

Background: Adherence to treatment guidelines and the optimization of therapy during the course of the disease remain significant issues that can influence patient outcomes. The complexity of AAV management, coupled with the potential side effects of the drugs used, underscores the necessity for a more personalized treatment approach that takes into account patient and course-specific factors.
Objectives: The primary objective of this study is to evaluate the adherence to treatment guidelines for the induction and maintenance treatments of AAV. To assess the rate and reasons for early treatment modifications during the remission induction phase.
Methods: In this retrospective study, Data were collected from medical records, including demographic information, treatment regimens adherence to guidelines, and treatment modifications during the disease course. Used treatments and their compliance with guidelines and suggested doses during remission induction and maintenance were evaluated. The time and reasons for the change in treatment were investigated.
Results: 112 ANCA vasculitis patients, consists of GPA 75 (67%), EGPA 22(19.6%), MPAN 15 (13.4 %), were included in the study. Median age of total cohort was 55 (Min: 18 - Max: 86]). Patients characteristics were summarized in Table 1.
93% of GPA patients, 85% of EGPA, and 75% of MPAN patients showed compliance to the guidelines in terms of cyclophosphamide induction. Full adherence to guidelines was observed in RTX administration.
Changes in treatment during the remission induction phase occurred in 21.3% of GPA patients, 9% of EGPA patients, and 13% of MPAN patients. The median time to treatment modification was 4 month (min max 0-5) for GPA patients, 2months (min-max 1-3) for EGPA patients, and 1 month (min-max 0-2) for MPAN patients. The primary reasons for treatment modification are summarized in Table 2. Side effects identified were cytopenia and liver function test abnormalities.
In the maintenance treatment phase, adherence and appropitate dosages of medications in such as Methotrexate (MTX), Mycophenolate Mofetil(MMF), Azathioprine(AZA) and RTX were evaluated. In whole cohort, adherence-using appropriate dose was 66% for MTX,36% for MMF, and 28% for AZA. Of note, adherence and optimal dosing of RTX was found 100%.
In the first stage of induction, rituximab was chosen as remission induction therapy in 13 patients. Of these,10 were diagnosed with GPA, 2 with EGPA, and 1 with MPA. When the reasons for choosing RTX are examined; RTX was preferred due to the risk of gonadal toxicity in 5 patients, physician preference in 3 patients, patient preference in 1 patient, malignancy in 1 patient, cytopenia in 1 patient, resistant disease in 1 patient, and chronic liver disease in 1 patient.
In comparison with patients who were chosen for cyclophosphamide during induction, it was statistically significant that those selected for RTX were younger (average ages 55 vs. 41, p=0.002). In follow-up, 25% of GPA patients were able to discontinue steroids, compared to 4.5% in EGPA and 6.7% in MPAN patients.
Conclusion: Even in the case of cyclophosphamide, a mainstay in vasculitis treatment, adherence to guidelines is not fully achieved. Considerable proportion of patients undergoing early modifications in their induction therapy. The decision to alter therapy early, particularly in severe cases, could be a reaction to the lack of a clear treat-to-target approach. Such discrepancies highlight the need to tailor treatment strategies for more flexible and personalized treatment approaches. We conclude that further research is necessary to evaluate the effects of early treatment changes and to develop more structured approaches, possibly including treat-to-target strategies, and strategies for maintenance therapy for optimal patient care.
REFERENCES: [1] Hellmich, B., Sanchez-Alamo, B., Schirmer, J. H., Berti, A., Blockmans, D., Cid, M. C., Holle, J. U., Hollinger, N., Karadag, O., … Jayne, D. (2024). EULAR recommendations for the management of ANCA-associated vasculitis: 2022 update.
Annals of the rheumatic diseases
,
83
(1), 30–47.
