

Background: Many individuals with neurodegenerative and immune-mediated inflammatory disorders, such as primary Sjogren’s syndrome (PSS), experience debilitating fatigue that substantially impacts their quality of life [1]. Currently, assessments of fatigue rely on patient reported outcomes (PROs), which are subjective and prone to recall bias. Wearable devices, however, can provide objective, reliable, and continuous estimates of human activity and physiology, which are essential components of health, and may provide objective evidence of fatigue [2]. However, current literature is very limited.
Objectives: This study aims to discriminate PSS patients with different fatigue levels using real-world measures of activity type and intensity.
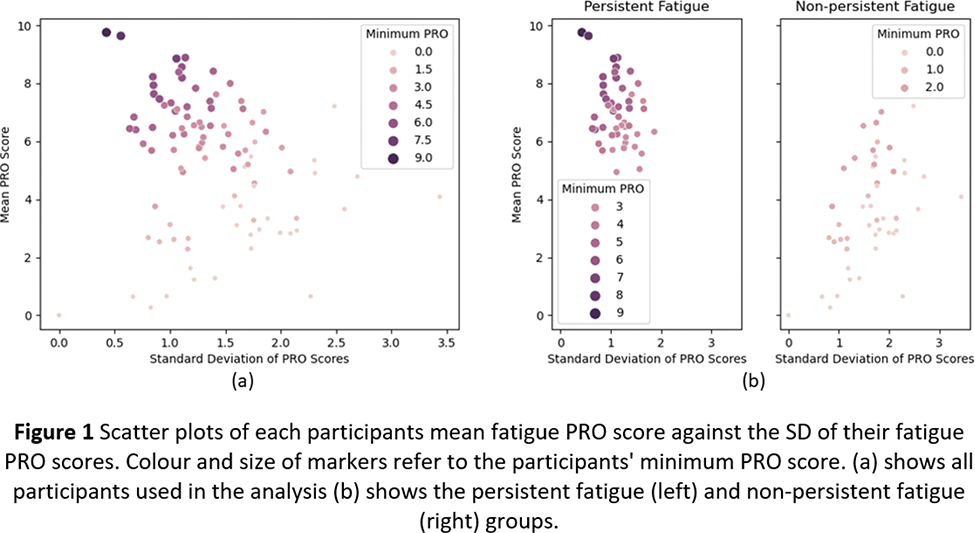
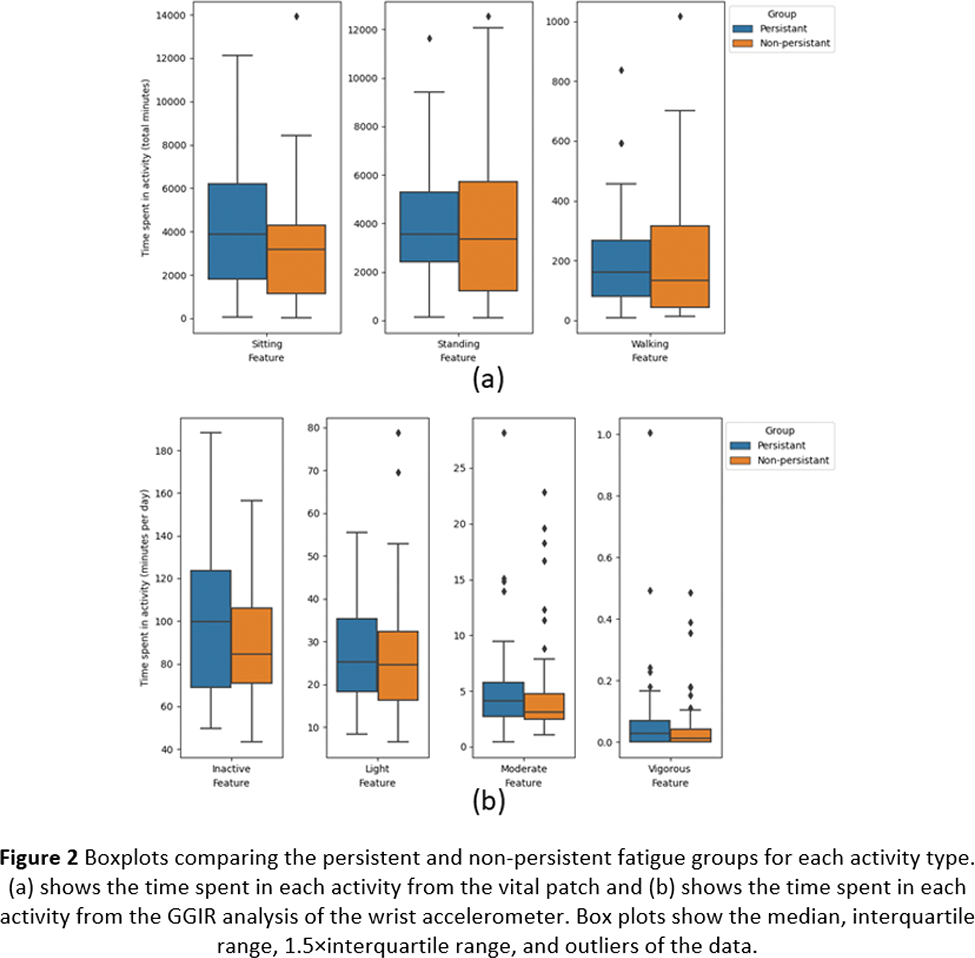
Methods: 105 participants with PSS recruited from an observational study wore an ECG-based VitalPatch sensor on the chest and a triaxial accelerometer (Actigraph GT9X Link) on the non-dominant wrist at home for two 7-day continuous periods. Concurrently, the participants completed paper-based PROs relating to fatigue up to 4 times a day: morning, afternoon, evening, and night (however night was not included in this analysis) with 0 meaning no fatigue and 10 meaning maximum fatigue. The participants were labelled as presenting with “persistent fatigue” if their minimum fatigue rating across the study was 3 out of 10 or higher and as “non-persistent fatigue” if it was 2 or lower. This separated the participants into those with consistently high ratings of fatigue and those with more variable ratings of fatigue, as seen in Figure 1. The time each participant spent in activity types (standing, walking, or sitting behaviours) were estimated by the VitalPatch sensor’s built-in software [3,4] and the time spent in activity intensity levels (inactive, light, moderate, and vigorous) were estimated from the wrist-worn accelerometer using GGIR [5,6,7]. The boxplots of the digital measures extracted from each group are shown in Figure 2, and the Mann-Whitney U test was used to assess if the distributions of the two groups differed significantly.
