

Background: Janus kinase (JAK) inhibitors block the signaling of multiple JAK-dependent cytokines. A number of studies have reported on the clinical efficacy of JAK inhibitors in patients with rheumatoid arthritis (RA). Post-hoc analyses of clinical trials suggest that JAK inhibitors may be more effective in sero-positive patients than in sero-negative patients [1, 2]. The efficacy of JAK inhibitors has been demonstrated both as monotherapy and in combination with methotrexate (MTX). However, few studies have directly compared JAK inhibitor monotherapy with MTX combination therapy.
Objectives: This study aimed to determine predictors of clinical remission in patients with rheumatoid arthritis treated with JAK inhibitors in clinical setting, and to investigate the influence of concomitant MTX on disease activity.
Methods: This study was performed based on data from a multicenter registry of patients with RA, and included 396 patients treated with JAK inhibitors (220 baricitinib, 66 peficitinib, 77 upadacitinib, and 92 filgotinib) who were observed for longer than 24 weeks. The impact of patient characteristics on remission [Simple Disease Activity Index (SDAI) ≤3.3] was assessed with logistic regression models. To assess the impact of concomitant use of MTX, patients were divided into two groups by MTX use: MTX (+) (n=216) and MTX (-) (n=180) groups. Propensity score matching was performed to address potential treatment-selection bias. Baseline data and disease activity were compared between the two groups using the unpaired t test for continuous variables and the chi-square test for categorical variables. The last observation carried forward method was used in each analysis.
