

Background: VEXAS (Vacuoles, E1 Enzyme, X-linked, Autoinflammatory, Somatic) syndrome is a severe autoinflammatory disease in adults resulting from somatic mutations in the UBA1 gene, associated with myelodysplastic syndromes (MDS) in 40-50% patients. The therapeutic management of VEXAS syndrome is poorly standardized, although the efficacy of azacitidine (AZA) has been reported in two French series: a prospective one (n=12) [1] and a retrospective one (n=11) [2].
Objectives: The aim of this study is to update data on the efficacy and safety of AZA in a significant cohort of VEXAS patients (n=57) and to investigate the impact of the treatment on the UBA1 clone.
Methods: Multicenter retrospective study conducted within the French national registry (n=305), including patients with genetically confirmed VEXAS syndrome who have received at least one full cycle of AZA. Complete inflammatory response (CR) was defined by a clinical remission, C-reactive protein (CRP) ≤ 10 mg/L, and a glucocorticoid therapy ≤ 10 mg/day with no other immunosuppressive treatment. Partial inflammatory response (PR) was defined by a significant improvement (>50%) in these parameters without meeting the CR criteria. Hematologic response was defined as the improvement of pre-existing cytopenias to reach the following thresholds: Hb > 10 g/dL or transfusion independence, ANC > 1000/mm3, and platelets > 100 G/L.
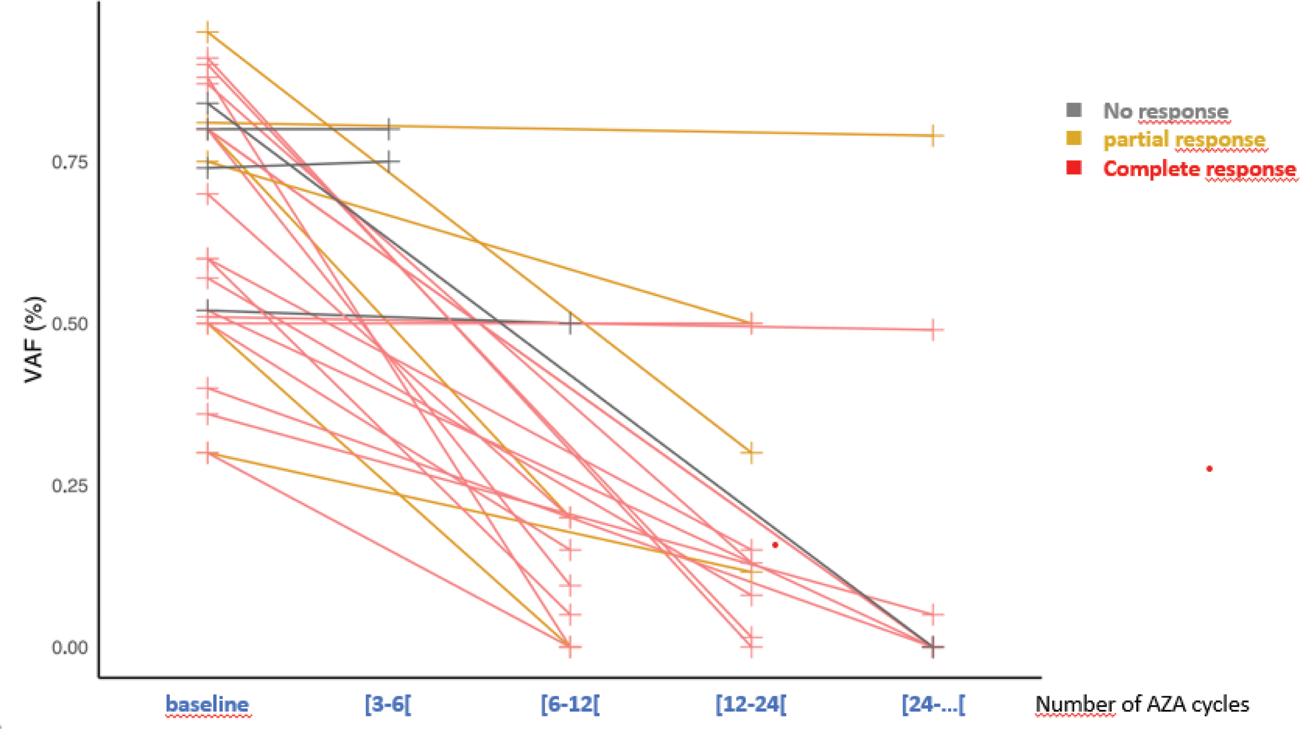
Results: Fifty-seven patients were included (100% male, median age 71 years), with 50 having MDS (mostly with multilineage dysplasia in 62%), predominantly classified as low risk according to the R-IPSS (very low/low/intermediate/high risk in 5/24/9/6 patients, respectively). NGS myeloid panel analysis (n=45) identified TET2/DNMT3A mutations in 22 (49%) and at least one other mutation in 13 (28%) patients. At AZA onset, 95% of patients were receiving glucocorticoids (median dose: 20 mg/day, median duration of exposure: 23 months); they had also received a median of 2 other immunosuppressants [0-12]. The median number of administered AZA cycles was 13 [1–114]. Twenty-eight patients (49%) discontinued AZA during follow-up: 3 before cycle 3, 25 after cycle 3 for infection (n=7), response (n=4), primary failure (n=3), secondary failure (n=4), allogeneic stem cell transplantation (n=6) and diagnosis of multiple myeloma (n=1). Response was assessed in 51 patients (45 with MDS and 6 without MDS) who had received at least 3 cycles of AZA. An inflammatory response (CR or PR) was observed in 39/51 (77%) patients, with 61% achieving a complete response. Five out of the 6 non-MDS patients achieved a response (complete response). Glucocorticoids could be reduced to <10 mg/day in 26/38 (68%) patients. Among responders, 29/39 maintained a response at the latest follow-up (74%), despite discontinuation of AZA in 7 patients. The median follow-up was 28 [4-163] months. Regarding hematologic response, anemia improved in 33/42 (79%) patients, among whom 25/32 (78%) achieved transfusion independence. Thrombocytopenia improved in 17/27 (63%), and neutropenia improved in 2/4 (50%). The follow-up of UBA1 mutation under AZA, currently available for 28 patients, showed Variant Allele Frequency (VAF) negativization in 9 (32%) patients and a decrease of more than 50% in 12 (43%) patients. This decrease was associated with clinical response in the majority of patients (Figure 1). During follow-up, 9 patients died under AZA: 3 from infections before cycle 3, 3 from infections after cycle 3, and 3 from causes unrelated to treatment.
Conclusion: We have confirmed the efficacy of AZA in a significant cohort of VEXAS patients, particularly in those without associated MDS. The treatment appears to have a direct impact on clonal evolution. Close monitoring seems necessary given the infectious risk in these patients who have received glucocorticoids and immunosuppressive drugs.
REFERENCES: [1] Mekinian et al, Leukemia 2022; 36:2739-2742.
[2] Comont, Br J Haematol 2022;196:969-974.
Acknowledgements: NIL.
Disclosure of Interests: None declared.