

Background: Systemic lupus erythematosus (SLE) is an autoimmune disease that affects women of childbearing age, multiplying pregnancy complications 2-4 times. According to EULAR recommendations, conception should occur with SLE in remission for at least 6 months, blood pressure under control and using safe drugs. But keeping the disease under control with safe drugs for pregnancy is no easy task. Belimumab (BEL) is an IgG1 monoclonal antibody indicated for the treatment of moderate-to-severe SLE. Although no risk of pregnancy loss or increased congenital malformations was observed in animals, it is classified as a class C drug for pregnancy. Both EULAR and ACR consider the information regarding foetal safety to be insufficient, advising to substitute BEL for another drug prior to conception, and only consider its use in the absence of another pregnancy-compatible drug capable of controlling SLE.
Objectives: To share our experience with belimumab in lupus pregnant women and to review the relevant published literature on its use in this scenario
Methods: A prospective observational study in clinical practice of pregnant patients with SLE seen in the multidisciplinary unit (consisting of an obstetrician and a rheumatologist) from January-2018 to August-2023, was conducted. Patients meeting EULAR/ACR 2019 SLE classification criteria were included, excluding those who refused to participate or who had not completed their pregnancy by August-2023. Demographic, clinical, drug, pregnancy evolution, and maternal and fetal outcomes were collected. The study was approved by the ISABIAL Ethics Committee (internal code: 2018-003). Patients gave informed consent and the study complied with the principles of the Declaration of Helsinki.
Additionally, MEDLINE and EMBASE databases were searched, and a secondary hand search of the literature was performed. Studies were evaluated and visualized descriptively. Descriptive data were summarized by median and interquartile range (IQR).
Results: During the period January 2018 to July 2023, 17 pregnancies of 13 women diagnosed with SLE were attended. All met the EULAR/ACR 2019 SLE classification criteria. All agreed to take part in the study. One woman had not completed her pregnancy by the end of the study and was therefore excluded. 16 pregnancies of 12 lupus women were included, 6 (involving 8 pregnancies) received BEL throughout their illness, five of them during some period of gestation. In this group, there was one miscarriage, one elective termination and seven live fetuses (including two live twins). There was one type I intrauterine growth retardation (IGR), and a preterm pregnancy due to premature rupture of membranes (PROM). One mild lupus flare was detected. There were no cases of pre-eclampsia, gestational diabetes mellitus or hypertension. All neonates had normal Apgar scores at birth, none needed critical care. There were no congenital anomalies. In the non-BEL group, there were six live fetuses from six mothers. Two patients had two pregnancies during this period. There were two miscarriages. The remaining six pregnancies went to term, three of them by caesarean section. One patient suffered a postpartum hemorrhage that needed reoperation. There was a gestational diabetes mellitus. There were no cases of pre-eclampsia, hypertension, or any flare during pregnancy. All but one neonate had normal Apgar scores at birth, and none required critical care. There were no IGR, preterm pregnancies, PROM, low birth weight babies or congenital anomalies.
