

Background: Primary antiphospholipid syndrome (PAPS), and systemic lupus erythematosus secondary APS (SAPS) are challenging diseases to predict and lack effective therapeutic drugs. Rituximab (RTX) can deplete CD20+ peripheral B cells and Telitacicept (TA) is a dual target inhibitor targeted both BAFF and APRIL. The strategy of TA following RTX immunotherapy is worth discussing in the treatment of APS.
Objectives: This study was to investigate the efficacy and safety of TA following RTX therapy for refractory/severe APS.
Methods: This prospective study enrolled PAPS/SAPS patients were treated with RTX monotherapy or a sequential therapy, that was 200-500mg RTX infusion after screening, then evaluated 4 weeks later (0w) and TA injection (mainly 160mg ih qw) for 24 weeks (24w). The standard background were glucocorticoid (GC), hydroxychloroquine, and anticoagulants/ antiplatelet medications. The complete remission rate (CR) was the proportion of increased antiphospholipid antibodies (aPL) or lupus anticoagulant (LA) value decreased to normal range and overall remission rate (OR) was the aPL titers decreased into two times above the normal value or LA decreased to normal range. The incidence of new APS related clinical events, overall remission ratio of thrombocytopenia (TP), and adverse events (AE) were also analysis.
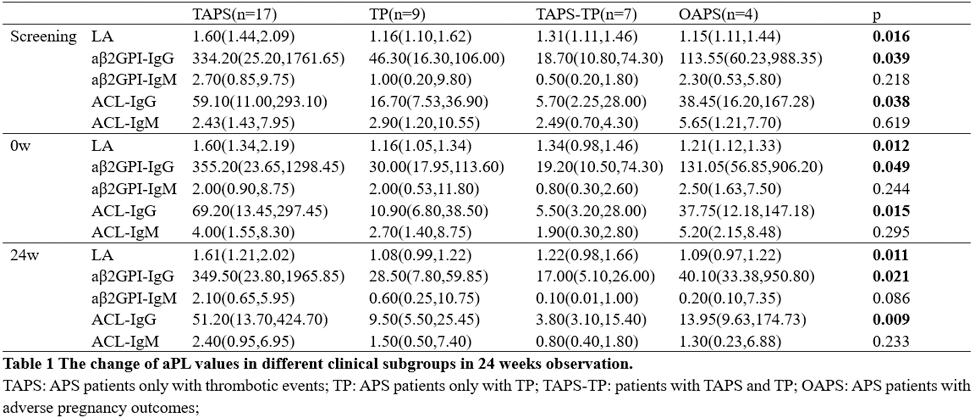
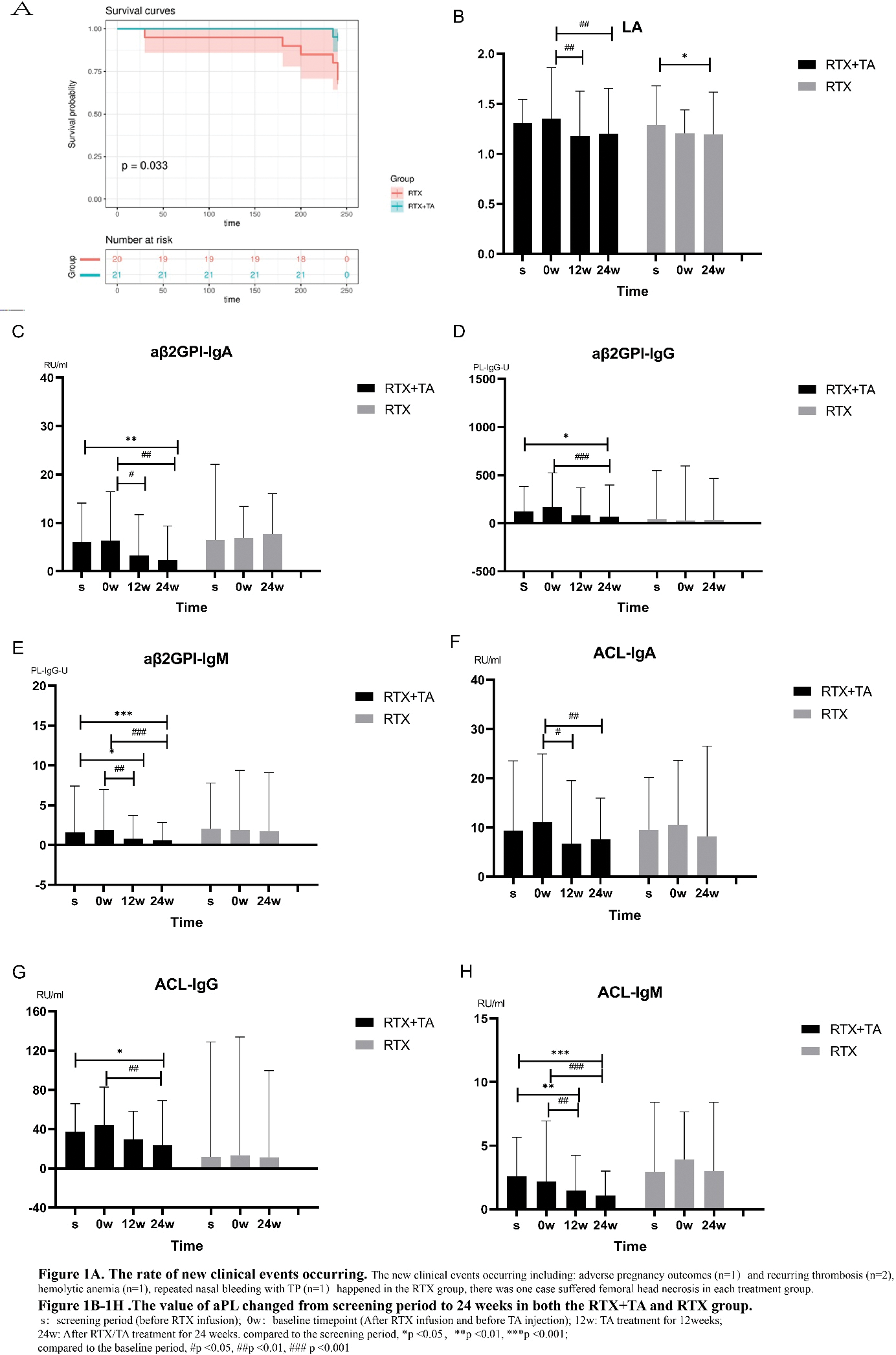
Results: There were 21 patients in RTA+TA group and 20 in RTX group who completed this study. During the screening period (before the use of RTX), the triple positivity of aPL in the study was 53.7%, among which the aβ2GPI-IgG showed the highest positivity rate at 95.1%. It was observed that the stratification of aβ2GP1-IgG and ACL-IgG were higher in the RTX+TA group than in RTX. Among the 4 difference clinical subgroups, the patients only with thrombotic APS (TAPS) showed the highest positive rate of LA, and much higher values of LA, aβ2GPI-IgG, ACL-IgG compared to TP, TP+TAPS and Obstetric APS (OAPS) subgroups. After RTA +TA immunotherapy, the values of these aPLs in TAPS group were still higher than others. The median values of ACL-IgG and ACL-IgM decreased sharply from 0w