

Background: Patients with lupus nephritis (LN) present with elevated type I interferon gene signatures (IFNGS) in blood, especially during active disease.[1] This supported the investigation of anifrolumab, a type I interferon receptor antagonist, as a potential treatment for LN.[2] There is continued need for noninvasive urinary markers of histological activity, renal inflammation, or disease progression in LN. Cluster of Differentiation 163 (CD163), a scavenger receptor mostly expressed by macrophages, was previously identified as a marker of renal inflammation[3] that, when persistently elevated upon treatment, precedes loss of kidney function.[4]
Objectives: To evaluate the impact of anifrolumab treatment on the urinary biomarker of renal inflammation, CD163, compared with placebo in the phase 2 TULIP-LN trial (NCT02547922).
Methods: In the randomized, placebo-controlled, double-blind TULIP-LN trial, adults with systemic lupus erythematosus (American College of Rheumatology SLE 1997 criteria) and active Class III/IV LN confirmed by biopsy within 3 months of screening were eligible for inclusion. Eligible patients had 24-hour urine protein–creatinine ratio [UPCR] >1 mg/mg and estimated glomerular filtration rate ≥35 mL/min/1.73 m 2 . Patients were randomized 1:1:1 to receive intravenous anifrolumab basic regimen (BR: 300 mg), intensified regimen (IR: 900 mg first 3 doses, 300 mg thereafter), or placebo every 4 weeks for 48 weeks, alongside standard therapy (mycophenolate mofetil and glucocorticoids).[2] Urine samples were collected at baseline, and Weeks 12 and 48, and analyzed for 192 proteins by Luminex. CD163 was selected for this analysis using an hypothesis-driven approach. CD163 levels were normalized to urinary creatinine levels. The impact of anifrolumab IR on CD163 levels from baseline to Week 12 was evaluated versus placebo using a linear mixed effects model, adjusted for baseline IFNGS (high/low) in blood and 24-hour UPCR >3/≤3 mg/mg. Normalized mean CD163 levels were compared by treatment group and Week 52 complete renal response[2] (CRR) status. Repeated measures correlation was used to measure the association of 21-gene type 1 interferon signature[5] in blood and urine levels of CD163 in a within-patient analysis.
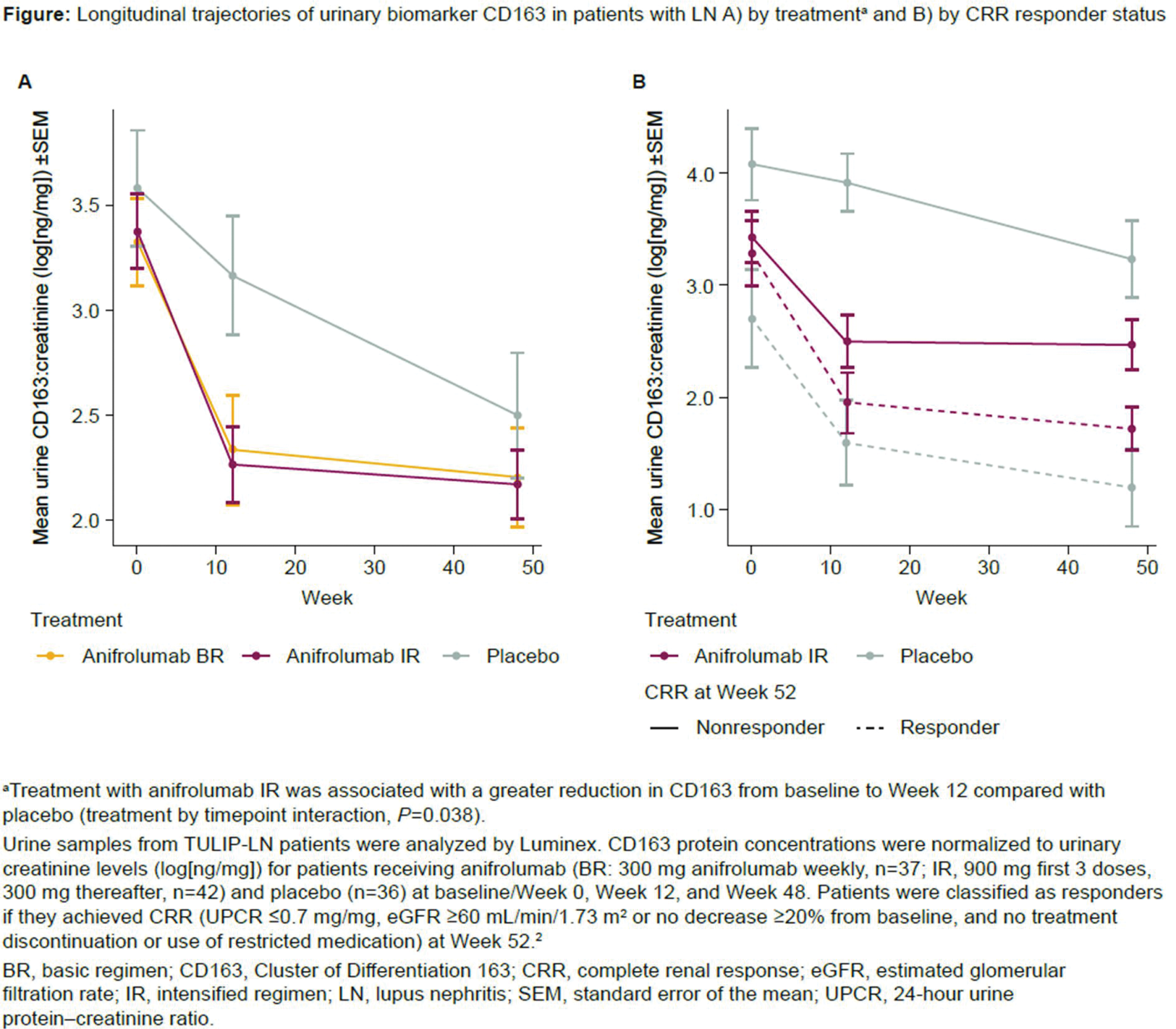
Results: CD163 protein levels declined over time in all treatment groups from baseline (anifrolumab BR, n=37; IR, n=42; placebo, n=36) to Week 48 (Figure 1A). Treatment with anifrolumab IR was associated with a greater reduction in CD163 from baseline to Week 12 compared with placebo (treatment by timepoint interaction, P =0.038). The CD163 levels were comparable among all treatment groups at Week 48. Patients with CRR had greater reductions from baseline in mean CD163 levels than nonresponders at Weeks 12 and 48, regardless of treatment (Figure 1B). Among nonresponders, patients receiving anifrolumab IR showed greater reductions in CD163 from baseline to Week 12 than patients receiving placebo. Within individual patients, urine CD163 levels correlated with blood IFNGS (repeated measures correlation, R=0.691; P <0.001).
