

Background: The British Isles Lupus Assessment Group (BILAG)-based Composite Lupus Assessment (BICLA) and the Systemic Lupus Erythematosus (SLE) Responder Index (SRI-4) are measures of response used as primary endpoints in randomized controlled trials (RCTs) within SLE. BICLA enables incremental response assessment, while SRI-4 reflects response based on the presence or absence of a lupus manifestation rather than its severity [1, 2].
Objectives: The purpose of this study was to determine belimumab efficacy assessed using BICLA in patients with SLE included in the phase III belimumab RCTs, in which SRI-4 outcomes were originally assessed.
Methods: A post-hoc meta-analysis was carried out using data from five RCTs of belimumab in active adult SLE: four with intravenous (IV) belimumab (BLISS-52 [3], BLISS-76 [4], BLISS-Northeast Asia (NEA) [5], and EMBRACE [6]), and one with subcutaneous belimumab (BLISS-SC [7]). The 52-week landmark assessments were analysed across all trials for the approved belimumab dose (10 mg/kg/month IV or 200 mg/week SC) or placebo, in addition to background therapy. Treatment response was defined in accordance with the BICLA definition, which requires BILAG improvement, no worsening of disease activity based on BILAG and SLE Disease Activity Index (SLEDAI-2K), no deterioration in Physician Global Assessment (PGA) ≥0.3 (0–3), and no treatment failure.
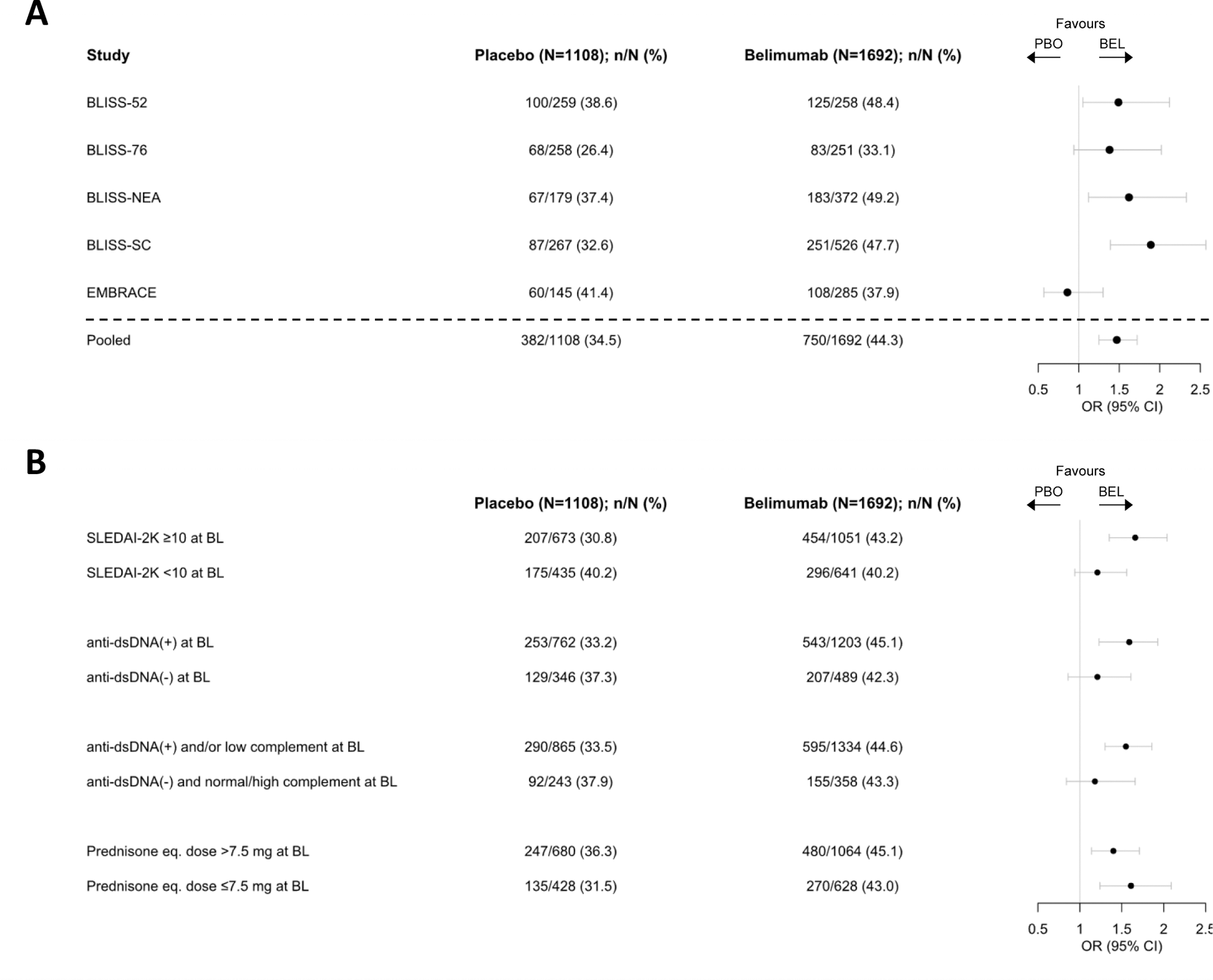
Results: A total of 3086 patients received belimumab (IV or SC, N=1869) or placebo (N=1217). Patient characteristics did not differ across the belimumab vs placebo-treated groups, including sex, age, ancestry, disease duration, SLEDAI-2K, PGA, anti-dsDNA antibody positivity, low C3 and/or C4, concomitant SLE medications, or glucocorticoid dose (3-7). Among 2800 patients with calculable BICLA response data at week 52, the following were analysed on an individual trial level (belimumab vs placebo, respectively): BLISS-52, N=258 vs 259; BLISS-76, N=251 vs 258; BLISS-NEA, N=372 vs 179; BLISS-SC, N=526 vs 267; EMBRACE, N=285 vs 145. Significant differences in BICLA response frequencies were observed in favour of belimumab vs placebo in BLISS-52 (OR: 1.49; 95% CI: 1.05–2.12; p=0.024), BLISS-NEA (OR: 1.62; 95% CI: 1.12–2.33; p=0.010), and BLISS-SC (OR: 1.89; 95% CI: 1.39–2.57; p<0.001). A statistically non-significant but marginal difference was observed in BLISS-76 (OR: 1.38; 95% CI: 0.94–2.02; p=0.098), and no difference was observed in EMBRACE (OR: 0.86; 95% CI: 0.57–1.30; p=0.484) (Figure 1). A highly significant difference in BICLA response in favour of belimumab vs placebo was observed in analysis of pooled data from all trials, after adjusting for trial variance (OR: 1.47; 95% CI: 1.25–1.75; p<0.001, Figure 1). In subgroup analyses, BICLA responses to belimumab were superior to placebo in patients with SLEDAI-2K≥10 and positive anti-dsDNA and/or low complement levels at baseline, but no difference was observed in BICLA response frequencies between belimumab and placebo recipients in relation to baseline glucocorticoid dose (Figure 1).
