

Background: Systemic lupus erythematosus (SLE) flares are associated with increased risk of organ damage and mortality; flare prevention is a key treatment goal. 1 Benefits of initiating treatment earlier in the disease course have been demonstrated in other autoimmune diseases; however, data are not available for SLE as early disease has not been clearly defined. Pooled analyses of data from registrational belimumab (BEL) clinical trials may provide insights into the impact of earlier use of BEL on SLE flares.
Objectives: To use pooled data from BEL clinical trials to evaluate flares in patients with active SLE treated with BEL or placebo (PBO) plus standard therapy (ST) across subgroups representing early and established disease, respectively, defined based on patients’ baseline SLE disease duration (<2 or ≥2 years), baseline Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index (SDI) score (0 or ≥1) and baseline immunosuppressant (IS) use (no or yes).
Methods: This post hoc analysis integrated data from five international Phase 3 BEL trials in adults with active SLE (BLISS-76, BLISS-52, North East Asia, EMBRACE and BLISS-SC) 2–6 randomised to BEL (10 mg/kg/month intravenously or 200 mg/week subcutaneously) or PBO, plus ST. Time to first severe Safety of Estrogens in Lupus Erythematosus National Assessment–SLE Disease Activity Index (SELENA-SLEDAI) flare index (SFI) flare, any SFI flare and any British Isles Lupus Assessment Group (BILAG) flare (1A/2B) over 52 weeks were evaluated in subgroup analyses (early disease [baseline SLE disease duration <2 years; baseline SDI=0; no