

Background: Baricitinib, a selective Janus kinase (JAK) 1 and JAK2 inhibitor, inhibits the signaling of multiple JAK-dependent cytokines [1]. A number of studies have reported on the clinical efficacy and safety profile of baricitinib in patients with rheumatoid arthritis (RA). In RA-BEAM, a Phase 3 clinical trial, baricitinib was associated with significant clinical improvements compared to adalimumab, an anti-tumor necrosis factor agent, in patients with RA who showed inadequate response to methotrexate (MTX) [2]. Our cohort study revealed that patients treated with baricitinib had a significantly higher proportion of patients who achieved Boolean remission at 24 weeks compared to those treated with tocilizumab, an anti-interleukin-6 agent [3].
Objectives: This study aimed to compare the effectiveness of baricitinib and abatacept, a T-cell co-stimulation modulator, in patients with RA.
Methods: This study was performed based on data from a multicenter registry of patients with RA, and included 513 and 220 patients treated with abatacept and baricitinib, respectively, who were observed for longer than 52 weeks. Propensity score matching was performed to address potential treatment-selection bias. Baseline data and disease activity were compared between the two treatment groups using the unpaired t test for continuous variables and the chi-square test for categorical variables. The last observation carried forward method was used in each analysis. The cumulative retention rate of each agent was estimated using Kaplan-Meier curves, and the log-rank test was used for comparisons between groups.