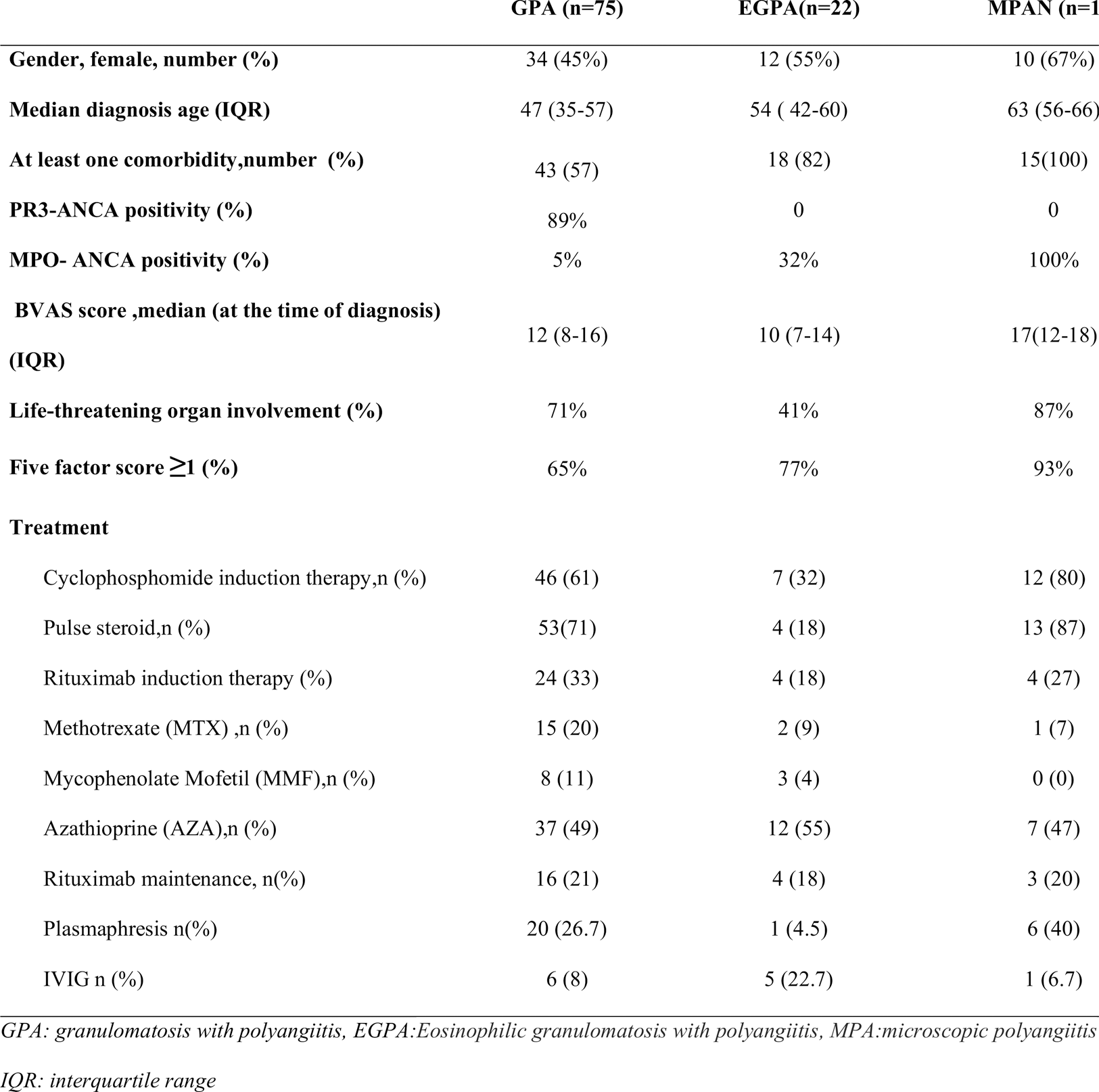
Table 2. Timing and reasons for treatment change

Acknowledgements: NIL.
Disclosure of Interests: None declared.