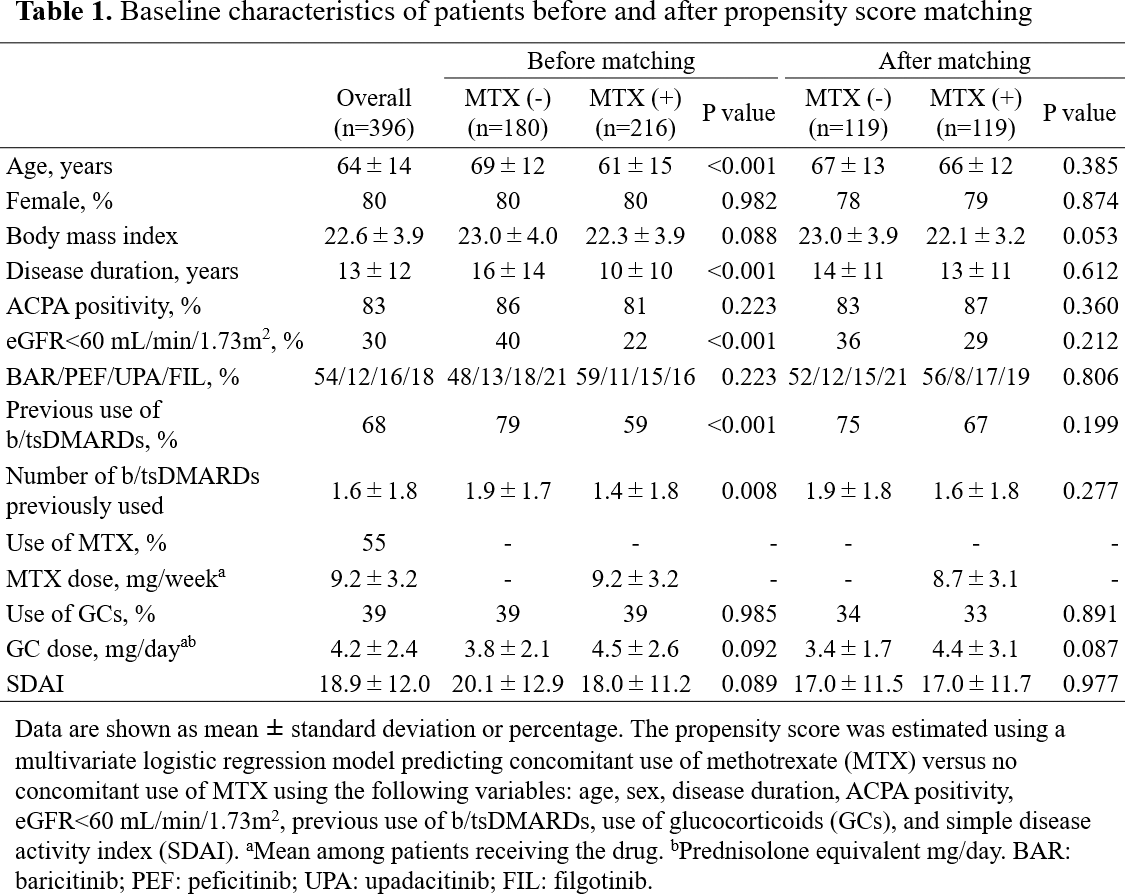
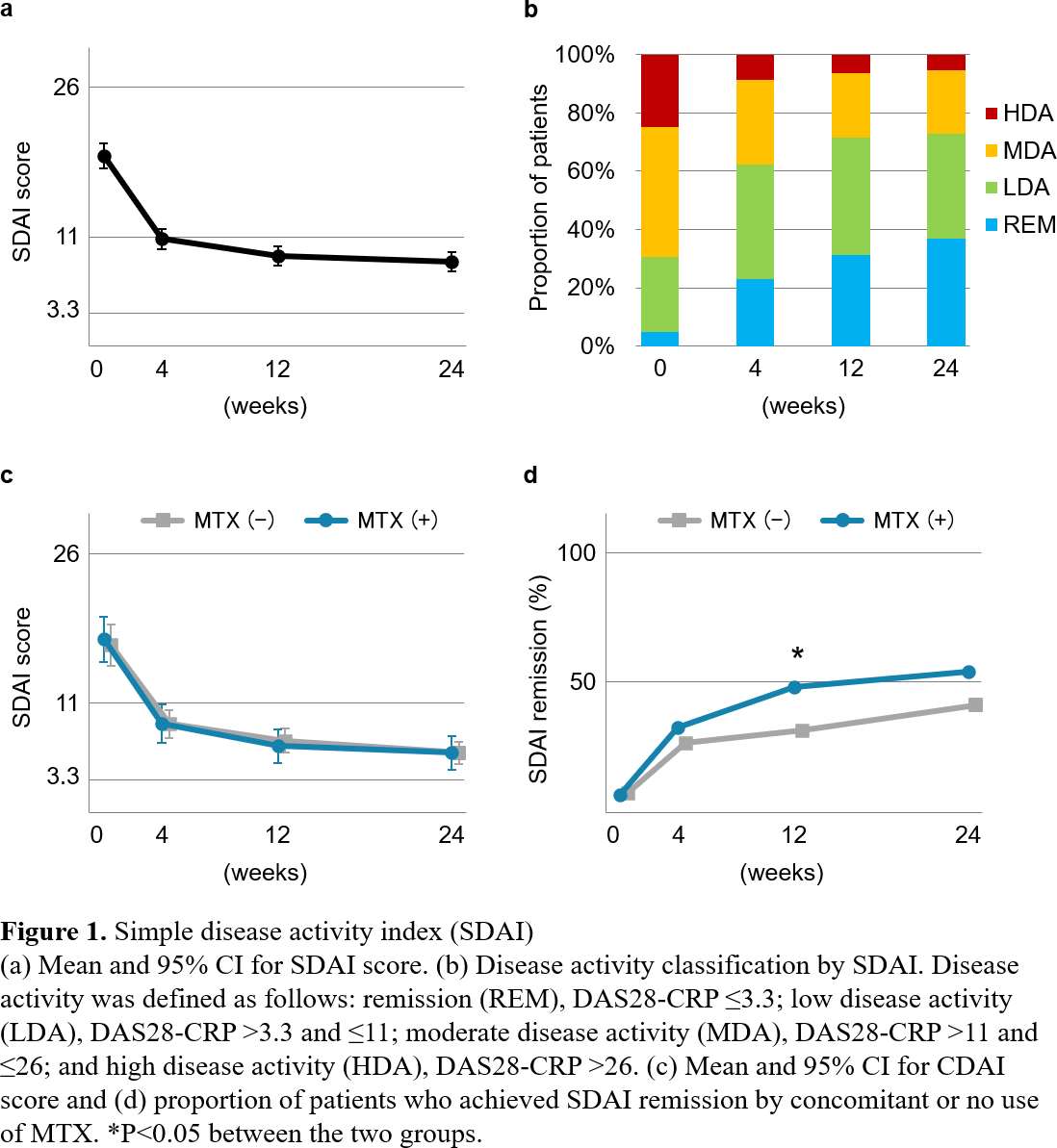
Results: Baseline characteristics of all patients included in this study (mean ± SD or %) was as follows: age 64 ± 14 years, 80% female, disease duration 13 ± 12 years, 83% anti-CCP antibody positive, 68% previous us of b/tsDMARDs, 55% MTX use, 39% glucocorticoid use (Table 1). SDAI score was 18.9 ± 12.0 at baseline, and significantly decreased to 10.7 ± 9.5 at 4 weeks (Figure 1a). 37% of patients were in SDAI remission at 24 weeks (Figure 1b). Multivariate analysis revealed that anti-CCP antibody positivity [odds ratio (OR): 2.09, 95% confidence interval (CI): 1.05-4.15], glucocorticoid use (OR: 0.40, 95% CI: 0.24-0.67) and SDAI score (OR: 0.96, 95% CI: 0.94-0.98) independently predicted SDAI remission at 24 weeks. None of the other variables, including concomitant use of MTX, predicted remission. Before propensity score matching, relative to the MTX (-) group, the MTX (+) group were more likely to be younger (mean ± SD, 61 ± 15 vs. 69 ± 12 years, P<0.001), and have a shorter disease duration (10 ± 10 vs. 16 ± 14 years, P<0.001), lower rates of chronic kidney disease defined as an eGFR<60 mL/min/1.73m 2 (22% vs. 40%, P<0.001), and lower rates of previous use of b/tsDMARDs (59% vs. 79%, P=0.008) (Table 1). A total of 119 matched pairs of patients were identified upon propensity score matching. There was good balance across all characteristics