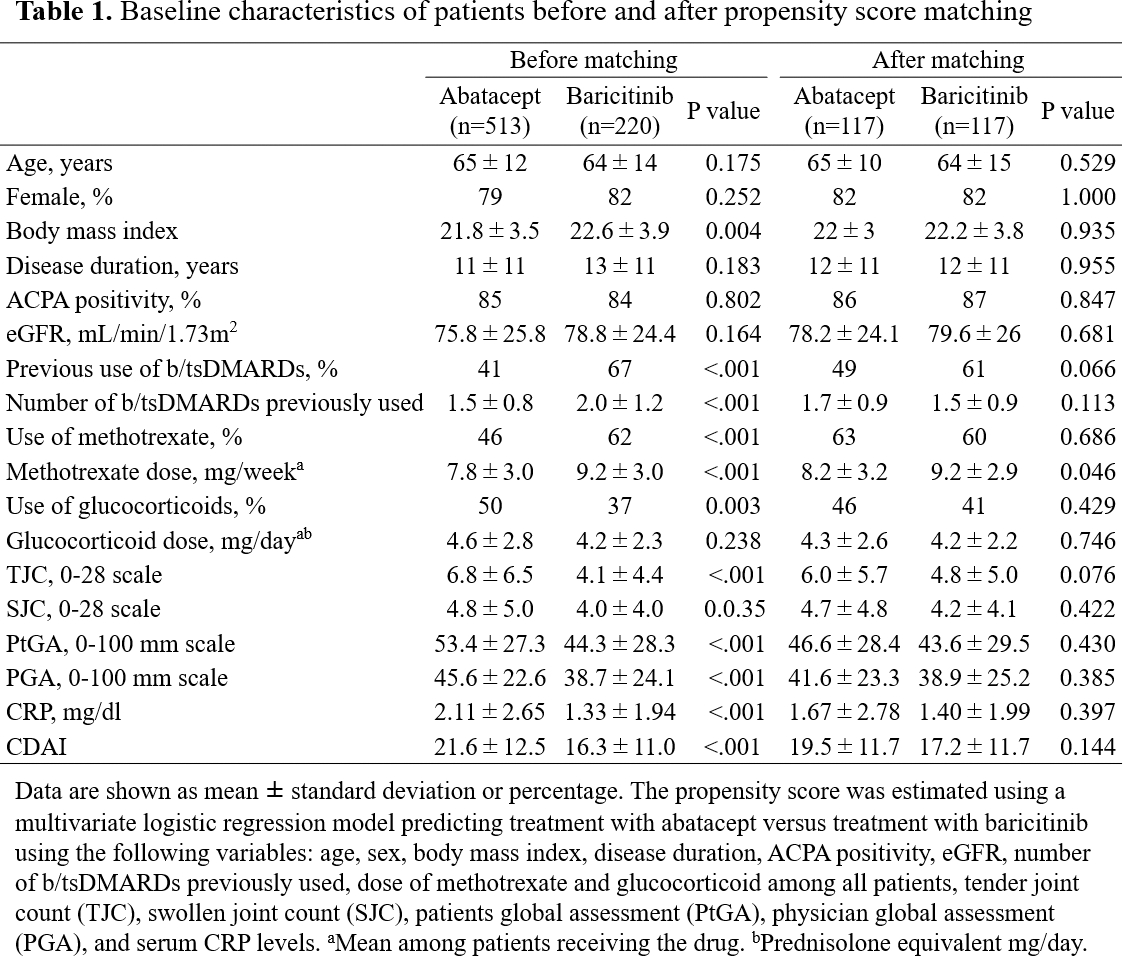
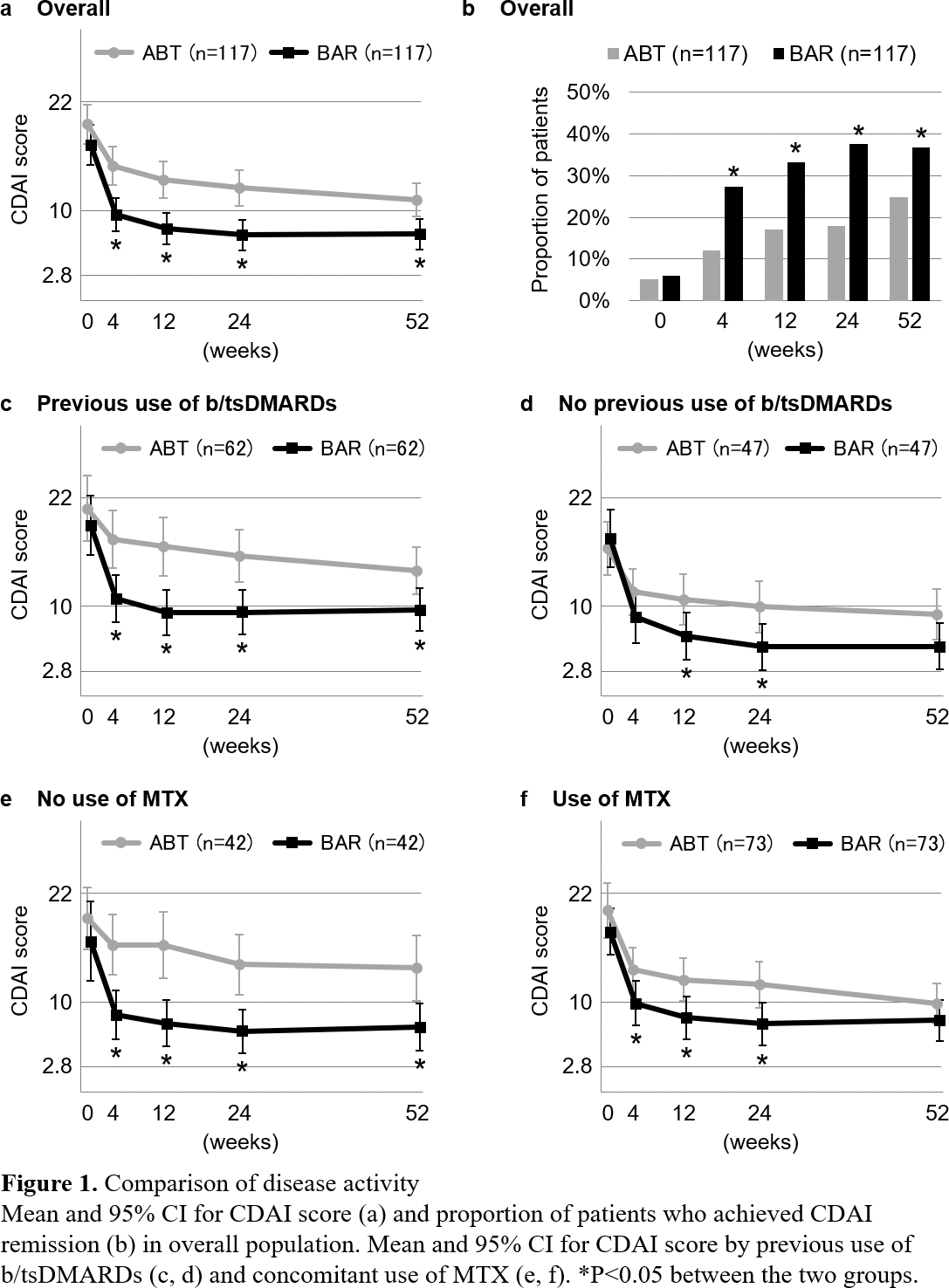
Results: Baseline characteristics of all patients included in this study are shown in Table 1. Before propensity score matching, relative to participants in the abatacept group, those in the baricitinib group were more likely to have a higher body mass index (22.6±3.9 vs. 21.8±3.5, P=0.004), higher rates of previous use of b/tsDMARDs (67% vs. 41%, P<0.001) and methotrexate use (62% vs. 46%, P<0.001), lower rates of glucocorticoid use (37% vs. 50%, P=0.003) and a lower clinical disease index (CDAI) score (16.3±11.0 vs. 21.6±12.5, P<0.001). A total of 117 matched pairs of patients were identified upon propensity score matching. There was good balance across all characteristics between the two groups. According to Kaplan-Meier estimates, there was no significant difference in drug retention rates between the abatacept and baricitinib groups (77% vs. 76% at 52 weeks, P=0.680). There were no significant differences in cumulative discontinuation rates due to inadequate response or adverse events between the two groups. Compared to the abatacept group, the baricitinib group demonstrated significantly lower CDAI scores (Figure 1a) and a higher proportion of patients who achieved CDAI remission (Figure 1b) at 4, 12, 24, and 52 weeks. Subgroup analyses stratified by previous use of b/tsDMARDs and concomitant use of MTX were performed, and propensity score matching was conducted in each subgroup. In the