

Background: Deucravacitinib, a first-in-class, oral, selective, allosteric tyrosine kinase 2 inhibitor, targets select immune pathways relevant to systemic lupus erythematosus (SLE) and is approved in multiple countries for the treatment of adults with moderate to severe plaque psoriasis. In the 48-week, double-blind, phase 2 PAISLEY trial in patients with active SLE (NCT03252587), a greater proportion of patients randomized to deucravacitinib 3 mg twice daily (BID) met the primary endpoint of SLE Responder Index-4 (SRI[4]) response and all key secondary endpoints, including global disease activity measures.
Objectives: This post hoc subanalysis focuses on the efficacy of deucravacitinib in musculoskeletal manifestations of SLE in the PAISLEY study.
Methods: Patients receiving standard of care were randomized 1:1:1:1 to placebo (n = 90) or deucravacitinib 3 mg BID (n = 91), 6 mg BID (n = 93), or 12 mg once daily (QD; n = 89). A 40-joint count (JC) measured joint swelling, tenderness, or both. Outcomes included achievement of ≥ 50% reduction (JC-50) and 100% reduction (JC-100) in active joint count (swollen + tender; AJC-50 and AJC-100, respectively), swollen joint count (SJC), and tender joint count (TJC) in patients with ≥ 6 joints involved at baseline or patients with any joint involvement at baseline. Responses were reported using a prespecified nonresponder imputation. The 2-sided 95% CI for responses was calculated using the asymptotic (including continuity) method. Results were descriptive.
