

Background: Deucravacitinib is a first-in-class, oral, selective, allosteric tyrosine kinase 2 (TYK2) inhibitor approved in multiple countries for the treatment of moderate to severe plaque psoriasis. The 48-week, double-blind, phase 2 PAISLEY trial in patients with active SLE (NCT03252587) met its primary endpoint and all key secondary endpoints at the deucravacitinib 3-mg-twice daily (BID) vs placebo.
Objectives: In this post-hoc analysis, response rates over time were evaluated for ≥50% reduction in Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI) score from baseline in patients with a baseline CLASI score ≥10 (CLASI-50), ≥50% reduction in active (swollen + tender) joint count in patients with ≥6 active joints at baseline (AJC-50), and other key secondary endpoints.
Methods: Patients with active SLE receiving standard of care were randomized 1:1:1:1 to placebo (n = 90), deucravacitinib 3 mg twice daily (BID; n = 91), 6 mg BID (n = 93), or 12 mg once-daily (QD; n = 89). Previous reporting from PAISLEY used strict nonresponder imputation criteria with several conditions considered a nonresponse applied after week 20, including not achieving a glucocorticoid dose ≤7.5 mg/day by week 20, resulting in different imputation methods used before vs after week 20. Here, we characterize disease activity improvement in 2 key lupus manifestations, imputing only missing data as nonresponse (M=NR) through week 48 and assessing response rate kinetics using the same imputation method throughout.
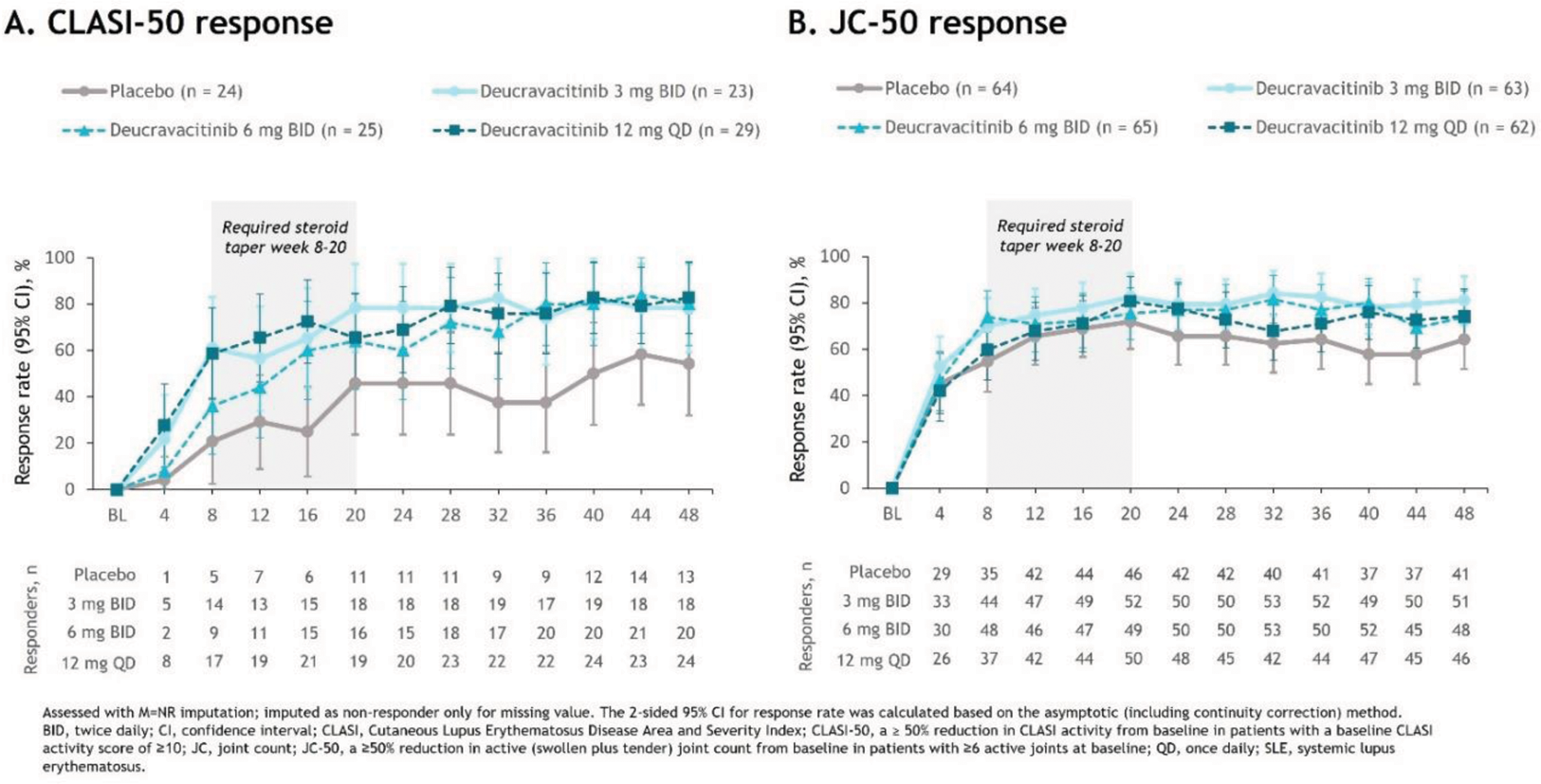
Results: Patients receiving deucravacitinib had numerically higher CLASI-50 response rates vs placebo starting at week 4 (placebo, 4.2%; 3 mg BID, 21.7%; 6 mg BID, 8.0%; 12 mg QD, 27.6%) (Figure 1A); robust differences were maintained through week 48. Numerical differences in AJC-50 response rates were first observed at week 8 (placebo, 54.7%; 3 mg BID, 69.8%; 6 mg BID, 73.8%; 12 mg QD, 59.7%) and became more notable from week 24 (Figure 1B). Differences in responses of both organs were maintained with deucravacitinib beyond week 20, after the protocol-mandated glucocorticoid taper.