subgroup of patients with previous use of b/tsDMARDs, the baricitinib group had a significantly lower CDAI score than the abatacept group throughout weeks 4-52 (Figure 1c), but partially not in the subgroup of patients without previous use of b/tsDMARDs (Figure 1d). In the subgroup of patients without MTX, the baricitinib group had a significantly lower CDAI score than the abatacept group throughout weeks 4-52 (Figure 1e), but partially not in the subgroup of patients with MTX (Figure 1f).
Conclusion: Baricitinib significantly improved disease activity compared to abatacept within the 52-week observation period, especially in patients with previous use of b/tsDMARDs and those without concomitant use of MTX. Further studies are needed to investigate longer-term efficacy and safety.
REFERENCES: [1] Fridman JS, et al. J Immunol 2010;184:5298-307
[2] Taylor PC, et al. N Engl J Med 2017;376:652-62
[3] Asai S, et al. Clin Rheumatol 2021;40:3143-51
Acknowledgements: NIL.
Disclosure of Interests: Shuji Asai Bristol-Myers Squibb, Eli Lilly, Astellas, Eisai, Nobunori Takahashi Bristol-Myers Squibb, Eli Lilly, Kenya Terabe Bristol-Myers Squibb, Eli Lilly, AbbVie, Yutaka Yoshioka: None declared, Toshihisa Kojima Bristol-Myers Squibb, Eli Lilly, Tomonori Kobayakawa: None declared, Yasumori Sobue: None declared, Tatsuo Watanabe: None declared, Yuji Hirano Bristol-Myers Squibb, Eli Lilly, Yasuhide Kanayama: None declared, Takefumi Kato: None declared, Masahiro Hanabayashi: None declared, Mochihito Suzuki Bristol-Myers Squibb,, Shiro Imagama: None declared