD after 12 weeks of treatment [4]. However, the time-dependency of FIL’s effect on rheumatoid arthritis remains unclear.
Objectives: The objective of this investigation is to assess whether the impact of FIL on rheumatoid arthritis exhibits time-dependent characteristics.
Methods: This constituted a retrospective, observational study involving 25 patients diagnosed with rheumatoid arthritis (RA) who met the 2010 American College of Rheumatology (ACR)/EULAR classification criteria. These patients were prescribed FIL and underwent a minimum 12-week follow-up at our hospital from November 2020 to October 2023. The efficacy of FIL was assessed based on the EULAR response at 12-week treatment, with concurrent measurement of the change in DAS28-CRP. Patients were categorized into three groups based on renal function and FIL dosage: FIL 200 mg group [FIL200], FIL 100 mg with chronic kidney disease (CKD) (eGFR < 60 ml/min/1.73m 2 ) [FIL100CKD], and FIL 100 mg without CKD [FIL100].
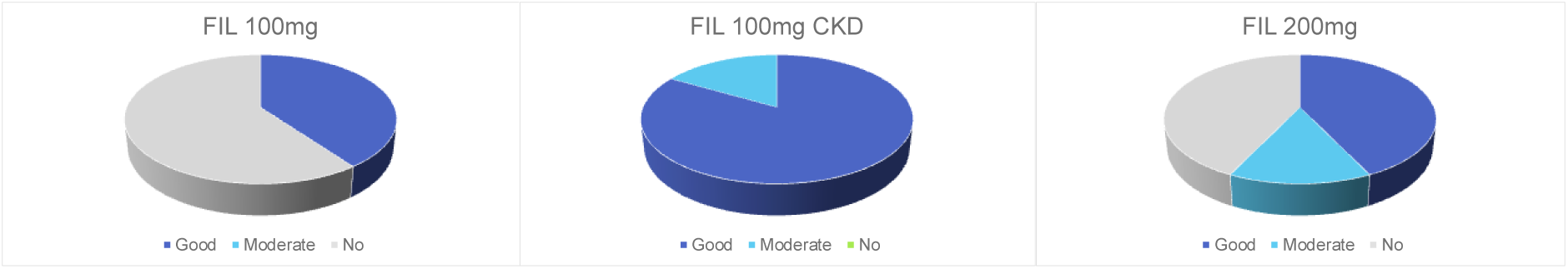
Results: The median age of the patients was 69.0 years (IQR: 61.0-76.0), and the median disease duration was 156 months (102-180). The median DAS28-CRP was 3.60 (3.16-4.49). Comparing the FIL100CKD group to the FIL200 group, the former exhibited a higher median age (80.0 vs. 73.0 year, P<0.05). However, other patient backgrounds showed no statistically significant differences. No significant variations were observed in the number of concomitant MTX, other csDMARDs, or prednisolone among the three groups. But, there was a tendency towards fewer patients being biologics- and JAKi-naive in the FIL200 group (FIL100 group; 60.0%, FIL100CKD group; 66.7% and FIL200 group; 14.3%, respectively). In terms of treatment outcomes, the EULAR response and ΔDAS28-CRP at 12 weeks tended to be higher in the FIL100, FIL100CKD, and FIL200 groups, in that sequential order (Figure 1).
Conclusion: FIL exhibits a Hill coefficient close to 2, implying a time-dependent pharmacodynamic effect rather than concentration dependence. In cases with eGFR <60, the time above IC5 for JAK1 is extended (FIL100; approximately 6 hours, FIL100CKD; approximately 8 hours, FIL200; approximately 9.5 hours) [5,6]. Clinical data derived from the current study imply that the efficacy of FIL is time-dependent. Further studies are needed to confirm this.
REFERENCES: [1] Czock D, Keller F. J Pharmacokinet Phar. 2007;34(6):727–51.
[2] Chimalakonda A, et al. Dermatology Ther. 2021;11(5):1763–76.
[3] Namour F, et al. Clin Pharmacokinet. 2015;54(8):859–74.
[4] Westhovens R, et al. Ann Rheum Dis. 2017;76(6):998.
[5] Namour F, et al. Brit J Clin Ph REFERENCES armaco. 2018;84(12):2779–89.
[6] Veeravalli V, et al. Drug Safety. 2020;1–15.
12W EULAR response
Acknowledgements: NIL.
Disclosure of Interests: None declared.