

Background: Renin-angiotensin-system inhibitors (RASi) have been recommended for patients with diabetic or nondiabetic nephropathy, but the studies focused on RASi in lupus nephritis are scaring.
Objectives: To assess the effect of RASi for LN in a Chinese real-world cohort.
Methods: The primary outcome was proteinuria partial recovery (PPR) rate at 6 months, other outcomes including the rate of complete remission (CR), total remission (TR), decline and decline rate of proteinuria. LN remission was compared by Chi-square tests. Inverse probability of treatment weighting (IPTW) was adopted to adjust for the baseline characteristics. Cox regression analysis was also performed.
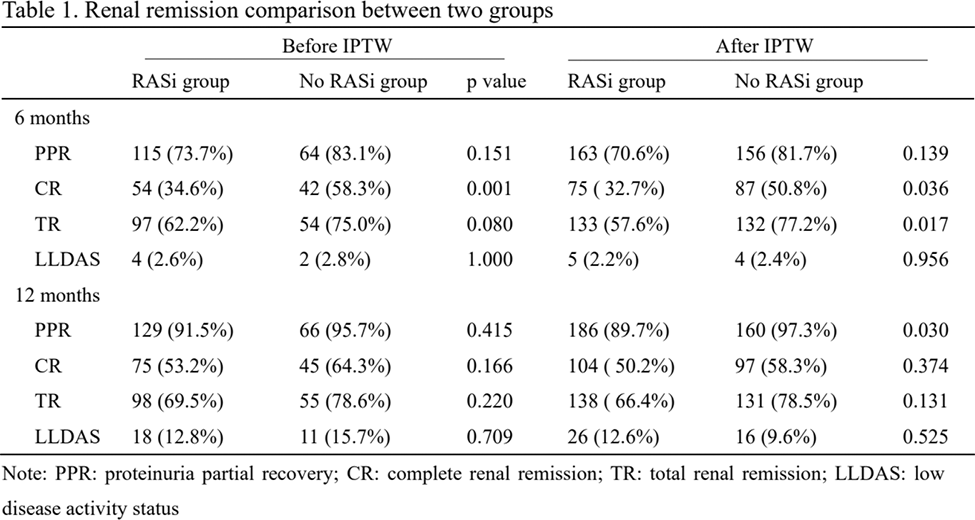
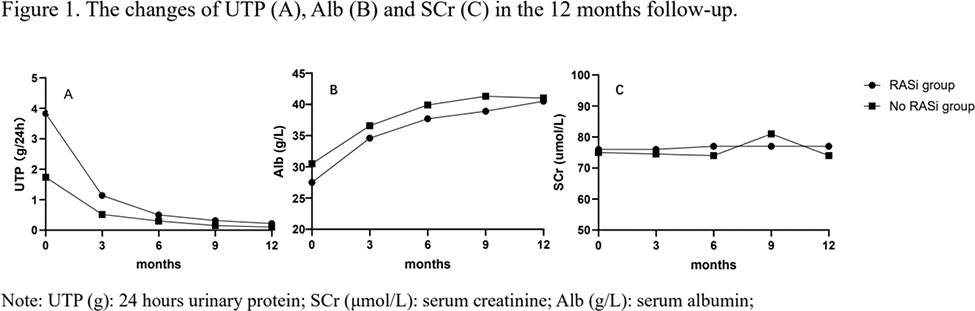
Results: There were 233 LN patients enrolled in the study, with 156 receiving RASi (RASi group) and 77 patients without exposure to RASi (no RASi group). At 6 months, 70.6% patients in RASi group and 81.7% in no RASi group reached PPR (p=0.139). Rates of CR and TR were significantly higher in no RASi group (CR: 32.7% vs. 50.8%, p=0.036; TR: 57.6% vs. 77.2%, p=0.017). At 12 months, the PPR rate was also significantly higher in no RASi group than RASi group (97.3% vs. 89.7%, p=0.030), but the rates of CR and TR were similar (CR: 50.2% vs. 58.3%, p=0.374; TR: 66.4% vs. 78.5%, p=0.131). The reduction of UTP and UTP declining rate were not significantly different between two groups. Meanwhile, the serum SCr was maintained stable and not different between two groups. The SLEDAI-2K and dose of prednisolone were also not statistically different.
Conclusion: Our real-world comparative study suggested that LN patients may not benefit from RASi treatment. and the results need to be interpreted with caution. Further high-quality randomized controlled trials and long-term follow-up are required to evaluate the RASi effects on proteinuria and renal prognosis.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.