

Background: An ongoing debate revolves around whether axial spondyloarthritis (axSpA) with psoriasis and psoriatic arthritis (PsA) with axial involvement (axPsA) constitute the same disease or separate entities. The discussion is intensified by the absence of a universally accepted definition for axPsA. With regards to biologic disease-modifying antirheumatic drugs (bDMARDs), both tumor necrosis factor and interleukin-17 inhibitors (TNFi and IL-17i, respectively) have proven effective in both axSpA and PsA. However, interleukin-23 inhibitors (IL-23i) have demonstrated efficacy in PsA but not in axSpA. Post-hoc analyses of randomized controlled trials (RCTs) of IL-23i in PsA suggest, however, that this drug class might also be efficacious in axPsA.
Objectives: To compare the retention of TNFi, IL-17i and IL-23i in patients with axPsA in a large national observational cohort of PsA patients treated under real-life conditions.
Methods: Patients diagnosed with PsA were included in this study if their treating rheumatologist confirmed the presence of axial involvement (based on their interpretation of clinical signs, symptoms, and imaging) in the online database of the national PsA registry at any time before the initiation of bDMARD treatment. Treatment with TNFi, IL-17i, and IL-23i was considered if started between 2015 and 2023, as all three modes of action were approved for the treatment of PsA during this period. Drug retention was investigated using mixed-effects Cox proportional hazards models adjusted for covariates, including age, sex, presence of enthesitis or peripheral arthritis, current smoking, obesity, degree of cutaneous psoriatic involvement (at least moderate-severe vs. ≤ mild-moderate), start of the treatment course (TC) 2019-2023 vs. 2015-2018, and the number of bDMARDs and targeted synthetic (ts-)DMARDs previously utilized. Sensitivity analyses were performed with additional adjustments for the level of C-reactive protein and co-medication with conventional synthetic (cs)DMARDs. The analyses accounted for the possibility that some patients might have been treated with several drugs of the same class, as well as with drugs of different classes.
Results: A total of 952 TCs in 543 patients with axPsA met the inclusion criteria. Among these, 364 TCs had available information on covariates and were included in the adjusted analyses (219 TNFi, 97 IL-17i (84 secukinumab and 13 ixekizumab), and 48 IL-23i (33 ustekinumab and 15 guselkumab)). Patients were older and had a longer symptom duration at start of IL-17i and IL-23i TCs compared to TNFi (Table 1). While the distribution of patients with peripheral arthritis was similar between the groups, the proportion of patients with enthesitis was lower at the start of IL-23i. AxPsA patients starting IL-17i and IL-23i exhibited more severe skin disease, aligning with the established superior efficacy of these drugs for psoriatic skin disease compared to TNFi. Patients initiating IL-17i and IL-23i had experienced a higher number of previous bDMARD failures. The adjusted analysis revealed no evidence for a difference between drug discontinuation between IL-17i vs. TNFi and IL-23i vs. TNFi (HR 0.98, 95% CI 0.68-1.41; and HR 1.01, 95% CI 0.62-1.63, respectively) (Table 2). These results were confirmed after additional adjustment for CRP and co-medication with a csDMARD (HR 1.08, 95% 0.71-1.63 for IL-17i vs. TNFi and HR 0.98, 95% CI 0.56-1.69 for IL-23i vs. TNFi).
Table 1. Patient characteristics at the start of individual treatment courses (N = 364) stratified by drug class: TNFi, IL-17i, and IL-23i, respectively.
