

Background: Despite the available range of disease-modifying antirheumatic drugs (DMARDs) for systemic lupus erythematosus (SLE) treatment, the issue of controlling disease activity has not been fully resolved. Our understanding of DMARDs withdrawal background in SLE remains limited.
Objectives: To explore the reasons for DMARDs discontinuation and estimate drug survival rates in patients with SLE.
Methods: A retrospective analysis of data obtained from SLE patients treated at the rheumatology department over the period 2011 to 2023 was conducted. The diagnosis was established according to the classification criteria available at that time. Our cohort represents the real-world structure of SLE clinical manifestations. Hydroxychloroquine (HCQ), methotrexate (MTX), azathioprine (AZA), mofetil mycophenolate (MMF), cyclophosphamide (CYC) and rituximab (RTX) were used as DMARDs in our cohort. The treatment duration and the reason for each case of DMARD discontinuation (lack of efficacy, side effects, de-escalation of therapy) were analysed. A marker of DMARDs efficacy was defined as the achievement of remission/low disease activity according to validated criteria [1]. HCQ in monotherapy was analysed as a separate group, and switching to combination therapy was considered as lack of its efficacy. Incomplete medical records were excluded from the analysis. Mean (standard deviation) or median (interquartile range) were reported. Chi-Square test was used with p-value<0.05. Kaplan-Meyer curves were produced to assess drug survival rates and compared by the log-rank test with p-value<0.05. CYC not included in the survival analysis due to a specific pattern of administration.
Results: Data from 160 SLE patients (mean age 37.1±11.2 years; 84.4% of females) were analyzed. Reasons for DMARDs discontinuation are presented in Table 1.
In our cohort HCQ had a higher “lack of efficacy” rate compared to MMF, CYC and RTX (p=0.014). The main reason could be its suboptimal prescription as monotherapy in cases with moderate disease activity (70.5% in “lack of efficacy” subgroup), when combination therapy was needed. The analysis of side effects as the reason for MTX and AZA withdrawal revealed rates 25.8% and 32.5% respectively, which are higher compared to other studies [2,3]. Among DMARDs used in lupus-nephritis, MMF revealed a substantially lower than CYC number of discontinuation cases due to side effects (p=0,02), while the levels of efficacy were comparable (p=0.27). RTX was used in cases of previous drugs inefficacy and provided disease activity control in 73,3 % patients. Remission or low disease activity was not achieved in 4 (2.5%) of patients with the administration of all available DMARDs. Figure 1 presents the Kaplan-Meier curves for DMARDs survival rates.
Reasons for DMARDs discontinuation in SLE.
| Reasons for DMARD discontinuation | HCQ (mono- therapy) | MTX | AZA | MMF | CYC | RTX | Chi-Square, p-value |
|---|---|---|---|---|---|---|---|
| N=38 | N=31 | N=40 | N=28 | N=32 | N=15 | ||
| Lack of efficacy, % | 44.7 | 25.8 | 30.0 | 17.9 | 9.4 | 13.3 | 0,014 |
| Side effects, % | 10.5 | 25.8 | 32.5 | 3.6 | 25.0 | 13.3 | 0,028 |
| De-escalation of therapy, % | 2.6 | 3.2 | 10.0 | 7.1 | 65.6 | 20.0 | <0,001 |
| Therapy is continued, % | 42.1 | 45.2 | 27.5 | 71.4 | - | 53.3 | 0,009 |
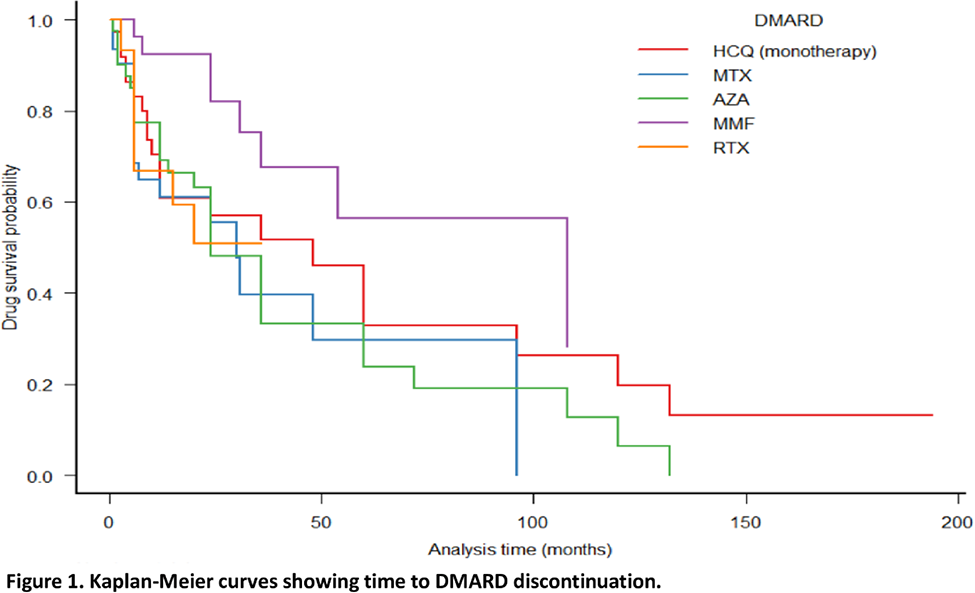
MMF demonstrated a better survival rate than MTX, AZA and RTX (log-rank test, p<0.05). The median duration (in months) of disease control for each DMARD was as follows: HCQ – 12 [4-38], MTX – 16 [6-29], AZA – 24 [6-36], MMF – 28 [19-42], CYC – 5 [3-6], RTX – 18 [6-26].
Conclusion: Our analysis describes the most common reasons for DMARDs discontinuation in SLE patients. Understanding of the revealed patterns of DMARDs use in our center could potentially reduce the number of DMARDs withdrawals and improve the drug survival profile. Our data demonstrate the unmet needs of DMARDs used in SLE patients in Ukraine.
REFERENCES: [1] Fanouriakis et al. Annals of the rheumatic diseases vol. 78,6 (2019): 736-745.
[2] Sato E.I. (2001). Lupus; 10(3): 162–164.
[3] Croyle L. et al. (2015) Lupus Science & Medicine., 2(1): e000105.
Acknowledgements: NIL.
Disclosure of Interests: None declared.