

Background: Systemic lupus erythematosus (SLE) is an autoimmune disease that can impact multiple organs. Among the various organs affected, lupus nephritis (LN) is a common complication related to increased morbidity and mortality [1]. According to the Taiwan National Health Insurance payment guidelines, mycophenolate mofetil or mycophenolate sodium are specifically recommended for patients with LN histopathologic classification III, IV, or V as determined by kidney biopsy. However, rheumatologists may have concerns about complications or contraindications associated with kidney biopsies for some SLE patients.
Objectives: To compare prognosis in SLE patients with or without kidney biopsy.
Methods: We utilized the Taiwan Chang Gung Research Database (CGRD) to assess the impact of undergoing kidney biopsy on the development of end-stage renal disease (ESRD) and mortality among SLE patients. A total of 10462 patients newly diagnosed with SLE between 2005 and 2020 were categorized into the biopsy and non-biopsy groups and were followed up until the end of 2022. Following propensity matching based on age, gender, and the diagnosis year of SLE, 345 and 1725 patients were included in the biopsy group and non-biopsy groups, respectively. The Cox proportional hazard regression model was employed, adjusting for factors such as age, gender, diagnosis year of SLE, comorbidities, drugs, creatinine, C3, C4, anti-dsDNA, and 24-hour proteinuria.
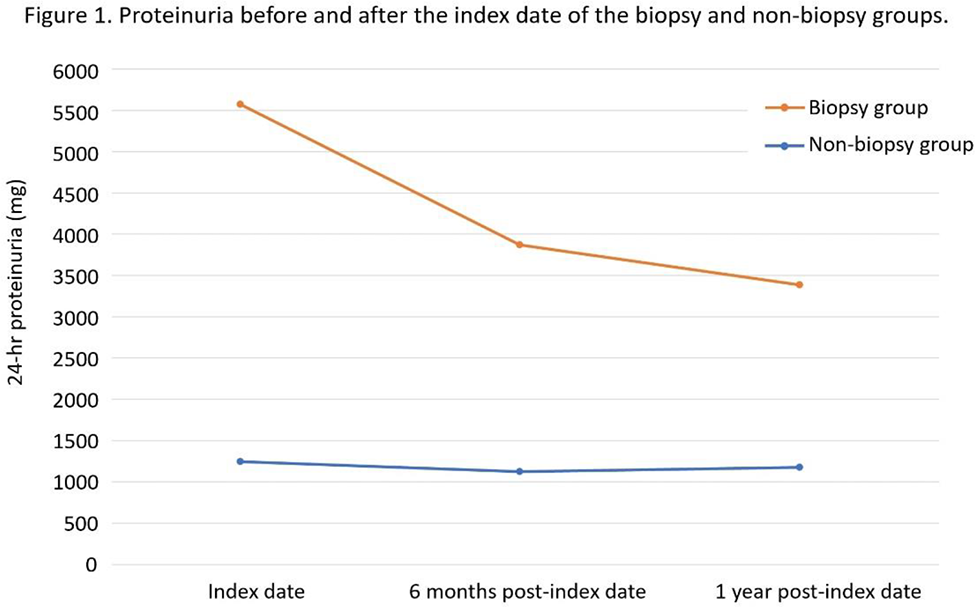
Results: In comparison to the non-biopsy group, SLE patients who undergo kidney biopsy exhibit a non-significant increase in the risk of ESRD (adjusted HR 1.58, 95% CI 0.91-2.74, p=0.11) and mortality (adjusted HR 1.51, 95% CI 0.83-2.73, p=0.17). Nevertheless, subgroup analyses based on different initial chronic kidney disease (CKD) stages reveal that SLE patients with an initial advanced CKD stage experience a non-significant decrease in the risk of both ESRD and mortality (Table 1). The reduction in 24-hour proteinuria before and after the index date (at 6 months and 1 year) between the two groups demonstrates a statistically significant difference (both with a p-value less than 0.01; Figure 1). The pathological results predominantly demonstrate LN classification of III, IV or V (76.5%). The percentages of each pathological result are as follow: Class I: 0.87%, Class II: 3.48%, Class III/ IV: 58.26%, Class III/ IV + V: 8.41%, Class V: 9.86%, Class VI: 0.29%, other than LN: 18.84%.
Conclusion: There is a non-significant increase in the risk of developing ESRD and mortality among SLE patients who undergo kidney biopsy. Conversely, the biopsy group shows a significantly higher reduction in proteinuria after kidney biopsy. The incongruent results regarding the risk of ESRD/ mortality and the decreased proteinuria warrant further investigation. In addition, SLE patients with initial advanced CKD stages who undergo kidney biopsy show a non-significant decrease in the risk of developing ESRD and mortality, suggesting a potential stricter indication for kidney biopsy in early CKD stages. The relatively small sample size after matching and unadjusted confounders such as immunosuppressant dosage may limit the interpretation of our results. Moreover, the relatively high percentage of histopathologic nephropathy other than LN may also restrict the benefits of kidney biopsy for specific drug prescriptions under insurance guidelines.
REFERENCES: [1] Croca SC, et al. Assessment of a lupus nephritis cohort over a 30-year period. Rheumatology (Oxford). 2011 Aug;50(8):1424-30.
Subgroup analyses based on different initial CKD stages for ESRD and mortality between the biopsy and non-biopsy groups.
| CKD Stage | Non-biopsy group
| Biopsy group
| ESRD
| p-value |
|---|---|---|---|---|
| 1-2 | 60/ 1692 | 52/ 257 | 1.41
| 0.35 |
| 3-5 | 11/ 33 | 41/ 88 | 0.69
| 0.48 |
| CKD Stage | Non-biopsy group
| Biopsy group
| Death
| p-value |
| 1-2 | 202/ 1692 | 48/ 257 | 1.66
| 0.16 |
| 3-5 | 16/ 33 | 24/ 88 | 0.87
| 0.84 |
Acknowledgements: The authors thank Po-Chuan Ko for the statistical assistance and wish to acknowledge for statistical and data analysis asistance and interpretation support by the Center for Big Data Analytics and Statistics, Chang Gung Memorial Hospital, Linkou.
Disclosure of Interests: None declared.