

Background: Anifrolumab (ANI) is a fully human monoclonal antibody against the type I interferon receptor that has recently been approved for the treatment of moderate to severe Systemic Lupus Erythematosus (SLE) as an add-on treatment to standard of care. Data from randomized controlled trials have demonstrated its efficacy and safety, but real-world data are still limited, especially regarding the impact of this new drug on patients’ quality of life (QoL).
Objectives: To evaluate the effect of ANI therapy on QoL and disease burden in a multicentric cohort of refractory SLE patients.
Methods: Consecutive adult SLE patients (2019 EULAR/ACR criteria) were prospectively enrolled at ANI prescription. Data on demographic features, medical history, previous therapies and SLICC-DI were collected from clinical charts at enrolment. Patients’ assessments were performed at the first ANI infusion and subsequently after one, three and six months of treatment. At each time-point, the clinical evaluation included SLEDAI-2K, SLE-DAS, Physician Global Assessment, number of tender and swollen joints and cutaneous activity and damage assessed using the Cutaneous LE Disease Area and Severity Index (CLASI-A for activity and -D for damage). Patients’ perspective was assessed by self-administration of validated Patient Reported Outcomes (PROs): Lupus Impact Tracker (LIT) to assess the overall impact of SLE on patients’ QoL, Functional Assessment of Chronic Illness Therapy – Fatigue scale (FACIT-F) to measure self-reported fatigue and its impact on daily activities and, in a subgroup of patients with mucocutaneous involvement, Skindex-16 which is a specific PRO to investigate the impact of the skin disease in symptom, emotional and functioning spheres.