Conclusion: These data suggest that higher response rates with deucravacitinib vs placebo occur early on for mucocutaneous and somewhat later for musculoskeletal manifestations. Permitted concomitant glucocorticoid use may partially explain the placebo group responses. Robust differences were seen after the glucocorticoid taper, further supporting the efficacy of deucravacitinib.
Response rates from weeks 4 to 48 in patients with active SLE
REFERENCES: NIL.
Acknowledgements: We would like to thank the patients and their families who made this study possible, as well as the clinical teams who participated. This study was sponsored by Bristol Myers Squibb. Professional medical writing assistance was provided by Angela R. Eder, PhD, of Nucleus Global, and funded by Bristol Myers Squibb.
Disclosure of Interests: Ronald F. van Vollenhoven Pfizer, Roche, UCB, Pfizer, AbbVie, Galapagos, and Janssen, UCB, Pfizer, AbbVie, Galapagos, and Janssen, AstraZeneca, Biogen, Biotest, Celgene, Gilead, and Servier, Bristol Myers Squibb, GSK, and Eli Lilly, UCB, Joseph F. Merola AbbVie, Amgen, Biogen, Bristol Myers Squibb, Dermavant, Eli Lilly, Janssen, LEO Pharma, Novartis, Pfizer, Regeneron, Sanofi, Sun Pharma, and UCB, Kathryn H. Dao Bristol Myers Squibb, Piotr Leszczyński Novartis, AbbVie, AstraZeneca, Bristol Myers Squibb, Pfizer, and UCB, Bristol Myers Squibb, Marilyn Pike AstraZeneca, Bristol Myers Squibb, Pfizer, and UCB, Samantha Pomponi Bristol Myers Squibb, Bristol Myers Squibb, Coburn Hobar Bristol Myers Squibb, Bristol Myers Squibb, Matthew J. Colombo Bristol Myers Squibb, Bristol Myers Squibb, Ravi Koti Employee of Syneos Health and provides statistical services to Bristol Myers Squibb, Subhashis Banerjee Bristol Myers Squibb, Bristol Myers Squibb, Thomas Wegman Bristol Myers Squibb, Bristol Myers Squibb, Eric Morand AstraZeneca, Biogen, Bristol Myers Squibb, EMD Serono, Genentech, Gilead, Novartis, and Takeda, AbbVie, Amgen, AstraZeneca, Biogen, Bristol Myers Squibb, Eli Lilly, EMD Serono, Genentech, GSK, Janssen, Takeda, and UCB.