

Background: Until now, prospective long-term observational data on the disease course of anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) were missing in Germany. Therefore, the Joint Vasculitis Registry in German-speaking countries (GeVas) has been established to follow the course of patients with AAV.
Objectives: To present baseline data of patients with newly diagnosed and relapsing AAV enrolled in the GeVas registry.
Methods: GeVas is a prospective, web-based, multicenter, clinician-driven registry for the documentation of organ manifestations, damage, long-term outcomes, and therapy regimens in various types of vasculitis [1]. Recruitment started in June 2019 and is currently being conducted by 15 centers in Germany to date.
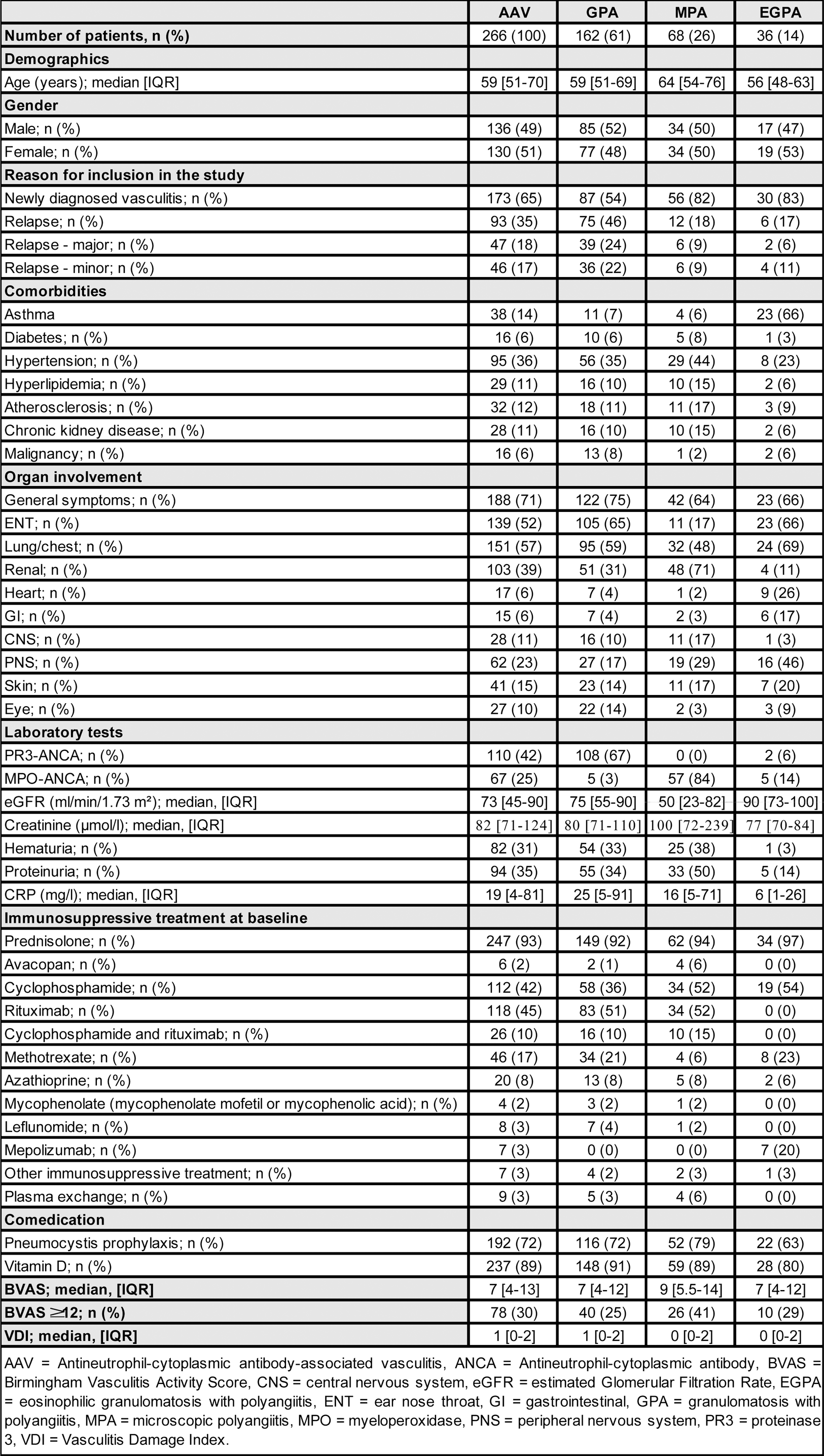
Results: Between June 2019 and October 2022, 266 patients with AAV were included in the GeVas registry, hereof 173 (65%) with new-onset and 93 (35%) with relapsing AAV. One hundred and sixty-two (61%) patients were classified as granulomatosis with polyangiitis (GPA), 68 (26%) as microscopic polyangiitis (MPA), and 36 (14%) as eosinophilic granulomatosis with polyangiitis (EGPA). The median age was 59 years (51-70 years, IQR), 130 (51%) patients were female, 136 (49%) males. Most patients were ANCA positive (177; 67%) and affected by general symptoms, pulmonary, ear nose throat (ENT), renal and neurological involvement. For induction of remission, the majority of patients received glucocorticoids (247, 93%) in combination with either rituximab (118, 45%) or cyclophosphamide (112, 42%) (Table 1).