Results: Twenty-five patients (96% female, 92% Caucasian) with a median age of 45 years (IQR 37-58) and a median disease duration of 11 years (IQR 7.5-21.5) were enrolled. In the whole cohort, 24 patients (96%) had a history of articular involvement, 23 (92%) of mucocutaneous involvement, 16 (64%) of haematological involvement, 7 (28%) of lupus nephritis, 5 (20%) of serositis and 4 (16%) of neuropsychiatric involvement. At baseline, 23 patients (92%) were on concomitant steroid therapy (median prednisone daily dose 7.5 mg, IQR 5-10), 19 (76%) on hydroxychloroquine and 23 (92%) on immunosuppressive treatment (10 methotrexate, 9 mycophenolate mofetil, 3 azathioprine, 1 cyclosporine). Active disease manifestations at the time of ANI prescription were in most cases mucocutaneous (18/25, 72%), followed by articular (10/25, 40%) and haematological (5/25, 20%) involvement.
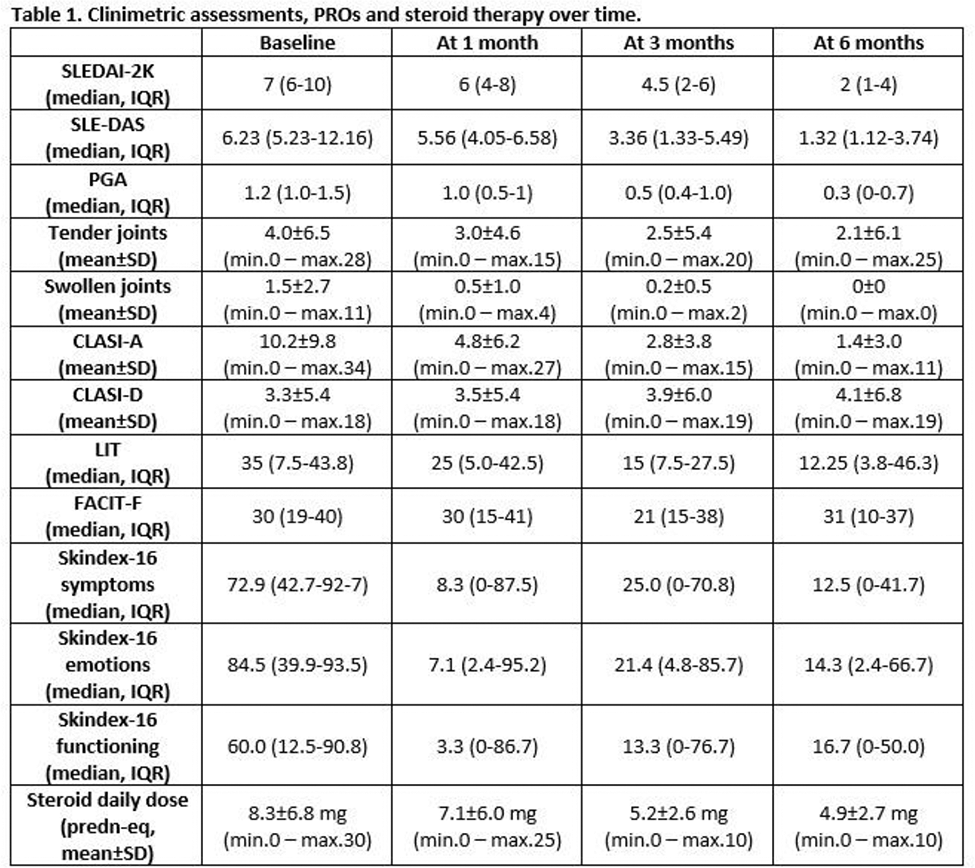
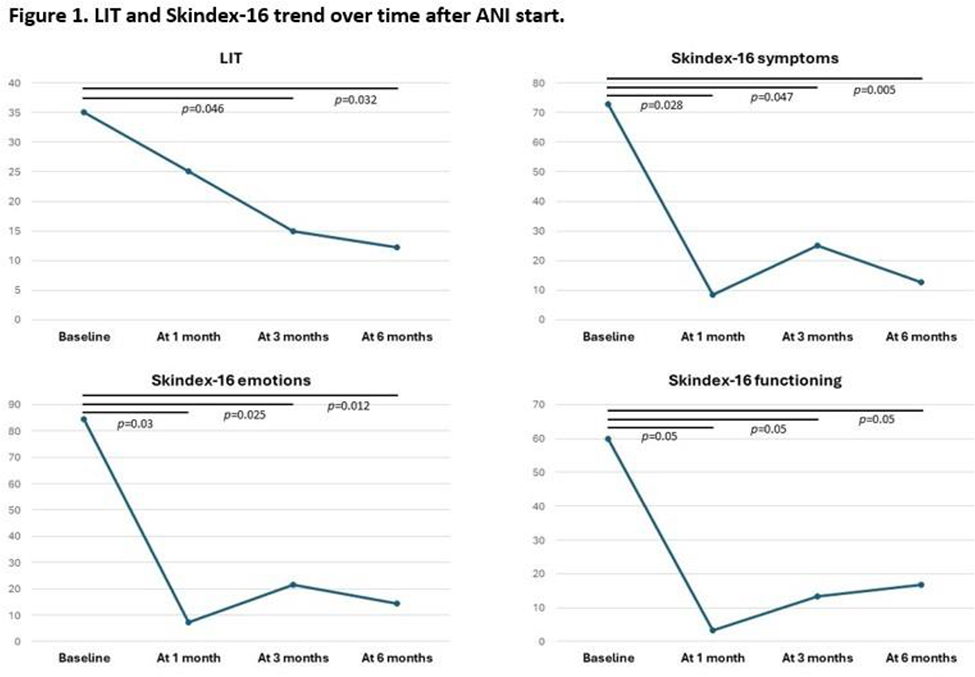
As reported in Table 1, after ANI start, all the disease activity measures showed a progressive improvement over time. A significant correlation was observed between LIT and joint count and Skindex-16 and CLASI-A at baseline (r≥0.464, p≤0.026 for LIT; r≥0.731, p≤0.04 for Skindex-16). During follow-up, LIT and Skindex-16 exhibited progressively and significantly improved scores, while no changes were observed in FACIT-F scores. Notably, Skindex-16 was significantly improved as early as 4 weeks after the first drug infusion (symptoms p=0.028, emotions p=0.03, functioning p=0.05), while LIT reached statistical significance after 12 weeks of treatment (p=0.046), maintaining in both cases the result achieved over time (Figure 1).
Conclusion: Our preliminary data show that ANI not only allows a rapid clinical improvement of SLE activity, but also of QoL as shown by the significant amelioration of PROs from the very first months of treatment. Further long-term studies on larger cohorts are needed to confirm and corroborate these results.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.