

Background: Systemic autoimmune diseases are based on an aberrant activation of B cells. Autologous CD19 chimeric antigen receptor (CAR) T cells allow deep depletion of B cells in humans and represent a new possibility to treat autoimmune disease. Previous observations have suggested that a single infusion of CD19-CAR-T cells is not only well tolerated in patients with SLE and other autoimmune diseases but also induces sustained drug-free remission [1-3]. However, safety and efficacy of CD19-CAR-T cell therapy in autoimmune disease has to be demonstrated in controlled clinical studies.
Objectives: To assess the safety and preliminary efficacy of CD19-CAR-T therapy in autoimmune diseases in a controlled clinical study.
Methods: CASTLE (CAR-T cells in systemic B cell-mediated autoimmune disease) is a phase I/II basket study that assesses the safety (primary endpoint) and preliminary efficacy (secondary endpoint) of CD19-CAR-T therapy in systemic lupus erythematosus (SLE), idiopathic inflammatory myositis (IIM) and systemic sclerosis (SSc). It consists of a first part with 8 patients followed by a second part with 16 patients. All patients receive standard cyclophosphamide/fludarabine conditioning therapy followed a single infusion of an advanced therapy medicinal product (MB-CART19.1) containing 1x106 CD19-CAR-T cells/kg body weight that were transfected with a lentiviral vector encoding for a 4-1BB based second generation CAR. To be included, patients had to have a diagnosis of SLE, IIM or SSc, (ii) active disease with organ involvement and (iii) failed treatment with a least two immunosuppressive drugs. Safety was assessed by recording cytokine-release syndrome (CRS), immune effector cell-associated neurotoxicity syndrome (ICANS), myelotoxicity and infections during the first 28 days. Preliminary efficacy was assessed by assessing B cell depletion, CAR-T cell expansion and clinical responses.
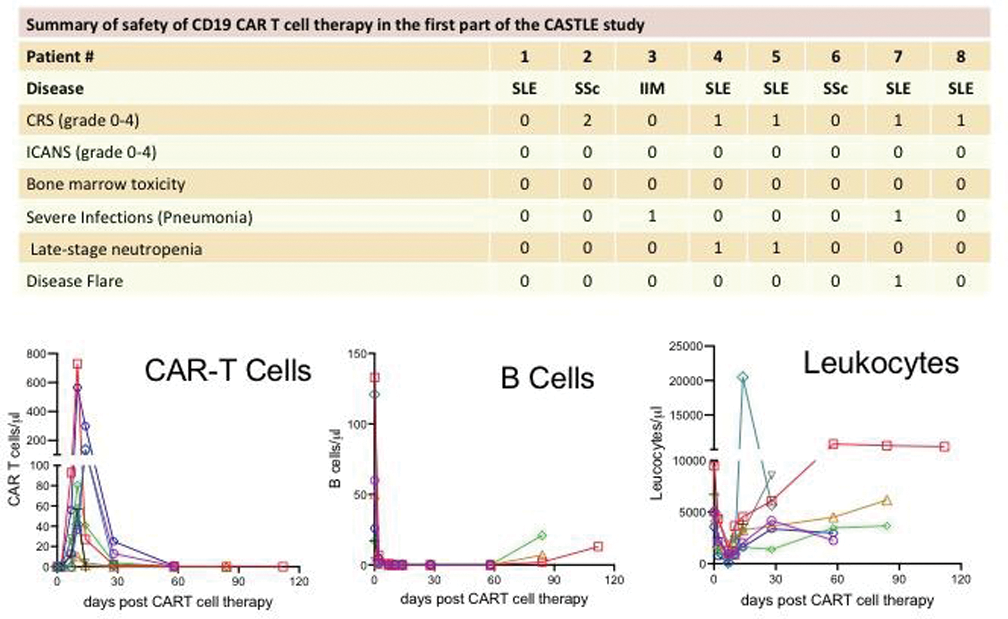
Results: This analysis is on the first part of the CASTLE study comprising 8 patients (5 SLE, 2 SSc, 1 IIM). 6 patients were females, 2 were males. Median age was 33.5 years (range 20-81 years), median disease duration 3 years [range: 1-9 years) and median follow up time 2.4 months (range: 1-5 months). Patients failed on a median number of 4 (range N=3-6) immunosuppressive treatments. From all 8 patients, safety data and B cell/CAR-T cell efficacy data were available, while clinical efficacy data were available from 5/8 patients with sufficiently long follow-up (6 weeks). No higher grade CRS (grade 3 or 4) was observed (grade 0: N=3; grade 1: N=4; grade 2: N=1). No ICANS and no myelotoxicity (grade III/IV neutropenia/leucocytopenia >28 days) were observed. AESI were two late-stage neutropenias that resolved with G-CSF treatment, one flare of SLE before CAR-T cell therapy that required glucocorticoids and two cases of pneumonia (SARS-CoV-2 and CMV) that resolved upon treatment. B cells were completely depleted in all patients within 10 days (Figure 1). CAR-T cells expanded in all patients. Among the 5 patients (3 SLE, 1 SSc, 1 IIM) that had sufficiently long follow up (≥ 6 weeks), three achieved DORIS remission (SLE), one achieved ACR Moderate/Major response (IIM) and one achieved no worsening of lung function (SSc). Furthermore, all patients could successfully stop glucocorticoids and immunosuppressive drugs after CAR-T cell infusion.
Conclusion: These data underline the safety of CD19-CAR-T therapy in autoimmune disease. No higher grade CRS or ICANS or no myelotoxicity is observed. Attention has to be given to late-stage neutropenia, exacerbation of the underlying diseases and infections.
REFERENCES: [1] Mougiakakos D et al., CD19-Targeted CAR T Cells in Refractory Systemic Lupus Erythematosus. N Engl J Med 2021;385:567-569.
[2] Mackensen A. et al., Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus; Nat Med 2022 Oct; 28(10):2124-2132.
[3] Mueller F. et al., A Case Series with Extended Follow-up of CD19 CAR-T cell therapy in Autoimmune Disease; N Engl J Med 2024; in press.
Acknowledgements: NIL.
Disclosure of Interests: None declared.