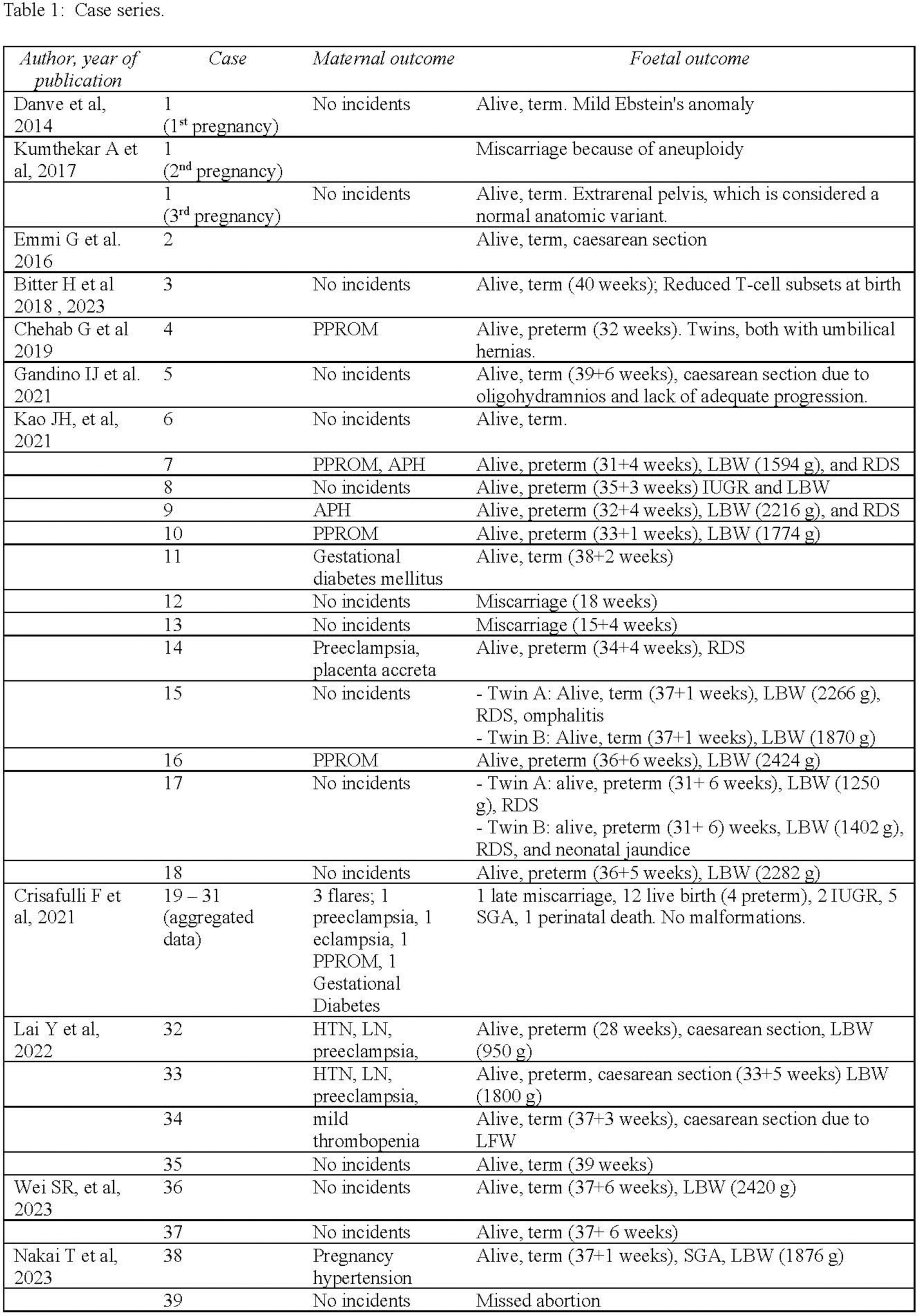
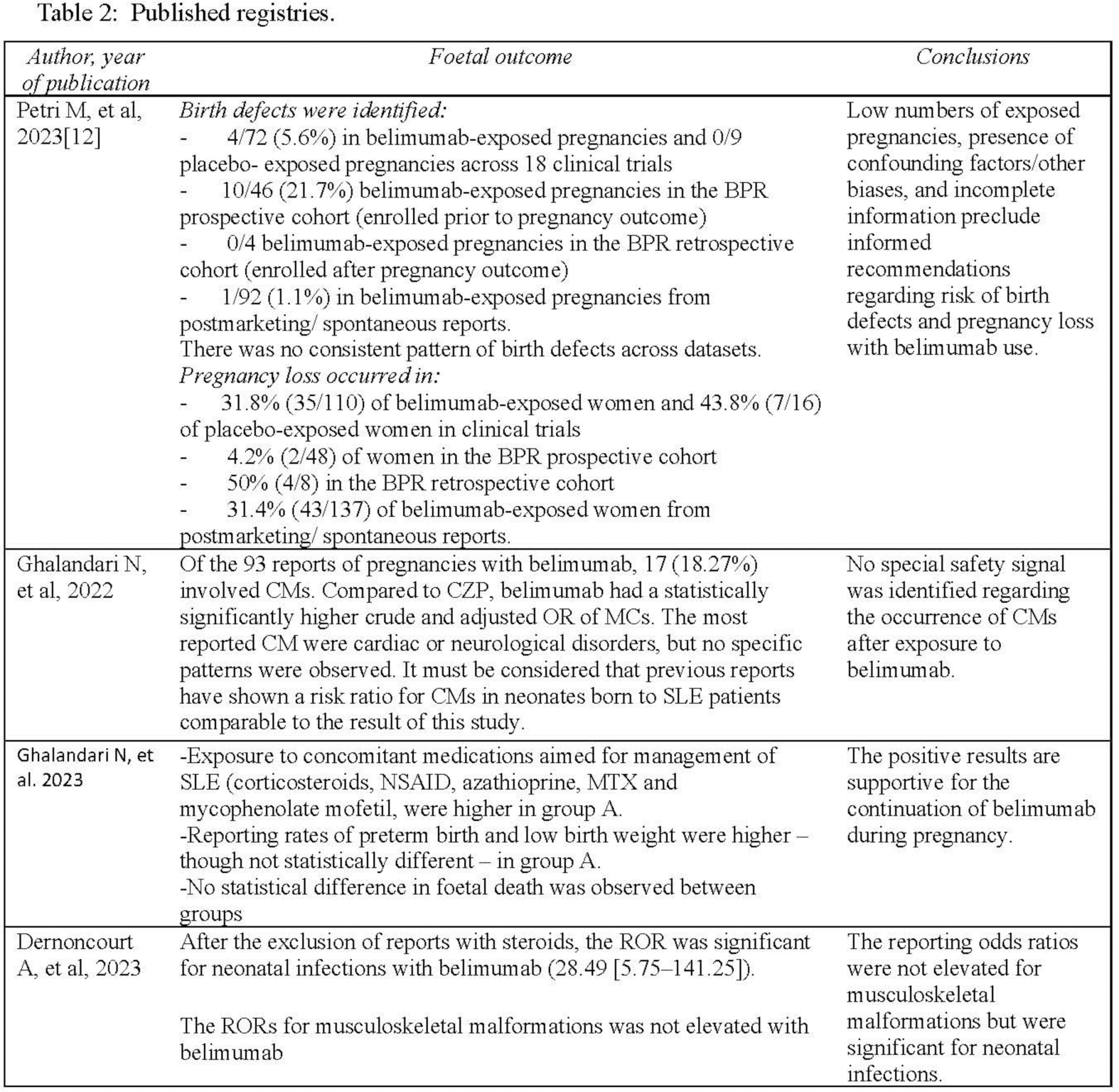
After the search, we identified 10 case reports and case series, and five registries. Among the 39 reported cases (41 pregnancies), there were 5 PROM, 4 pre-eclampsia, and 1 eclampsia. All women made full recoveries. 19 new-borns had low birth weight. There were no malformations. (Table 1). While registries did not indicate an increased risk of birth defects or pregnancy loss, there was a higher risk of neonatal infections (Table 2).
Conclusion: Belimumab may be an option for pregnant women with difficult-to-control lupus. Further research is needed to confirm the absence of association between belimumab and foetal harm.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Paloma Vela Casasempere Abbvie, GSK, Galapagos, Lilly, Amgen, GSK, AstraZeneca, Pfizer, GSK, Abbvie, Roche, Novartis, Lilly, AstraZeneca, Pfizer, Rocío Caño: None declared, Silvia Gómez: None declared, Silvia Cortell Aznar: None declared, Encarnacion Perez Pascual: None declared.