baseline IS use] or established disease [baseline SLE disease duration ≥2 years; baseline SDI≥1; baseline IS use]). Hazard ratios (HRs), 95% confidence intervals (CIs) and p-values were from Cox proportional hazards models for comparison between BEL and PBO adjusting for study and baseline SELENA-SLEDAI score (≤9 vs ≥10).
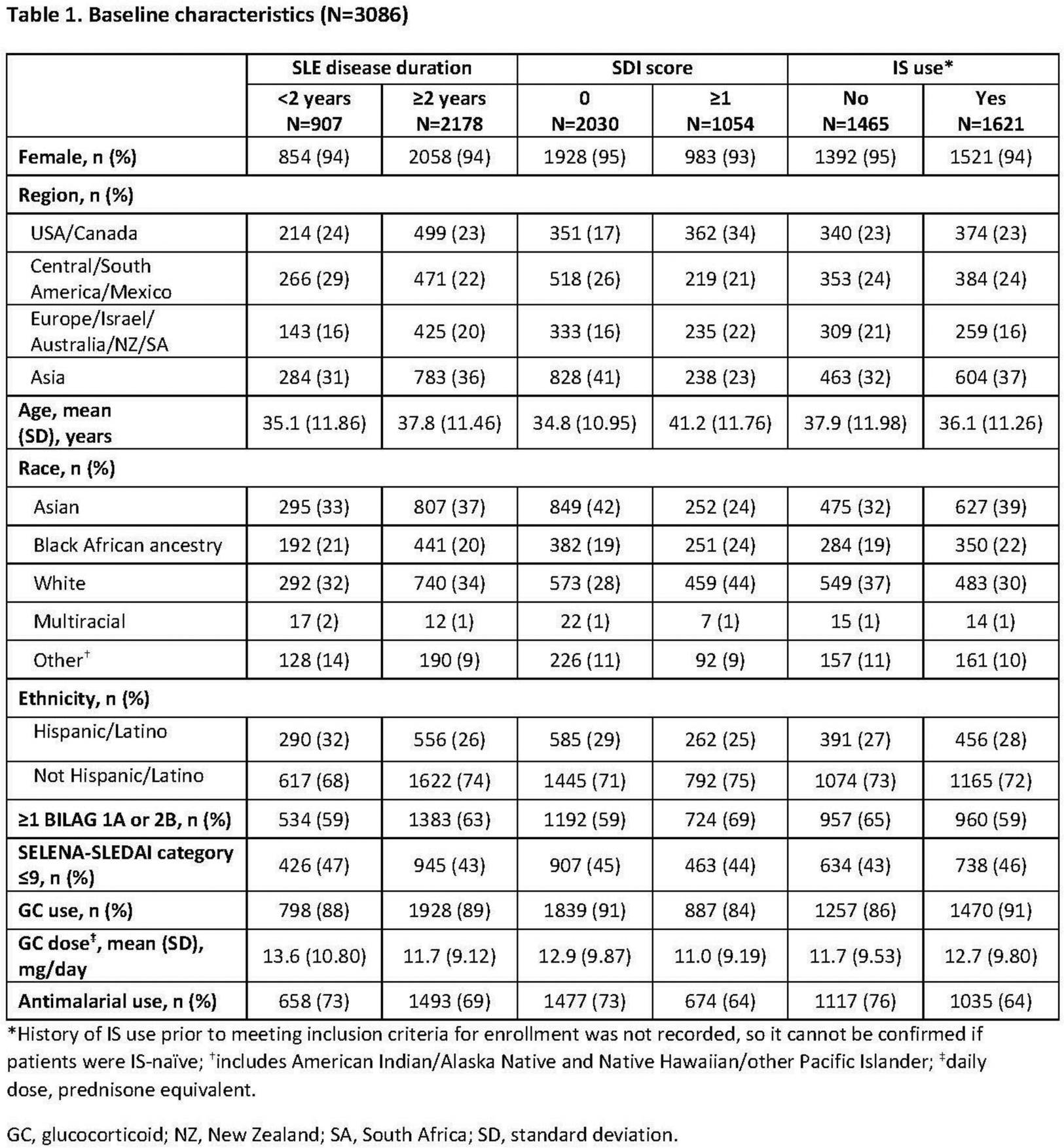
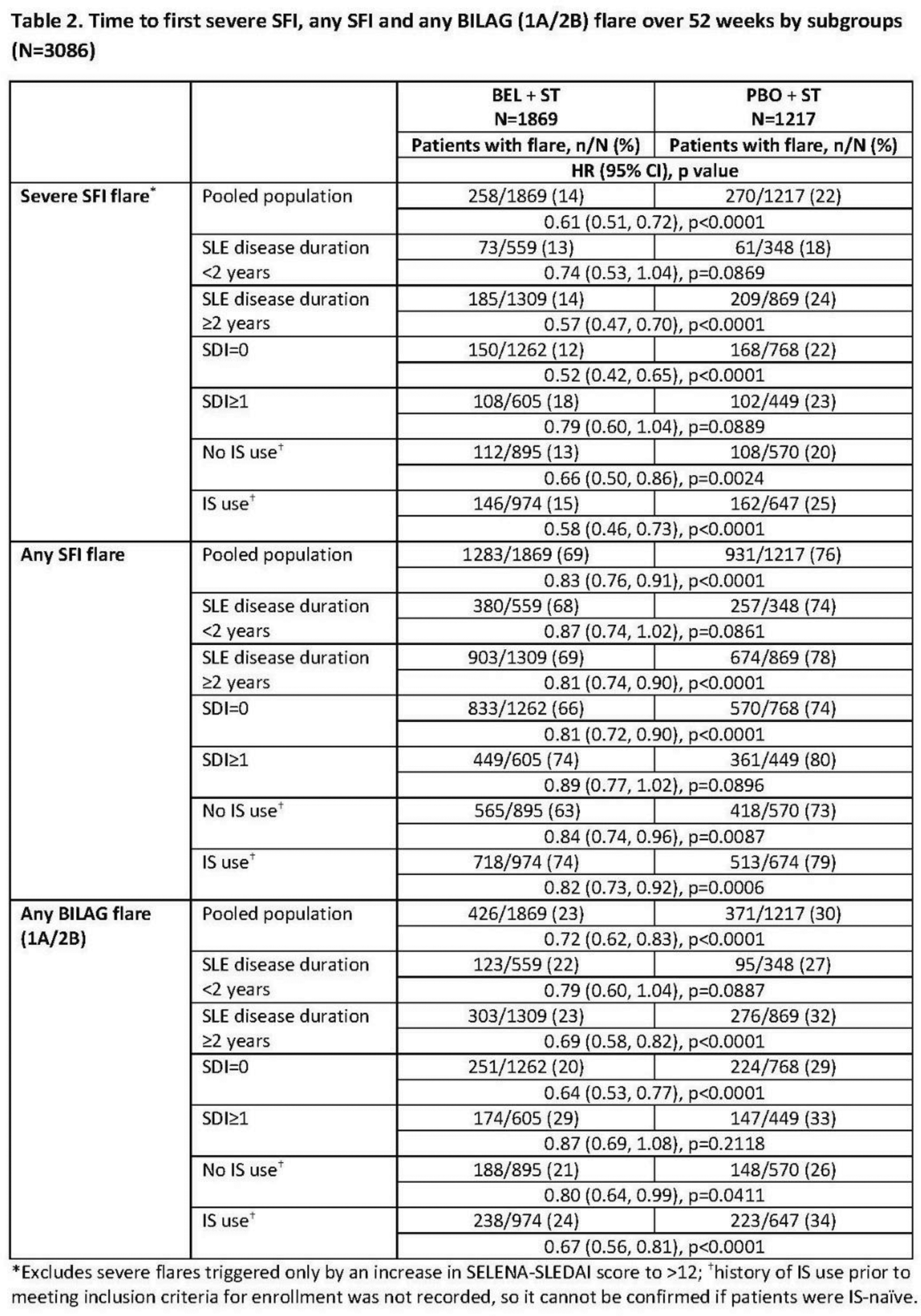
Results: We studied 1869 BEL- and 1217 PBO-treated patients. Differences in baseline characteristics were observed between subgroups for some parameters (Table 1). In the pooled population, significantly fewer patients experienced a severe SFI flare (HR [95% CI]: 0.61 [0.51, 0.72]; p<0.0001), any SFI flare (0.83 [0.76, 0.91]; p<0.0001) or any BILAG flare (0.72 [0.62, 0.83]; p<0.0001) over 52 weeks with BEL versus PBO (Table 2). Over 52 weeks, fewer BEL- versus PBO-treated patients experienced a severe or any SFI flare or any BILAG flare in all early and established disease subgroups, though treatment differences did not reach statistical significance for all comparisons (Table 2). Treatment differences (BEL vs PBO) consistently reached statistical significance for patients with SDI=0 or no IS use at baseline, with greater reduction in flares observed in patients with SDI=0 compared with the pooled population and patients with SDI ≥1. The observed proportion of patients with a severe or any SFI flare or any BILAG flare over 52 weeks was numerically lower in the early versus established disease subgroups and pooled population for BEL and PBO, though differences were small (Table 2).