Conclusion: Herein, we present baseline data from an inception cohort of AAV patients in the GeVas registry. Demographic characteristics are largely comparable to those in other European countries. Hoewever, differences were found regarding ANCA-status, frequencies of organ manifestations, and therapeutic regimen [2-4]. The GeVas registry will allow continued longitudinal observation and prospective outcome measures in AAV.
REFERENCES: [1] Iking-Konert C, et al. The Joint Vasculitis Registry in German-speaking countries (GeVas) – a prospective, multicenter registry for the follow-up of long-term outcomes in vasculitis. BMC Rheumatol 2021;5:40.
[2] Wójcik K, Wawrzycka-Adamczyk K, Włudarczyk A, et al. Clinical characteristics of Polish patients with ANCA-associated vasculitides-retrospective analysis of POLVAS registry. Clin Rheumatol 2019;38(9):2553-2563. (In eng). DOI: 10.1007/s10067-019-04538 w.
[3] Solans-Laqué R, Fraile G, Rodriguez-Carballeira M, et al. Clinical characteristics and outcome of Spanish patients with ANCA-associated vasculitides: Impact of the vasculitis type, ANCA specificity, and treatment on mortality and morbidity. Medicine (Baltimore) 2017;96(8):e6083. DOI: 10.1097/md.0000000000006083.
[4] Ponte C, Khmelinskii N, Teixeira V, et al. Reuma.pt/vasculitis - the Portuguese vasculitis registry. Orphanet J Rare Dis 2020;15(1):110. (In eng). DOI: 10.1186/s13023-020-01381-0.
Table 1. Demographics and clinical characteristics of AAV patients enrolled in GeVas registry (06/2019 – 10/2022).
Acknowledgements: GeVas was supported by unrestricted grants by DGRh, John Grube Foundation, Vifor Pharma, and Roche Pharma.
Disclosure of Interests: Sabrina Arnold: None declared, Pia Wallmeier: None declared, Arlette Tais: None declared, Gabriele Ihorst: None declared, Marco Janosche: None declared, Fabian Schubach: None declared, Peer Malte Aries: None declared, Raoul Bergner personal fees for lectures from Abbvie, BMS, Chugai, GSK, Galapagos, MSD, Novartis, Roche,, consultant of: VIFOR, GSK, Phillip Bremer personal fees for lectures from: UCB Pharma, Chugai, Celgene, Abbvie, Janssen, Galapagos, Lilly, Sanovi-Aventis, Novartis, AstraZeneca, GSK, Roche, Consultant of: Galapagos, Norman Görl: None declared, Eva Gutdeutsch: None declared, Bernhard Hellmich personal fees for lectures or advisory services from Amgen, AstraZeneca, BMS, Boehringer Ingelheim, Chugai, InflaRx, GSK, Pfizer, Phadia, MSD, Roche, Novartis and Vifor outside the submitted work, personal fees for lectures or advisory services from Amgen, AstraZeneca, BMS, Boehringer Ingelheim, Chugai, InflaRx, GSK, Pfizer, Phadia, MSD, Roche, Novartis and Vifor outside the submitted work, Jörg Henes: None declared, Bimba Hoyer: None declared, Antje Kangowski: None declared, Ina Kötter: None declared, Martin Krusche: None declared, Tim Magnus: None declared, Claudia Metzler: None declared, Ulf Müller-Ladner: None declared, Jana Petersen: None declared, Anke Reichelt de Tenorio: None declared, Matthias Schaier: None declared, Jan Schirmer Grant/research support from: John-Grube Foundation, Deutsche Gesellschaft für Rheumatologie, Ulf Schönermark Lecture fees from Janssen-Cilag and Alexion/AstraZeneca, Study fees and consultancy fees from Alexion/AstraZeneca, Ablynx/Sanofi and Chemocentryx/Vifor., Jens Thiel personal fees for lectures or advisory services from Abbvie, AstraZeneca, BMS, GSK, Pfizer, Novartis and Vifor, personal fees for lectures or advisory services from Abbvie, AstraZeneca, BMS, GSK, Pfizer, Novartis and Vifor, Leonore Unger: None declared, Nils Venhoff Speakers bureau: Roche and Vifor: speaker honoraries, Consultant of: Roche and Vifor: advisory boards, Grant/research support from: John- Grube Research Award 2021, Julia Weinmann-Menke: None declared, Christoph Iking-Konert Speakers bureau: Lecture fees from: Chugai, GSK, Roche, and Vifor, SConsultant of: Consulting fees from: Chugai, GSK, Roche, and Vifor, Grant/research support from: Research grants for GeVas: Roche, Vifor, DGRh, John Grube Foundation,, Peter Lamprecht Speakers bureau: BMS, FOFM, GSK, Janssen, UCB, and Vifor Pharma, Consultant of: GSK, and Vifor Pharma, Grant/research support from: BMBF, DFG, DGRh, John Grube Foundation, and Vifor Pharma.