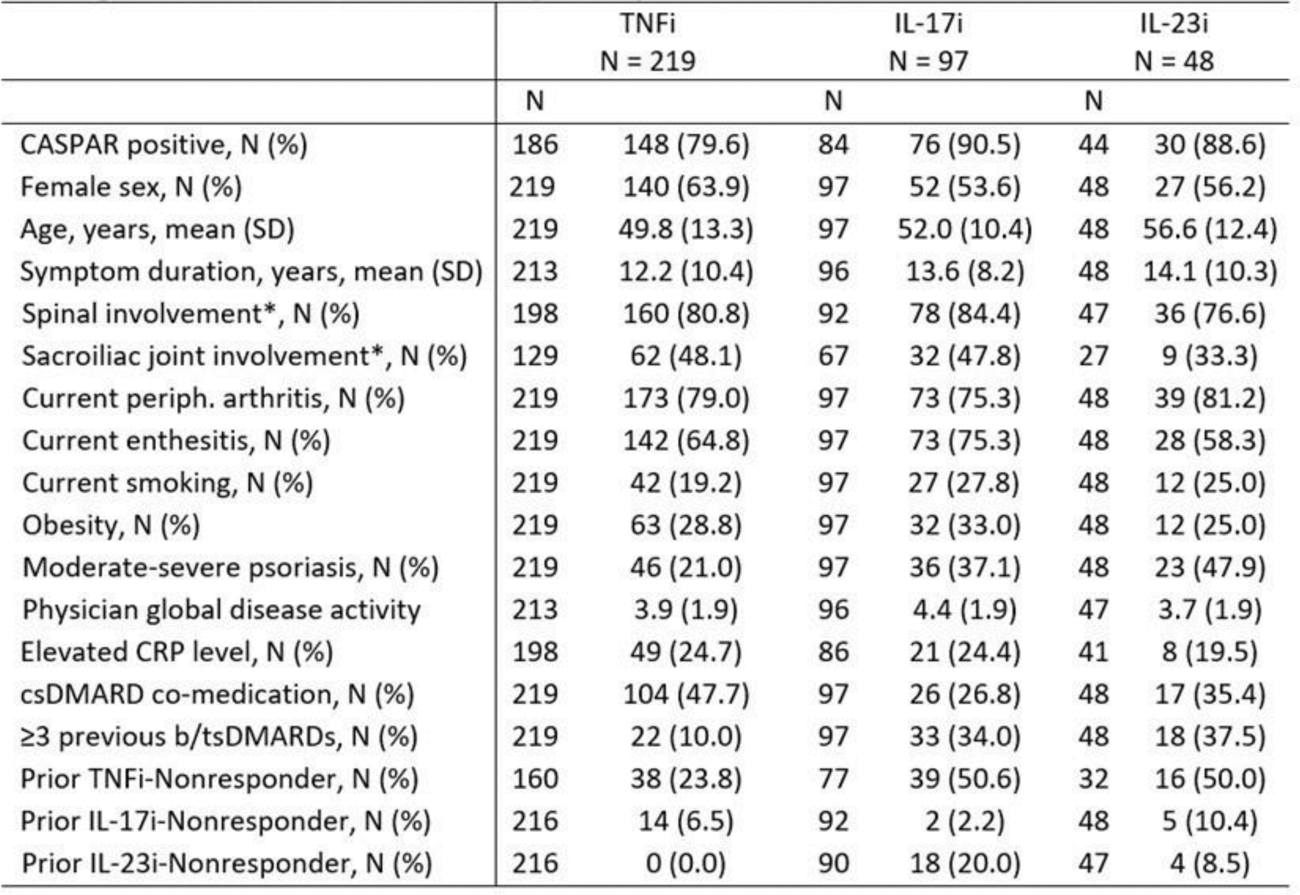
Table 2. Mixed-effects Cox proportional hazards models for analysis of drug discontinuation of a bDMARD in PsA patients with axial involvement. (Analysis performed in 364 treatment courses in 275 patients with 244 discontinuation events).