Conclusion: In this meta-analysis of five RCTs, belimumab conferred significantly greater benefit than placebo based on the BICLA criteria, especially in patients with highly active SLE and/or active serology at baseline, further substantiating the efficacy of belimumab in SLE through multiple efficacy endpoints.
REFERENCES: [1] Wallace DJ, Kalunian K, Petri MA, et al. Ann Rheum Dis. 2014;73(1):183-90.
[2] Furie RA, Petri MA, Wallace DJ, et al. Arthritis Rheum. 2009;61(9):1143-51.
[3] Navarra SV, Guzman RM, Gallacher AE, et al. Lancet. 2011;377(9767):721-31.
[4] Furie R, Petri M, Zamani O, et al. Arthritis Rheum. 2011;63(12):3918-30.
[5] Zhang F, Bae SC, Bass D, et al. Ann Rheum Dis. 2018;77(3):355-63.
[6] Ginzler E, Guedes Barbosa LS, et al. Arthritis Rheumatol. 2022;74(1):112-23.
[7] Stohl W, Schwarting A, Okada M, et al. Arthritis Rheumatol. 2017;69(5):1016-27.
Meta-analysis of the five phase III randomized controlled trials assessing belimumab: BICLA response assessment at 52 weeks of follow-up.
Acknowledgements: NIL.
Disclosure of Interests: Ioannis Parodis I have received research funding and/or honoraria from Amgen, AstraZeneca, Aurinia, Bristol Myers Squibb (BMS), Elli Lilly, Gilead, GlaxoSmithKline (GSK), Janssen, Novartis, Otsuka, and Roche., Julius Lindblom: None declared, Nursen Çetrez: None declared, Leonardo Palazzo: None declared, Shereen Oon: None declared, Henri Ala: None declared, Ronald F. van Vollenhoven I have received support for educational programs from AstraZeneca, Galapagos, MSD, Novartis, Pfizer, Roche, Sanofi, and UCB., I have received speaking fees and/or honoraria from AbbVie, AstraZeneca, BMS, Galapagos, GSK, Janssen, Pfizer, and UCB., I have received consulting fees from AbbVie, AstraZeneca, Biogen, BMS, Galapagos, Janssen, Pfizer, RemeGen, and UCB., I have received grant/research support from BMS and UCB., Eric Morand I have received consulting fees and/or honoraria from AstraZeneca, Biogen, BMS, EMD Serono, Galapagos, GSK, IgM, Janssen, Novartis, Roche, and Takeda., I have received grant support from AbbVie, Amgen, AstraZeneca, Biogen, BMS, Janssen, Eli Lilly, EMD Serono, Genentech, GSK, Takeda, and UCB., Adrian Levitsky I report employment with BMS outside the submitted work., Mandana Nikpour I have received research grant support from Janssen and Boehringer Ingelheim. I have received honoraria from Janssen, GSK, AstraZeneca, Pfizer, and Boehringer Ingelheim.