

Background: The pro-inflammatory interleukin (IL)-16 has been implicated in various rheumatic disorders. Urinary (u-) IL-16 has been associated with proliferative nephritis in systemic lupus erythematosus (SLE) and is also expressed in inflammatory foci in the kidney [1]. Nevertheless, there is a lack of data on plasma (p-) and u-IL-16 in other immune-mediated kidney diseases including antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV).
Objectives: To determine the IL-16 (p- and u-) levels in a cross-sectional cohort of AAV patients compared to controls and to assess associations with clinical variables.
Methods: P- and u-IL-16 were measured using a commercial sandwich ELISA. The cut-off for detection was set to ≥ 10 pg/mL. Disease activity was assessed using the Birmingham Vasculitis Activity Score (BVAS). Kidney biopsies were classified according to histopathological class [2]. Immunohistochemical staining of IL-16 in kidney biopsies was performed. Healthy controls were included for comparison (plasma n=38 and urine n=48).
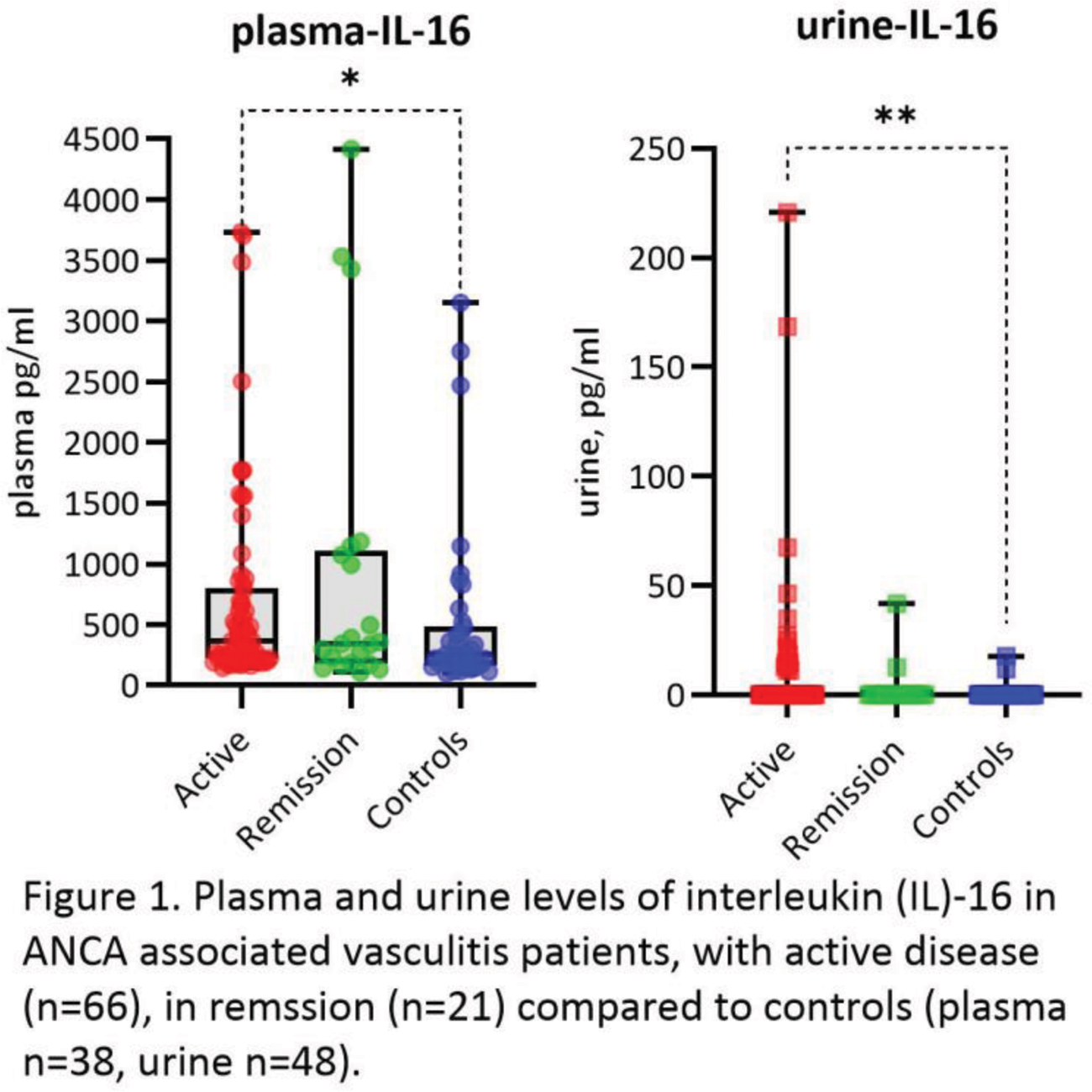
Results: There were 87 AAV patients included in the study, 47 (54%) males, 57 (65.5 %) anti-PR3 positive and 30 (34.5 %) anti-MPO positive whereas 63 (72.4 %) patients had GPA and 24 (27.6 %) MPA. There were 66 (76%) patients with active disease (BVAS >0, median 14, range 23) and 39 (44.8%) had kidney involvement. All patients had detectable p-IL-16 levels. U-IL-16 was found in 18 (21%) AAV patients, 15 with active disease and kidney involvement, 1 active disease without kidney involvement, and 2 in remission with prior kidney involvement. Two controls had detectable u-IL-16. Active patients had higher levels of p- and u-IL-16 compared to controls (median 364.7 pg/ml vs 254.5 pg/ml, U = 911.0, p = 0.021 and median (range) 0 (220.8) pg/ml vs 0 (17.8) pg/ml, U = 1258, p = 0.003), but there was no difference between patients in remission and controls, Figure 1. U-IL-16 significantly correlated with BVAS (r s = 0.365, p<0.001). Both p- and u-IL-16 correlated with eGFR (r s = - 0.283, p = 0.022 and r s = - 0.347, p = 0.005) in active patients and u-IL-16 also correlated with urine albumin-creatinine ratio (r s = 0.336, p=0.009). Active patients with kidney involvement had higher levels of u-IL-16 compared to patients without (median(range) 0 (220.8) pg/ml vs 0 (67.4) pg/ml, U = 349.0, p = 0.002), but no differences in p-IL-16 levels were seen between groups. There were no differences in levels of p- or u-IL-16 in active patients with ongoing immunosuppression compared to those without. Furthermore p- or u-IL-16 levels did not differ between diagnosis (GPA/MPA) or ANCA-serotype. Kidney biopsies were obtained from 43 patients. P-IL-16 levels correlated negatively with the percentage of normal glomeruli (r s = - 0.311, p = 0.043) and u-IL-16 levels positively with the percentage of crescentic glomeruli (r s = 0.329, p = 0.031). Thirty-seven kidney biopsies were classified according to histopathological type: Focal (n=20), Crescentic (n=8), Mixed (n=6), and Sclerotic (n=3). There was a significant difference in u-IL-16 levels between these groups (H(3) = 8.68, p = 0.034) with the highest u-IL-16 levels in the crescentic and sclerotic classes. A pilot staining of kidney biopsies showed detectable IL-16 in the interstitium and inflammatory infiltrates.
Conclusion: The increased u- IL-16 levels found in AAV patients with active disease and kidney involvement strongly imply a role in the inflammatory kidney process. Moreover, IL-16 was expressed in the kidney tissue and urine levels reflected the severity of kidney involvement which strengthens the findings. The results suggest that urine IL-16 could be used as a biomarker of kidney involvement, as previously shown in severe types of lupus nephritis.
REFERENCES: [1] Hayry A, et al. Interleukin (IL) 16: a candidate urinary biomarker for proliferative lupus nephritis. Lupus Sci Med. 2022;9(1).
[2] Berden AE, et al. Histopathologic classification of ANCA-associated glomerulonephritis. J Am Soc Nephrol. 2010;21(10):1628-36.
Acknowledgements: NIL.
Disclosure of Interests: Anna Juto: None declared, Francesca Faustini: None declared, Aliisa Häyry: None declared, Anders Larsson: None declared, Vivianne Malmström: None declared, Annette Bruchfeld Alexion, Amgen, Astra-Zeneca, Bayer, Boehringer-Ingelheim, Chemocetryx, Fresenius, CSL-Vifor (during the last three years)., Alexion, Amgen, Astra-Zeneca, Bayer, Boehringer-Ingelheim, Chemocetryx, Fresenius, CSL-Vifor (during the last three years)., Aleksandra Antovic: None declared, Vilija Oke: None declared, Iva Gunnarsson: None declared.