Results: 97 subjects had useable data for analysis (84 female, 12 male, 1 undefined and mean±SD age of 59.7±13 years), 49 of which were classified as persistently fatigued. Figure 2 shows the time spent in different activity levels as estimated by the vital patch (part a) and the wrist-worn accelerometer (part b). From the vital patch, the persistent fatigue group spent more time sitting, but the times spent standing and walking were indistinguishable between the groups. Similarly, the actigraphy sensor found that the persistently fatigued group spent more time inactive, and slightly more time in moderate activity, but the light and vigorous activities were indistinct. However, the p-values from the Mann-Whitney U tests were >0.05 for all activity levels for both sensors. Therefore, persistent fatigue did not significantly impact the activity levels of these patients. While this is counter-intuitive, this is consistent both with patient experience and previous studies [8-11], where patients who are fatigued do not do significantly less than those who are not but such activities might require more “efforts”, and they may be slightly more inclined be sedentary when possible.
Conclusion: This analysis has shown that the persistently fatigued participants tended to spend more time sedentary compared to the non-persistent group, but this separation was not statistically significant. Therefore, future research should explore more nuanced measures to investigate the relationships between habitual movement, other physiological signals, and fatigue in PSS.
REFERENCES: [1] Segal et al. A&R 2008.
[2] Viceconti et al. Front. Med. 2022.
[3] Haveman et al. JFR. 2022.
[4] Chan et al. IEEE EMBC 2013.
[5] Migueles et al. J Meas Phys Behav. 2019.
[6] van Hees et al. PLoS ONE. 2013.
[7] Sabia et al. Am J Epidemiol. 2014.
[8] Micheal et al. Rehabilitation Nurs. 2006.
[9] Nagaki et al. Healthcare 2022.
[10] Liguori et al. Neurorehabilitation 2021.
[11] Vogelaar et al. Scand. J. Gastroenterol. 2015.
Acknowledgements: NIL.
Disclosure of Interests: Chloe Hinchliffe: None declared, Bing Zhai: None declared, Victoria Macrae: None declared, Jade Walton: None declared, Wan-Fai Ng: None declared, Silvia Del Din S. Del Din reports consultancy activity with Hoffmann-La Roche Ltd. outside of this study.