Conclusion: This post hoc analysis showed fewer patients experienced flares with BEL versus PBO in the pooled population and in early or established disease subgroups, with particular benefit suggested in patients with no baseline organ damage. The proportion of patients with flares was lower in those with early versus established disease, for BEL and PBO. Although the original trials were not designed to evaluate flares in these subgroups, these findings suggest that BEL use in early SLE may improve outcomes. Confirmation in longer prospective studies is needed.
REFERENCES: [1] Fanouriakis A, et al Ann Rheum Dis 2024;83:15
[2] Navarra SV, et al Lancet 2011;377:721
[3] Furie R, et al Arthritis Rheum 2011;63(12):3918
[4] Zhang F, et al Ann Rheum Dis 2018;77:355
[5] Stohl W, et al Arthritis Rheum 2017;69(5):1016
[6] Ginzler E, et al Arthritis Rheum 2022;74(1):112
Acknowledgements: This analysis was funded by GSK. Editorial support (GSK-funded): Evelin O. Szalai, Fishawack Indicia Ltd, UK, part of Avalere Health.
Disclosure of Interests: Marta Mosca GSK, AstraZeneca, Janssen, Otsuka, AbbVie, Lilly, GSK, AstraZeneca, Otsuka, UCB, AbbVie, Idorsia, GSK, Karen Costenbader GSK, BMS, AstraZeneca, Cabaletta Bio, Exagen Diagnostics, Merck, Gilead, Joan Merrill AbbVie, BMS, Sanofi/Provention, RemeGen, Takeda, Xencor, For CME compliant talks: AbbVie, AstraZeneca, Aurinia, BMS, GSK, AbbVie, Amgen, AstraZeneca, Aurinia, BMS, GSK, EMD Serono, Genentech, Gilead, Kezar, Lilly, MSD, Pfizer, Sanofi/Provention, RemeGen, Takeda, UCB, Xencor, Zenas, AstraZeneca, BMS, GSK, Holly A. Quasny GSK, GSK, Christine Henning GSK, GSK, Steven Bloom GSK, Julia H. N. Harris GSK, GSK, Ciara O’Shea GSK, GSK, Tatsuya Atsumi AbbVie Inc., Alexion Inc., Asahi-Kasei Co,. Astellas Pharma Inc., AstraZeneca plc., Bayer Yakuhin, Bristol-Myers Squibb Co., Chugai Pharmaceutical Co., Ltd., Daiichi Sankyo Co., Ltd., Eisai Co. Ltd., Eli Lilly Japan K.K., Gilead Sciences K.K., GlaxoSmithKline K.K, Janssen Pharmaceutical K.K., Novartis Pharma K.K., Nippon Boehringer Ingelheim Co., Ltd., Mitsubishi Tanabe Pharma Co., Pfizer Inc., Taiho Pharmaceutical Co., Ltd. and UCB Japan Co. Ltd., GlaxoSmithKline K.K, AstraZeneca plc., Nippon Boehringer Ingelheim Co., Ltd., Novartis Pharma K.K., Otsuka Pharmaceutical Co., Ltd, GlaxoSmithKline K.K, Ronald F. van Vollenhoven AstraZeneca, Galapagos, MSD, Novartis, Pfizer, Roche, Sanofi, UCB, AbbVie, AstraZeneca, BMS, Galapagos, GSK, Janssen, Pfizer, UCB, AbbVie, AstraZeneca, Biogen, BMS, Galapagos, GSK, Janssen, Pfizer, RemeGen, UCB, BMS, UCB.