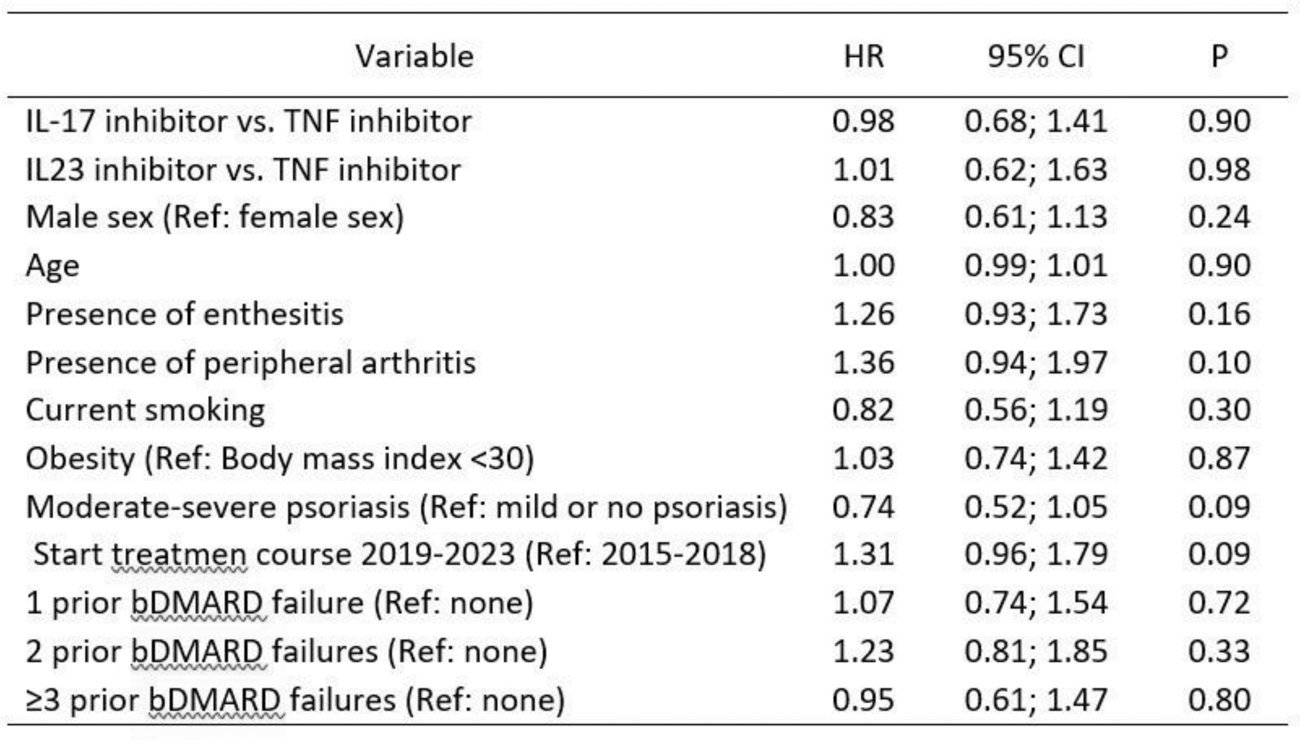
Conclusion: While the profiles of axPsA patients treated with IL-17i and IL-23i differed from those treated with TNFi, our data do not demonstrate a significant difference in drug retention among the three drug classes after adjusting for potential confounding.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Adrian Ciurea: None declared, Andrea Goetschi: None declared, Burkhard Moeller Speaking fees from Janssen, Novartis, Pfizer, Eli Lilly, Grant/Research support from Amgen, Michael J. Nissen Speaking fees from AbbVie, Eli Lilly, Janssen, Novartis, Pfizer, Amgen, Consulting fees from AbbVie, Eli Lilly, Janssen, Novartis, Pfizer, Research grant from Novartis, Pfizer, Kristina Buerki: None declared, René Braem: None declared, Michael Andor: None declared, Thomas Hügle Payments for lectures and presentations from Pfizer, Fresenius Kabi, AbbVie, Merck Sharp and Dohme, Galapagos, Eli Lilly and Novartis, Holds stock or stock options of Atreon SA and Vtuls, royalties from Curmed, Pariticapted on Advisory Boards for DETECTRA, Andrea Rubbert-Roth Honoraria for lectures from AbbVie, Janssen, Novartis and Pfizer, Consulting fees from AbbVie, Janssen and Pfizer, Support for attending meetings from Janssen, Pfizer, Diego Kyburz Honoraria for presentations from AbbVie, Eli Lilly, Payments for participation on advisory boards from AbbVie, Eli Lilly, Janssen, Novartis, Pfizer, Roche, Research grant from AbbVie, support for attending meetings from Janssen and Eli Lilly, Sabine Adler: None declared, Oliver Distler: None declared, Almut Scherer Employed by Bristol-Myers-Squib in 2007-2008, Raphael Micheroli Honoraria for lectures or presentations from AbbVie, Eli Lilly, Janssen, Gilead, Pfizer.