

Background: Rheumatoid arthritis (RA) is the most common chronic inflammatory joint disease, leading to structural damage and loss of quality of life if left untreated. Many effective disease modifying anti-rheumatic drugs (DMARDs) can limit disease progression, however therapies that prevent the onset of RA are currently not available. As it is nowadays possible to identify individuals at risk of developing RA (RA-risk) [1], several clinical trials have been performed to prevent disease development, but so far the selected drugs could not prevent onset of disease [2]. Synovial gene signatures associated with the development of disease in RA-risk individuals have been identified [3]. We postulate that therapeutic compounds that could reverse these signatures, could be effective in preventing the disease development in RA-risk individuals.
Objectives: To identify druggable compounds that could reverse the synovial gene signature related with high risk to develop RA.
Methods: The pre-RA gene signature was defined by genes, differentially expressed in synovial tissue of RA-risk individuals who later developed RA, compared to RA-risk individuals who did not develop the disease. We used the iLINCS, CLUE and L1000CDS 2 platforms [4] to identify existing drugs that could potentially reverse this pre-clinical RA specific profile. Next we applied the CoDReS software, which allowed us to further rank these compounds based on their druggability potential. As a second approach, we identified transcription factors (TF) that could target the pre-RA specific signature using the TRRUST database. Subsequently, a gene regulatory network, integrating