

Background: Sjögren’s syndrome, a chronic autoimmune disease characterized by inflammation of exocrine glands, may be difficult to diagnose due to frequently non-specific symptoms. Therefore, there is a considerable diagnostic delay.
Objectives: This study explores the journey of patients with primary Sjögren’s syndrome from symptom onset to diagnosis and assesses its impact on patient-reported outcomes.
Methods: Participants included Hannover Medical School outpatient clinic patients aged ≥18, diagnosed with primary Sjögren’s syndrome (ICD-10 M35.0) at least 12 months ago. Using a standardized questionnaire demographics, details about symptoms and therapy, pre-diagnosis visits and interactions with physicians were recorded. The patient-reported outcomes assessed emotional burden, impact on daily life and general health status reported on a scale from 0-100 (with 100 being very good health). General performance was measured based on a four-point Likert-scale ranked from no negative impact to very large negative impact. Descriptive analyses and statistical tests were conducted using IBM SPSS Statistics Version 28.0.1.0. A Chi 2 test was used to determine the effect on the patient reported outcomes measured with a Likert-scale. A t-test was used to determine the difference in general health status between the groups of long and short diagnosis delay.
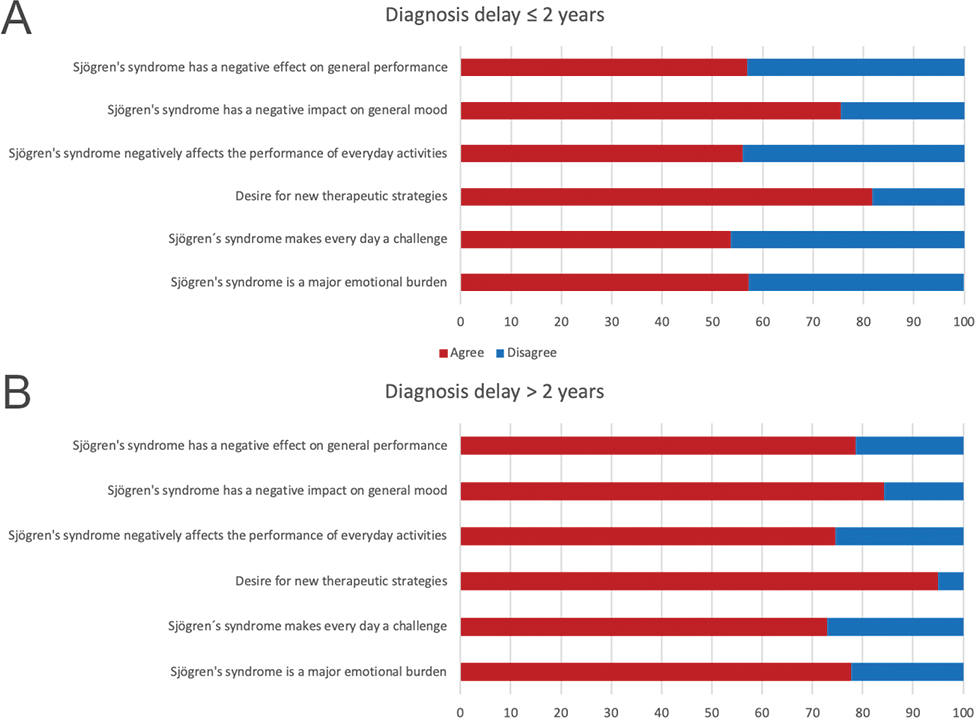
Results: 278 patients with primary Sjögren’s syndrome were recruited, 82.8% were female (Table 1). Initial symptoms included dry eyes (56%), dry mouth (52.4%), and dry skin (40.6%). Joint and muscle pain (59.8%) and fatigue (53.9%) were prevalent. Before the diagnosis was made, general practitioners (80.2%) were commonly consulted, along with ophthalmologists (39.2%), ENT physicians (37.3%), neurologists (36.1%), and orthopedists (35.0%). Rheumatologists diagnosed the disease in 66.0% of cases. The top complaints limiting everyday life at the time of the questionnaire was completed were fatigue (59.5%), joint and muscle pain (56.4%), dry eyes (53.7%), dry mouth (43.6%), and intestinal complaints (23.2%). The average age at diagnosis was 52.3 years, with a delay between the first symptoms indicating a Sjögren´s syndrome to diagnosis averaging 6.3 years (median of 2 years; Table 1). The participants were divided in two groups based on the median (>/≤ 2 years until final diagnosis). Patients reporting a longer diagnostic delay (more than 2 years) indicated a greater emotional burden (77.7%) compared to those with shorter delays (57.2%, p<0.001; Figure 1). Regarding the impact on general performance, 78.6% of patients with a prolonged diagnostic delay reported a negative effect compared to 56.9% with a shorter delay (p=0.012; Figure 1). This trend is also consistent with the general health status in which the general health status was 9.93 units worse in patients with a prolonged diagnostic delay (95% confidence level [-15.55; -4.31], p<0.001) compared to patients with a shorter delay.
Conclusion: The study highlights a significant 6.3-year delay from symptom onset to Sjögren’s syndrome diagnosis, underscoring adverse effects on reported patient outcomes. Particularly, a prolonged delay >2 years negatively affected the patients reported general health status and the impact of the Sjögrens´s syndrome on the general performance significantly.
Patient characteristics
| Sex | n=279 (%) |
| Woman | 231 (82.8) |
| Man | 48 (17.2) |
| Nationality | n=274 (%) |
| German | 264 (96.4) |
| Other | 10 (3.6) |
| Ethnicity | n=272 (%) |
| Caucasian | 269 (98.9) |
| Other | 3 (1.1) |
| General health status | |
| All patients, mean (n=276) | 53.8 (±23.1) |
| Diagnostic delay ≤ 2years, mean (n=129) | 58.2 (±23.1) |
| Diagnostic delay > 2years, mean (n=122) | 48.2 (±22.1) |
| Age | |
| Actual Age (n=279) | 60.1 (±14.0) |
| Age of symptom begin (n=255) | 45.8 (±15.1) |
| Age of diagnosis (n=276) | 52.3 (±14.0) |
| Latency time | n=254 |
| Mean | 6.3 (±9.0) |
| Median | 2.0 |
Patient reported outcomes in long and short diagnosis delay patients (>/≤ 2 years until final diagnosis)
REFERENCES: NIL.
Acknowledgements: The study was financed by an unrestricted grant from Novartis Pharma GmbH without any involvement in the scientific work.
Disclosure of Interests: Anna Meinecke: None declared, Kristine Kreis: None declared, Paul Olson: None declared, Juliana Rachel Hoeper: None declared, Diana Ernst Abbvie, Amgen, Boehringer Ingelheim, Bristol-Myer Squibb, Celgene, Chugai, Gilead, GlaxoSmithKline, Lilly, Medac, Novartis, Pfizer, Roche, UCB, Abbvie, Amgen, Boehringer Ingelheim, Bristol-Myer Squibb, Celgene, Chugai, Gilead, GlaxoSmithKline, Lilly, Medac, Novartis, Pfizer, Roche, UCB, Abbvie, Novartis, Kirsten Hoeper no COI in the field of Sjögren’s syndrome, no COI in the field of Sjögren’s syndrome, in the field of Sjögren’s syndrome from Novartis, Torsten Witte in the field of Sjögren’s syndrome from Novartis, in the field of Sjögren’s syndrome from Novartis, in the field of Sjögren’s syndrome from Novartis.