

Background: Pregnancies in patients with systemic lupus erythematosus (SLE) are associated with an increased risk of adverse maternal and fetal outcomes. Hydroxychloroquine (HCQ) decreases disease activity and the occurrence of flares, but little is known about the usefulness of monitoring HCQ blood levels during pregnancy in these patients.
Objectives: The objective of this study was to evaluate the correlation between HCQ blood levels in the first trimester of pregnancy and the maternal and fetal outcomes in patients with SLE.
Methods: We included pregnant SLE patients enrolled in the French prospective “Groupe de recherche sur la Grossesse et les Maladies Rares” (GR2) study, with at least one available first-trimester whole-blood HCQ level. We evaluated several cut-offs: 500 ng/ml, 750, and 1000 ng/ml, as a therapeutic target, and a threshold of 200 ng/ml for assessing severe non-adherence. The primary outcomes were maternal flares (mild/moderate or severe, identified by SLEDAI Flare Index) during the 2nd and 3rd trimesters of pregnancy, and the occurrence of any adverse pregnancy outcomes (APOs: fetal/neonatal death, placental insufficiency resulting in preterm delivery, and/or small-for-gestational-age neonates).
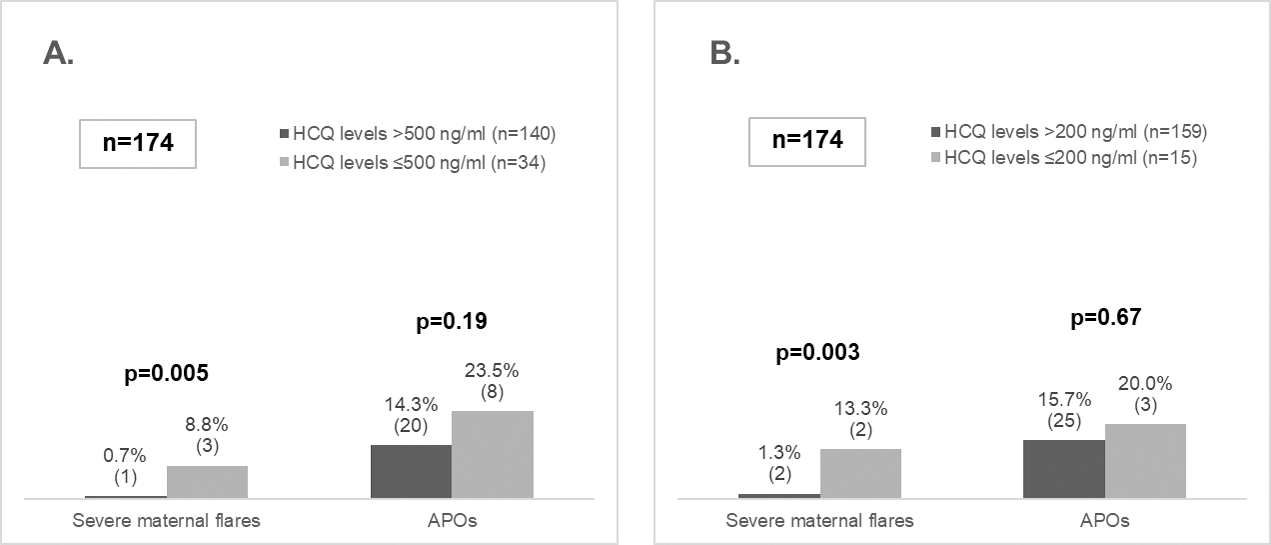
Results: We included 174 patients (median age 32.1 years -IQR 28.8-35.2-). Thirty (17.2%) patients had flares, 4 (2.3%) being severe. APOs occurred in 28 patients (16.1%). There were no significant differences in APOs according to HCQ levels, neither among patients with infra-therapeutic HCQ levels ≤500 ng/ml vs >500 ng/ml (23.5% vs 14.3%, p=0.19), nor among those with non-adherent HCQ levels ≤200 ng/ml vs >200 ng/ml (20.0% vs 15.7%, p=0.67). For overall maternal flares, there were no significant differences based on varying HCQ cut-offs. However, there was a significant increase in severe flares among patients with infra-therapeutic (HCQ ≤500 ng/ml) (8.8% vs 0.7% in those > 500 ng/ml, p=0.005), and among non-adherent (≤ 200 ng/ml) HCQ levels (13.3% vs 1.3% in those > 200 ng/ml, p=0.003) (Figure 1).
Conclusion: First-trimester HCQ blood levels did not predict APOs (possibly due to a lack of power), but infra-therapeutic (≤500 ng/ml) and non-adherent HCQ levels (≤200 ng/ml) were associated with severe maternal flares during pregnancy. Therefore, this study supports HCQ blood level monitoring in pregnant women with SLE, as a predictor of severe maternal disease activity during pregnancy.
Severe maternal flares and APOs according to different thresholds for HCQ blood levels. A. Therapeutic threshold (therapeutic > 500 ng/mL; subtherapeutic ≤ 500 ng/mL). B. Adherence threshold (adherence > 200 ng/mL; non-adherence ≤ 200 ng/mL).
APOs: adverse pregnancy outcomes; HCQ: hydroxychloroquine
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Gelsomina Alle: None declared, Gaelle Guettrot Imbert: None declared, Estibaliz Lazaro: None declared, Anna Moltó Abbvie, Bristol Myers Squibb, Biogen, Pfizer, UCB, and Lilly, Janssen and UCB, UCB and Pfizer, Maddalena Larosa UCB, Anne Murarasu: None declared, Christophe Richez Astra Zeneca, GSK and Novartis, Lilly and Biogen, Nathalie Morel: None declared, Pauline Orquevaux: None declared, Laurent Sailler: None declared, Viviane Queyrel GSK, Astra Zeneca and Boehringer Ingelheim, Benoit BLANCHET Bristol Myers Squibb, EISAI, Pierre Fabre Oncology, Glaxo Smith Kline, Pfizer and Ipsen., Emmanuelle Pannier: None declared, Luc Mouthon Boehringer Ingelheim and CSL Behring, LFB Biotechnologies, CSL Behring, and Roche, Véronique Le Guern Bristol Myers Squibb, Novartis, Nathalie Costedoat-Chalumeau UCB and Roche.