to 24w in the TAPS group with additional TA treatment (Table 1). With 24 weeks treatment, the new clinical events occurring ratio was significant lower in the RTX+TA group than in the RTX group (1/21 vs. 6/20) (Figure 1A). There were eight patients with TP in each treatment group. The PLT count in the RTX+TA group was significantly lower than that in the RTX group during the screening period, while at baseline and 24 weeks timepoint, the data in the RTX+TA group were higher than in the RTX group. The OR rates of aβ2GPI-IgA, aβ2GPI-IgM, ACL-IgM, ACL-IgA, and ACL-IgG in the RTX+TA group were more than 50%, especially the OR of aβ2GPI-IgA and aβ2GPI-IgM were significantly higher than RTX group. It was obviously that all aPL values in the RTX+TA group were significantly lower compared to both the screening and baseline timepoint (after RTX treatment and before TA administration). However, in the RTX group, only the LA value at 24 weeks significantly decreased from that of the screening timepoint (Figure 1B-1H ). The decreased values were also different between the two groups, the reduction of aPL values was higher in the RTX+TA group. Both groups received a GC dose of 10 mg/d during the screening period, and the RTX+TA group exhibited a lower dose than the RTX group at 24 weeks (5.0 mg/d vs 7.5 mg/d, p=0.011). AE was mild in both treatment patients, including 14 RTX infusion-related reactions and two RTX-associated secondary infections, primarily occurring within one month after infusion. TA injection had 5 slight and one moderate local reactions. No additional infections were reported after the administration of TA.
Conclusion: In this 24w observational study, TA followed with RTX therapy was a promising strategy for refractory/severe APS. This immunotherapy strategy effectively reduced the incidence of new-onset APS-related events and lowered aPL values without increasing the number of new infections using TA.
REFERENCES: NIL.
Acknowledgements: Funded by ECCM Program of Clinical Research Center of Shandong University (No.2021SDUCRCB010), Shandong Innovation and Entrepreneurship Community of Antibody Drug Project (SDUZHJ(2023) 0716.
Disclosure of Interests: None declared.