

Background: Behçet’s syndrome (BS) significantly increases morbidity and mortality, especially among patients with vascular (i.e. vascular-BS) and neurological involvement (i.e. neuro-BS). Cyclophosphamide and glucocorticoids have been for long the standard remission-induction therapy for severe BS, but disease flares require repeated treatment courses and lead to high cumulative doses. Current International guidelines advocate the combination of high-dose steroids with either cyclophosphamide or TNFi as induction therapy for severe Vascular- and Neuro-BS.
Objectives: We aimed to assess the efficacy and safety of infliximab and cyclophosphamide in severe BS.
Methods: In this phase 2, Bayesian, randomized, multicenter, controlled trial, we enrolled patients aged 12 years or older fulfilling the ISG criteria for BS and presenting major vascular or CNS involvement. They were randomly assigned to receive either intravenous Infliximab (5mg/kg at week 0, 2, 6, 12, and 18) or Cyclophosphamide (0.7-1.2 g/m 2 at week 0, 4, 8, 12, 16 and 20), combined with a standardized corticosteroid tapering regimen. Randomization was stratified by the main BS involvement at baseline (vascular or neurological involvement) and according to newly diagnosed or relapsing BS status. The primary outcome was a complete response at week 22, defined by resolution of all baseline vascular-BS or neuro-BS clinical manifestations, CRP level normalization and radiological remission under a prednisone dose ≤0.1mg/kg/day. Patients were analyzed in an intent-to-treat basis.
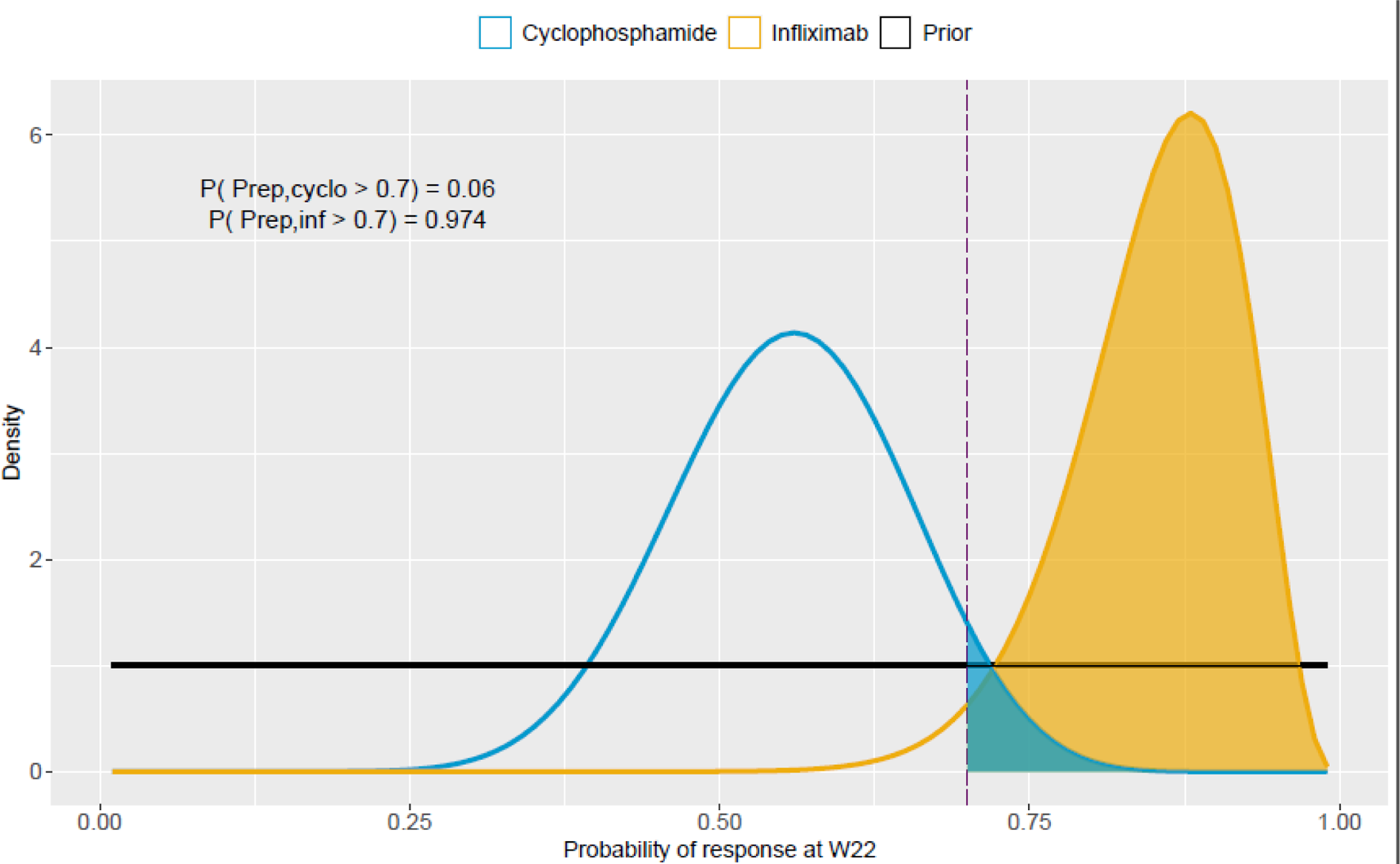
Results: Between May 2018 and April 2021, 52 patients with BS were enrolled; baseline characteristics are presented in Table 1 . Complete response was achieved by 22/27 (81%) and 14/25 (56%) patients in the infliximab and cyclophosphamide groups, respectively (estimated difference 22.9; credible interval 2.3 to 42.7). The posterior probability that the rate of complete response achieve at least 70% by week 22 was 97.4% for infliximab and 6.0% for cyclophosphamide ( Figure 1 ). By week 22, 17/19 (94%) and 10/18 (56%) vascular-BS patients achieved complete response in the infliximab and cyclophosphamide groups, respectively. For neuro-BS, 5/8 (71%) and 4/7 (57%) patients receiving infliximab and cyclophosphamide, respectively, achieved complete response. Relapse occurred in one (4%) and four (16%) patients receiving infliximab and cyclophosphamide, respectively. Forty-two mild-to-moderate adverse events (mainly infections) were recorded in 8/27 (29.6%) and 16/25 (64%) patients under infliximab and cyclophosphamide, respectively. Serious adverse events were similar between groups.