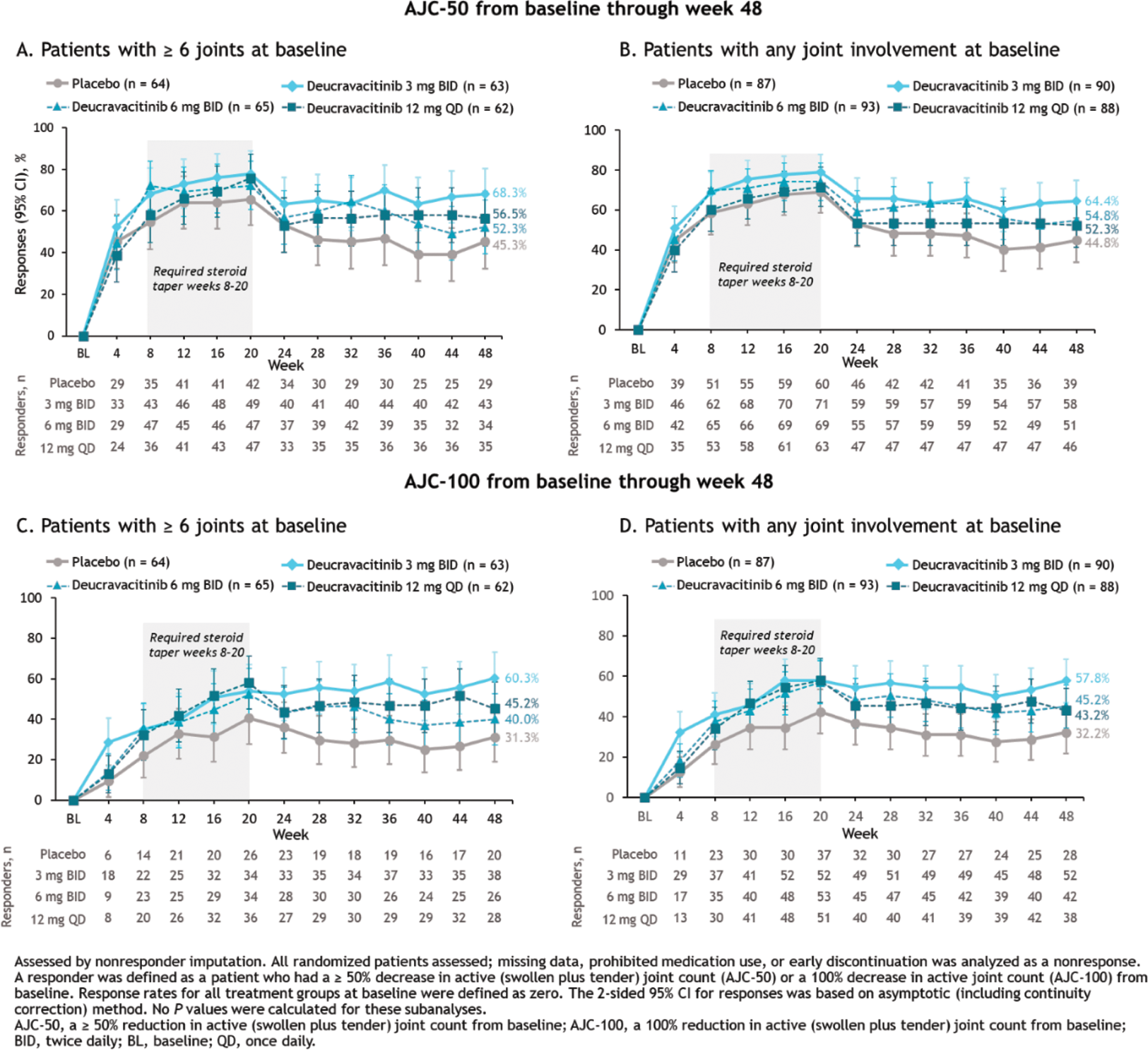
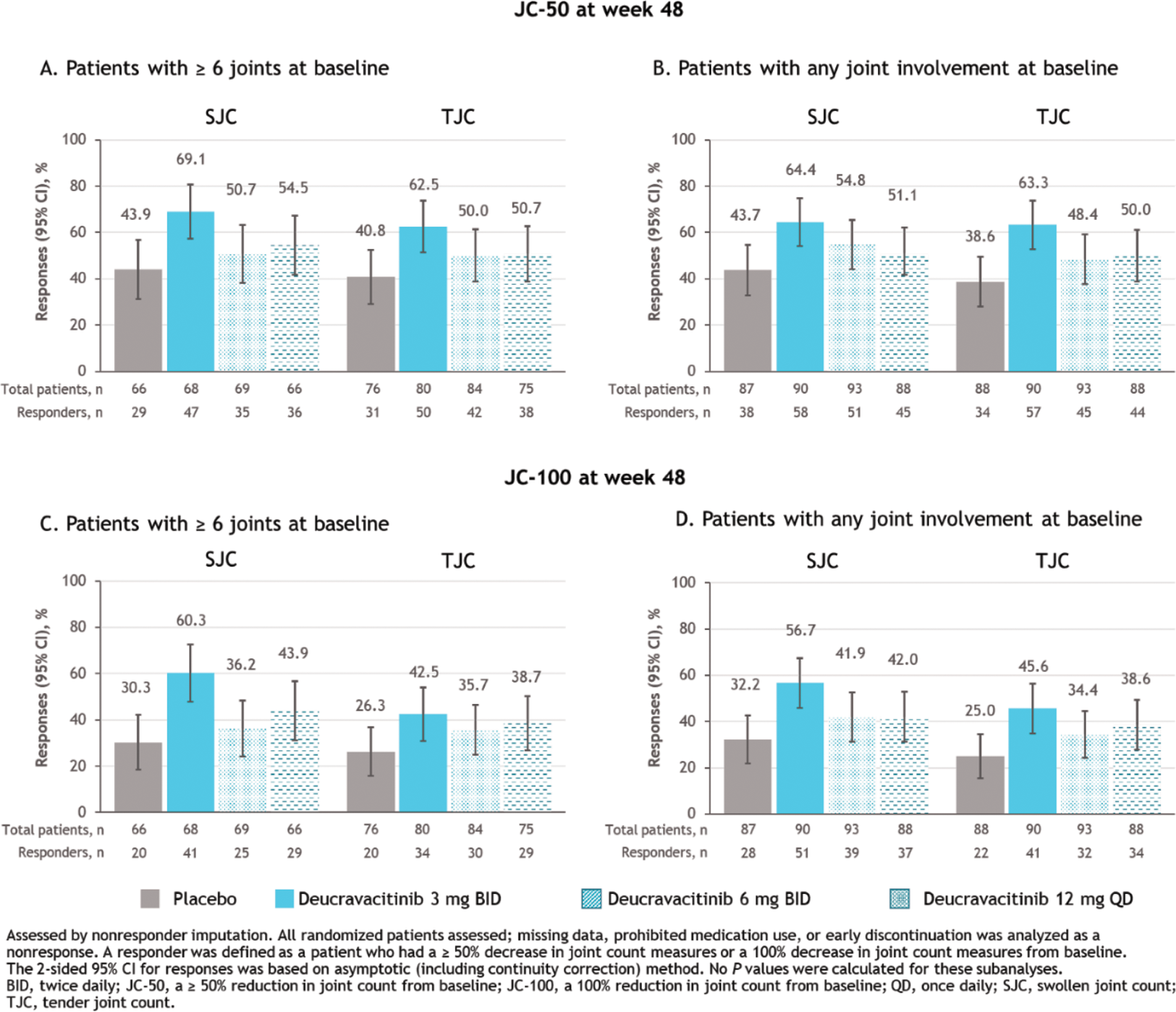
Results: At week 32 (primary endpoint analysis time point), more patients with ≥ 6 active joints at baseline receiving deucravacitinib achieved AJC-50 vs those receiving placebo (placebo, 45.3%; deucravacitinib 3 mg BID, 63.5%; 6 mg BID, 64.6%; 12 mg QD, 56.5%; Figure 1A), in line with previously reported JC-50 findings at week 48. Patients with any joint involvement at baseline receiving deucravacitinib achieved AJC-50 more often than those receiving placebo (week 32: placebo, 48.3%; deucravacitinib 3 mg BID, 63.3%; 6 mg BID, 63.4%; 12 mg QD, 53.4%) through week 48 (Figure 1B). AJC-100 results were similar to AJC-50 results in both populations through week 48 (Figures 1C and 1D). Consistent patterns of higher JC-50 and JC-100 responses with deucravacitinib were observed in separate analyses of SJC and TJC through week 48 (Figure 2).
Conclusion: Deucravacitinib was associated with reductions in joint counts vs placebo using a range of assessment time points, different joint assessments, and different cutoffs for musculoskeletal involvement at baseline.
AJC-50 and AJC-100 responses from baseline through week 48
JC-50 and JC-100 responses at week 48 for SJC and TJC assessments
REFERENCES: NIL.
