

Background: Over the past few years, the efficacy of several biological drugs for the treatment of Systemic Lupus Erythematosus has been evaluated. The efficacy of belimumab in reducing disease flares and organ damage is now strongly established [1].
Objectives: We will analyse the efficacy of belimumab in reducing disease activity after 3 and 6 months of treatment.
Methods: All the 74 patients from the Lupus Clinic of our Rheumatology Unit who received belimumab between 2014 and 2023 were selected for this study. 69% of the patients were also taking another DMARD (azathioprine, mycophenolate mofetil, cyclophosphamide, methotrexate), while 75% were also taking hydroxychloroquine. Clinical and epidemiological date were assessed at baseline and 3 and 6 months after first belimumab administration, with SLEDAI-2K, C3 and C4, anti-dsDNA antibodies, proteinuria over 24 hours and steroid dosage being particularly highlighted. Adverse events were also analysed.
Results: 94% of the patients selected were female, mean age 41±13.1 years, median 39 (range 15-83). The mean disease duration was 15.2±9 years, median 14 (range 3-37). The mean prednisone dosage was 9.3±5.7 mg, median 10 mg (range 0-25) and the mean SLEDAI-2K score was 7.3±3.7, median 8 (range 0-16). 32.8% of the patients had biopsy-confirmed Lupus nephritis (7% class II, 49% class III, 43% class IV, 1% class V). 27.74% of patients had arthritis, 39% muco-cutaneous manifestations, 8% serositis, 32.8% haematological changes and 2.73% neuropsychiatric involvement. 7.6% of the patients had C3 and C4 reduced. The mean anti-dsDNA values were 113.5±153 IU/mL, median 79 (range 0-1000).
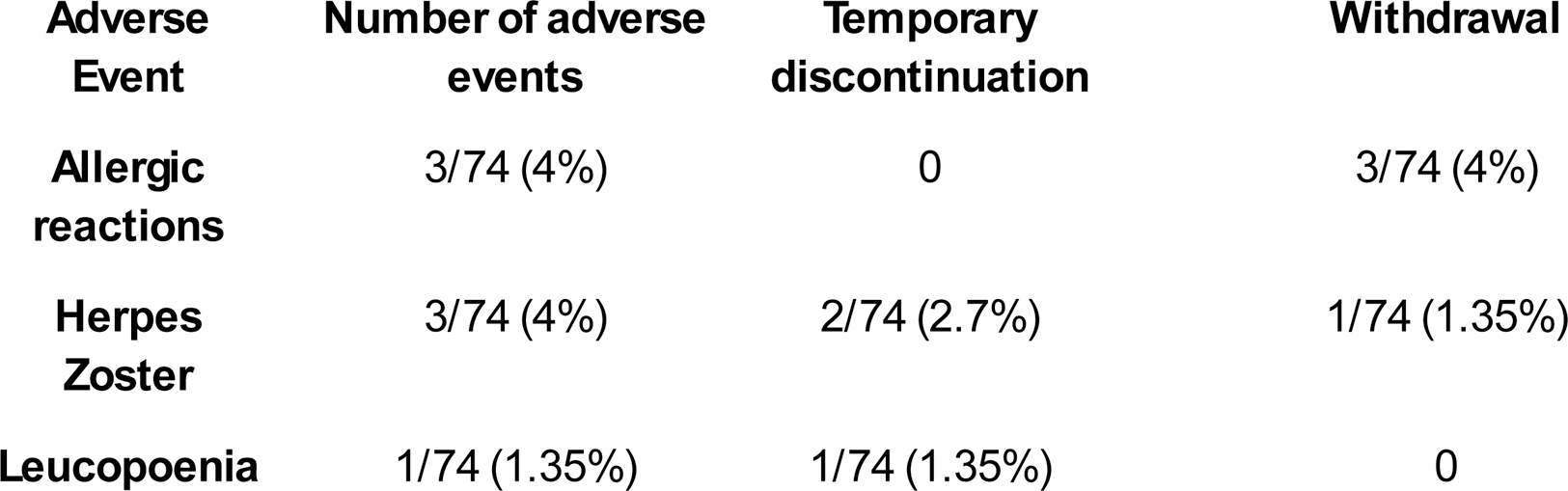
Clinical features at T0, T3 and T6 are summarised in Table 1. The data have confirmed a significant reduction of SLEDAI-2K, C3 and C4, anti-dsDNA antibodies, proteinuria and prednisone dosage from the first months of treatment. No significant differences were found regarding ESR, CRP, haemoglobin and creatinine. Adverse events are summarised in the Table 2.