Conclusion: Though reductions were similar by Week 48, anifrolumab was associated with greater CD163 reductions in the first 3 months of treatment compared with placebo. CD163 levels were lower in patients achieving CRR than in nonresponders in both treatment groups. These results support that CD163 holds promise as a biomarker of renal response to therapy. Additional studies are needed to further explore the impact of anifrolumab treatment on this urinary biomarker of renal inflammation/histologic activity.
REFERENCES: [1] Feng X. Arthritis Rheumatol . 2006;54:2951–62.
[2] Jayne D. Ann Rheum Dis . 2022;81:496–506.
[3] Endo N. Nephrol Dial Transplant . 2016;31:2023–33.
[4] Fava A. Persistence of urinary biomarkers of intrarenal inflammation precedes loss of kidney function in lupus nephritis [abstract]. Arthritis Rheumatol . 2023;75 (suppl 9).
[5] Yao Y. Arthritis Res Ther . 2010;12(Suppl 1):S6.
Acknowledgements: This study was sponsored by AstraZeneca. Writing assistance was provided by Katey Glunt, PhD of JK Associates Inc., part of Avalere Health, and funded by AstraZeneca.
Disclosure of Interests: Nicola Ferrari Shareholder at: AstraZeneca, Employee of: AstraZeneca, Andrea Fava Consultant of: Annexon Bio, AstraZeneca, Sanofi, UCB, Michelle Petri Paid instructor at: AstraZeneca, Speakers bureau: Aurinia, Consultant of: Alexion, Amgen, AnaptysBio, Annexon Bio, Argenx, Arthros-FocusMedEd, AstraZeneca, Aurinia, Axdev, Biogen, Boxer Capital, Cabaletta Bio, Caribou Biosciences, CTI Clinical Trial and Consulting Services, CVS Health, Eli Lilly, Emergent Biosolutions, Exo Therapeutics, Gilead Biosciences, GSK, Horizon Therapeutics, iCell Gene Therapeutics, IQVIA, Idorsia Pharmaceuticals, Kezar Life Sciences, Kira Pharmaceuticals, MedShr, Merck EMD Serono, Momenta Pharmaceuticals, Nexstone Immunology, Nimbus Lakshmi, PPD Development, Proviant, Regeneron, Sanofi, Seismic Therapeutic, Sinomab Biosciences, Tenet Medicines Inc, UCB, Zydus, Grants/research support from: AstraZeneca, Aurinia, Eli Lilly, Exagen, GSK, Janssen, Thermofisher, Patrick G. Gavin Employee of: AstraZeneca, Eszter Csomor Shareholder at: AstraZeneca, Employee of: AstraZeneca, Philip Z. Brohawn Shareholder at: AstraZeneca, Employee of: AstraZeneca, Daniel Muthas Employee of: AstraZeneca, Adam Platt Shareholder at: AstraZeneca, Employee of: AstraZeneca, Catharina Lindholm Shareholder at: AstraZeneca, Employee of: AstraZeneca.