Acknowledgements: We would like to thank the patients and their families who made this study possible, as well as the clinical teams who participated. This study was sponsored by Bristol Myers Squibb. Professional medical writing assistance was provided by Stephanie V. Koebele, PhD, of Nucleus Global, and funded by Bristol Myers Squibb.
Disclosure of Interests: Marilyn Pike AstraZeneca, Bristol Myers Squibb, Pfizer, UCB, Amit Saxena AstraZeneca, Bristol Myers Squibb, Eli Lilly, GSK, Kezar Life Sciences, Richard A. Furie Bristol Myers Squibb, Bristol Myers Squibb, Peter Nash AbbVie, Amgen, Bristol Myers Squibb, Celgene, Janssen, Eli Lilly, Novartis, Servatus, UCB, Thomas Dörner AbbVie, Aurinia, Bristol Myers Squibb, Eli Lilly, Novartis, Sanofi, UCB, AbbVie, Novartis, Bristol Myers Squibb, Janssen, Roche/Genentech, Sanofi, Eli Lilly, UCB, Deutsche Forschungsgemeinschaft, EU Horizon/HarmonicSS, Coburn Hobar Bristol Myers Squibb, Bristol Myers Squibb, Samantha Pomponi Bristol Myers Squibb, Bristol Myers Squibb, Ravi Koti Employee of Syneos Health, providing statistical services to Bristol Myers Squibb, Subhashis Banerjee Bristol Myers Squibb, Bristol Myers Squibb, Thomas Wegman Bristol Myers Squibb, Bristol Myers Squibb, Joan Merrill AbbVie, Alexion, Amgen, AstraZeneca, Aurinia, Biogen, Bristol Myers Squibb, EMD Serono, Equillium, Genentech, Gilead, GSK, IQVIA, Janssen, Eli Lilly, LFA, Merck, Provention, RemeGen, Sanofi, Takeda, Zenas.