Table 1
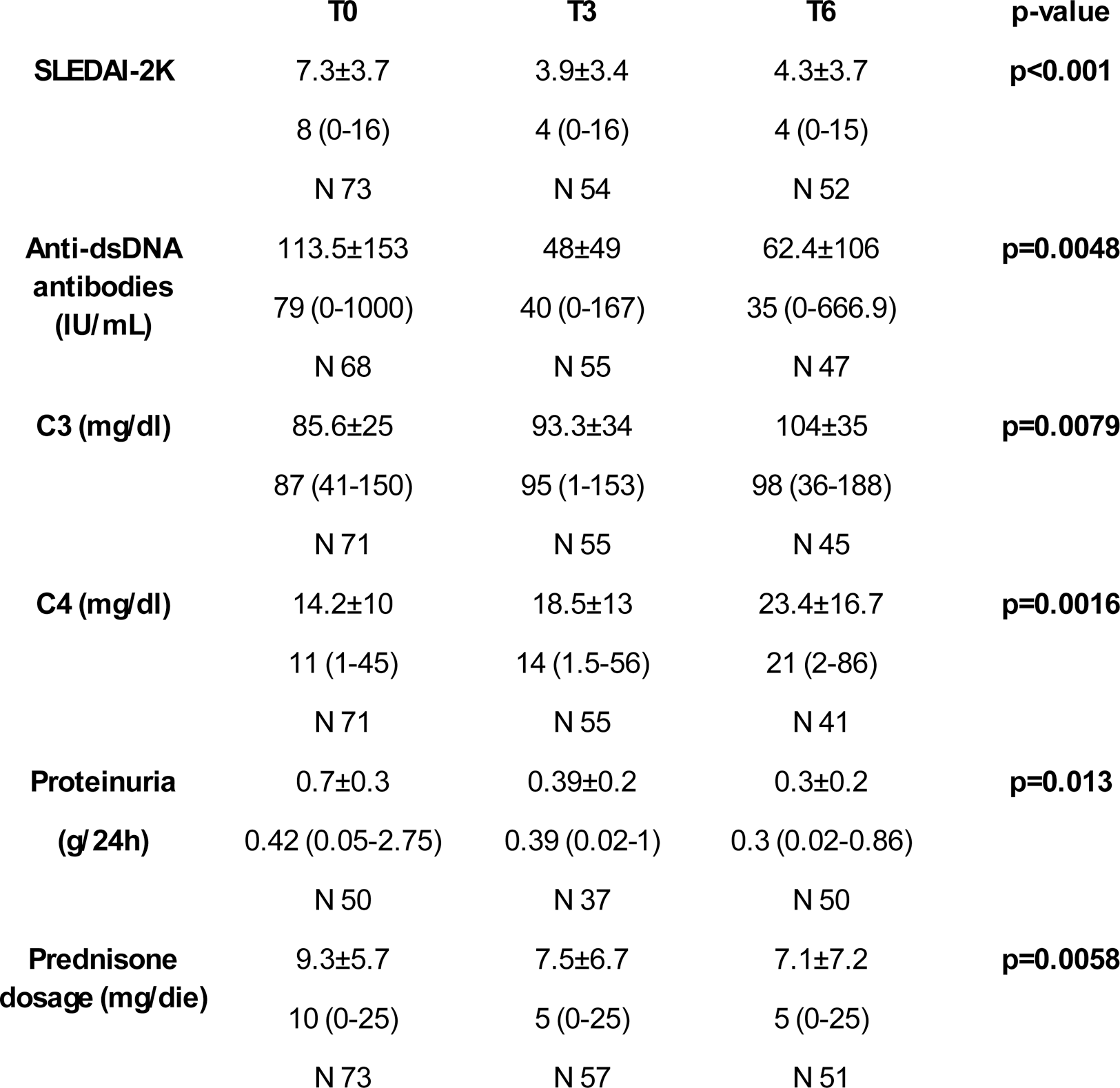
Table 2
Conclusion: Belimumab is effective in reducing disease activity already after 3 months of treatment, acting on the different clinical features of the disease, especially proteinuria. Adverse events are extremely rare. Therefore, the use of this drug is crucial in ensuring rapid steroid-sparing.
REFERENCES: [1] Cureus.2022 Jun 13;14(6):e25887
Acknowledgements: NIL.
Disclosure of Interests: None declared.