the identified TF and genes from the initial pre-RA signature (protein coding genes) was constructed, and potential drugs targeting central components of this network were retrieved via the DGIDb database, in order to build a final drug-gene interaction network.
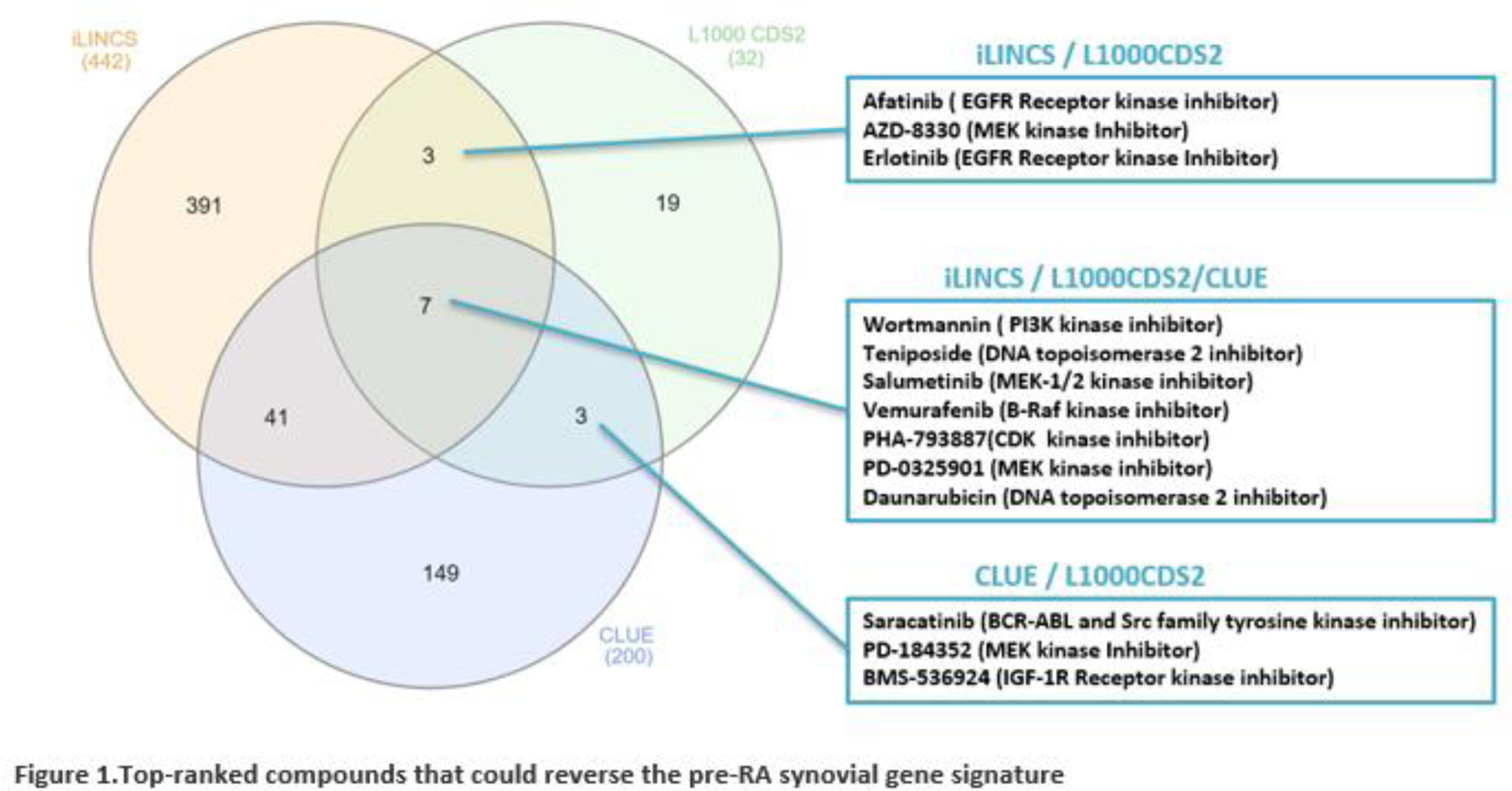
Results: Each platform (iLINCS, CLUE and L1000CDS 2 ), separately identified drugs that could reverse the pre-RA gene signature. When focusing on common drugs identified by at least two of the platforms, inhibitors of the PI3K/Akt/mTOR pathway, compounds targeting the Src kinase family, as well as MEK kinase inhibitors, were predicted to reverse the synovial pre-RA gene signature and were included amongst the top ranked compounds based on the CoDReS platform output (Figure 1).
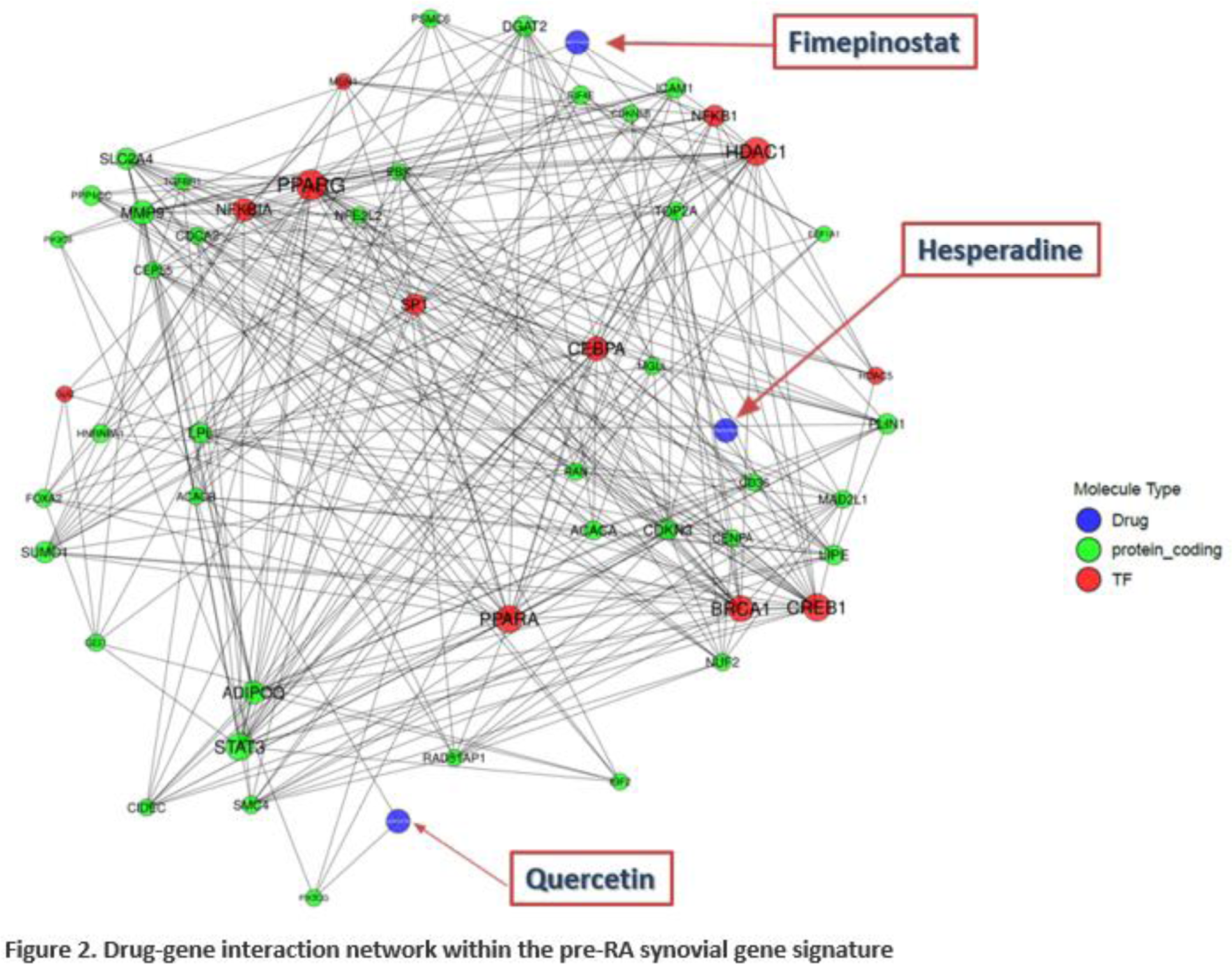
According to our network-based drug repurposing approach a drug-gene interaction network was built (Figure 2). In this network we identified a dual HDAC/PI3K kinase inhibitor (fimepinostat), an aurora kinase inhibitor (Hesperadine) and the flavonoid natural product quercetin, to represent potential drug candidates.
Conclusion: Application of two independent in silico drug repurposing strategies, uncovered novel compounds that have the potential to counteract the synovial gene signature associated with RA development in RA-risk individuals. These findings provide the support for further in vitro testing of these compounds and may guide future clinical trials aimed at halting disease development in RA-risk individuals.