Conclusion: Among patients with severe BS, induction therapy with infliximab had better complete remission rate at 22 weeks and a safer profile as opposed to cyclophosphamide.
Baseline characteristics of patients within the ITAC trial
| Variable | Cyclophosphamide
| Infliximab
| Total
|
|---|---|---|---|
| Age, years | 40.9 (34.7;48.2) | 38.1 (29.3;46.0) | 39.4 (34.5;47.4) |
| Female sex | 10 (40.0) | 12 (44.4) | 22 (42.3) |
| BS newly diagnosed at enrollment | 19 (76.0) | 20 (74.1) | 39 (75.0) |
| Major vascular involvement | 18 (72.0) | 19 (70.3) | 37 (71.1) |
| Arterial aneurysm | 8 (32.0) | 7 (25.9) | 15 (28.8) |
| Arterial thrombosis or stenosis | 10 (40.0) | 3 (11.1) | 13 (25.0) |
| Deep venous thrombosis | 10 (40.0) | 11 (40.7) | 21 (40.4) |
| Central nervous system involvement | 7 (28.0) | 8 (29.6) | 15 (28.8) |
| Brainstem lesions | 4 (16.0) | 4 (14.8) | 8 (15.3) |
| Supra-tentorial lesions | 2 (8.0) | 5 (18.5) | 7 (13.4) |
| Myelitis | 0 | 3 (11.0) | 3 (5.7) |
| Cerebral thrombophlebitis | 1 (4.0) | 1 (3.7) | 2 (3.8) |
| C-reactive protein, mg/L | 4 (2.1;12.6) | 4 (1.6;11.5) | 3 (1.0;6.7) |
| Previous treatments | |||
| Corticosteroids | 10 (40.0) | 14 (52) | 24 (46.0) |
| Colchicine | 4 (16.0) | 5 (18.5) | 9 (17.0) |
| Azathioprine | 4 (16.0) | 4 (15) | 8 (15.0) |
| Mycophenolate mofetil | 0 | 1 (3.7) | 1 (2.0) |
Categorical data are presented as n (%), whereas quantitative data as median [interquartile range].
Posterior probability of the difference in complete response rates at Week 22
The posterior probabilities with a prior (Beta (7,3)) are provided.
REFERENCES: NIL.
Acknowledgements: We thank Dr Delphine Leclercq, Dr Marine Bravetti, and Dr Alessandra Bartoli (Pitié - Salpétrière Hospital) for composing the Endpoint Adjudication Committee. We also thank all the patients who participated, and their relatives; the staff involved in caring for trial participants at all trial sites, and Cendrine Chaffaut and Nabil Raked for their involvement in the data monitoring and the statistical analysis.
Disclosure of Interests: None declared.