between the two groups. There was no significant difference in SDAI scores between the two groups throughout 0-24 weeks (Figure 1c). The proportion of patients who achieved SDAI remission was significantly higher in the MTX (+) group than in the MTX (-) group at 12 weeks, but it became comparable at 24 weeks (Figure 1d).
Conclusion: Anti-CCP antibody positivity, no use of glucocorticoid, and lower SDAI score at baseline independently predicted SDAI remission at 24 weeks in patients treated with JAK inhibitors. Moreover, concomitant MTX contributed to achieving remission earlier.
REFERENCES: [1] Genovese MC, et al. Rheumatology (Oxford) 2018;57:900-8
[2] Weinblatt ME, et al. ACR Annual Meeting 2019 [Abstract 516]
Acknowledgements: NIL.
Disclosure of Interests: Shuji Asai AbbVie, Astellas, Eisai, Eli Lilly, Gilead Sciences, UCB Japan, Astellas, Kenya Terabe AbbVie, Astellas, Eisai, Eli Lilly, Gilead Sciences, UCB Japan, AbbVie, Nobunori Takahashi AbbVie, Astellas, Eisai, Eli Lilly, Gilead Sciences, UCB Japan, Yutaka Yoshioka: None declared, Tatsuo Watanabe: None declared, Toshihisa Kojima AbbVie, Astellas, Eisai, Eli Lilly, Gilead Sciences, UCB Japan, Tomonori Kobayakawa: None declared, Yasumori Sobue: None declared, Yasuhide Kanayama: None declared, Takuya Matsumoto: None declared, Yuji Hirano AbbVie, Astellas, Eisai, Eli Lilly, Gilead Sciences, UCB Japan, Takefumi Kato: None declared, Takayoshi Fujibayashi: None declared, Masahiro Hanabayashi: None declared, Tsuyoshi Nishiume: None declared, Mochihito Suzuki Eisai, Shiro Imagama: None declared.