REFERENCES: [1] Gerlag, Danielle M et al. “EULAR recommendations for terminology and research in individuals at risk of rheumatoid arthritis: report from the Study Group for Risk Factors for Rheumatoid Arthritis.” Annals of the rheumatic diseases vol. 71,5 (2012): 638-41.
[2] Frazzei, Giulia et al. “Prevention of rheumatoid arthritis: A systematic literature review of preventive strategies in at-risk individuals.” Autoimmunity reviews vol. 22,1 (2023): 103217.
[3] de Jong, Tineke A et al. “Synovial gene signatures associated with the development of rheumatoid arthritis in at risk individuals: A prospective study.” Journal of autoimmunity vol. 133 (2022): 102923.
[4] Keenan, Alexandra B et al. “The Library of Integrated Network-Based Cellular Signatures NIH Program: System-Level Cataloging of Human Cells Response to Perturbations.” Cell systems vol. 6,1 (2018): 13-24.
Acknowledgements: NIL.This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 847551 (ARCAID). The authors Dimitrios Nikolakis and Panagiotis Garantziotis contributed equally in the data analysis part. The authors Marleen van de Sande and Lisa van Baarsen, contributed equally in this project and share last authorship.
Disclosure of Interests: Dimitrios Nikolakis: None declared, Panagiotis Garantziotis: None declared, Giulia Frazzei: None declared, George K. Bertsias: None declared, Dimitrios Boumpas: None declared, Ronald F. van Vollenhoven Consultancy and/or speaker: AbbVie, AstraZeneca, Biogen, BMS, Galapagos, GSK, Janssen, Pfizer, RemeGen, UCB, Support for Research or Educational programs (institutional grants): AstraZeneca, BMS, Galapagos, MSD, Novartis, Pfizer, Roche, Sanofi, UCB, Lisa G.M. van Baarsen: None declared, Marleen G.H. van de Sande: None declared.