Background: Sodium-glucose cotransporter type 2 inhibitors (SGLT2i) have revolutionized the management of type 2 diabetes, heart failure, and chronic kidney disease (CKD), all comorbidities which are also highly prevalent among individuals with gout. SGLT2i have also been reported to lower serum urate (SU), the causal mediator of gout, by up to 1.8 mg/dL among those with hyperuricemia. 1 However, no data are available specifically among patients with gout.
Objectives: To investigate the SU change among patients with gout initiating SGLT2i, and to compare with sulfonylurea, the second-most widely used glucose-lowering medication after metformin.
Methods: Using electronic health records data from an academic, tertiary healthcare network in Boston, Massachusetts, we conducted a new-user comparative effectiveness cohort study of patients with gout with baseline SU >6 mg/dL who had SU measured within 90 days before and after SGLT2i or sulfonylurea initiation. Patients with gout were identified using an algorithm with a positive predictive value of 0.9 against the 2015 ACR/EULAR gout classification criteria. A rheumatologist reviewed all identified patient records to confirm their gout diagnosis and the initiation and continuation of SGLT2i or sulfonylurea during the study period (up to 6 months). Individuals who had SU measured <7 days after medication initiation or in the setting of an acute gout flare were excluded, as were patients who initiated or changed the dose of urate-lowering therapy (ULT) just before or during the study period. We first calculated the within-group SU change. Using multivariable linear regression, we compared SU change among SGLT2i initiators between those with and without diabetes and then compared SU change between SGLT2i and sulfonylurea.
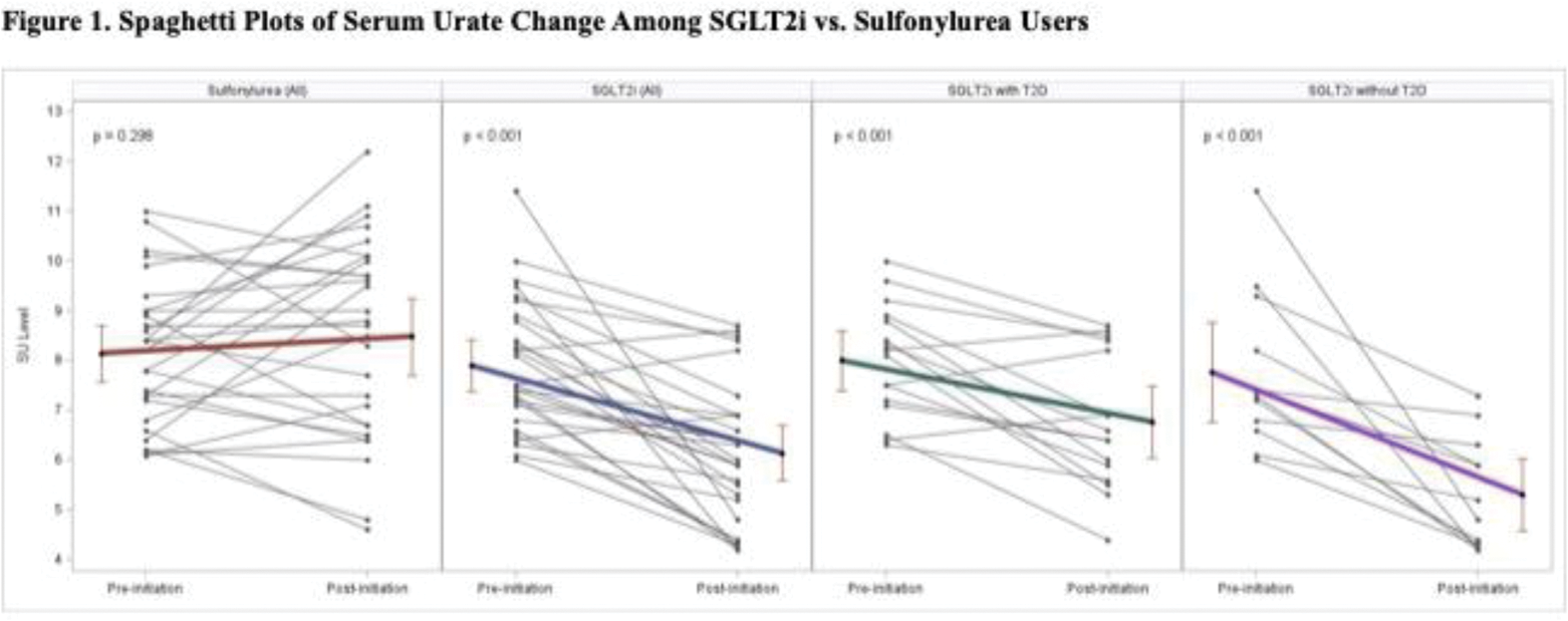
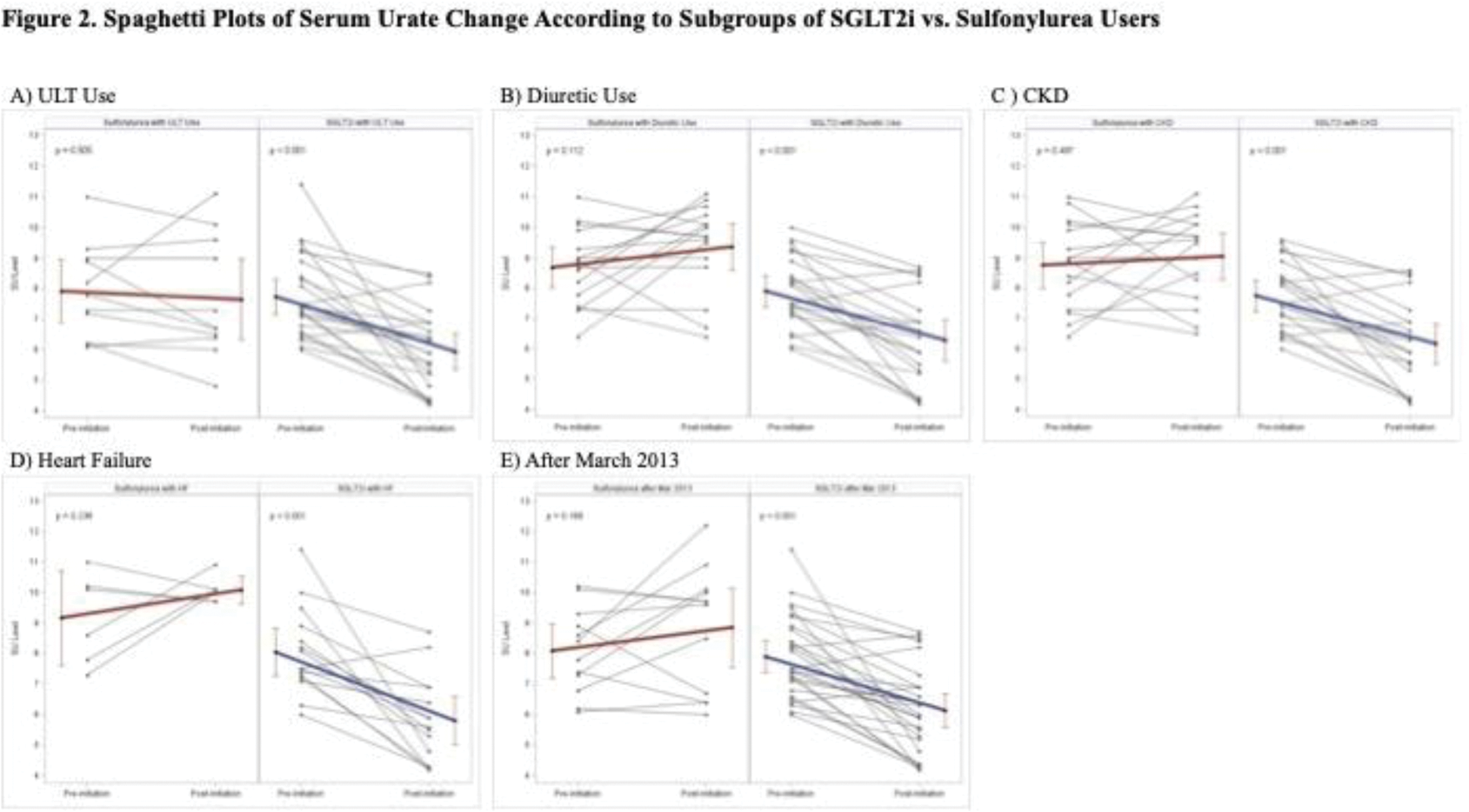
Results: We identified 28 gout patients initiating SGLT2i (including 16 with diabetes) and 28 patients initiating sulfonylurea (all with diabetes). The SGLT2i group tended to be older (mean age 66.8 years vs. 61.9 years) and included fewer females (7.1% vs. 14.3%) than the sulfonylurea group. Mean baseline SU were similar at 7.9 (1.3) mg/dL and 8.1 (1.4) mg/dL in the SGLT2i and sulfonylurea groups, respectively. The prevalence of CKD stage 3 or greater and heart failure tended to be higher in the SGLT2i group compared to the sulfonylurea group, consistent with the approved indications for SGLT2i beyond glycemic control. Both ULT and diuretic use were higher among SGLT2i users compared with sulfonylurea users. Most SGLT2i users were on empagliflozin (78.6%, compared with 17.9% dapagliflozin and 3.6% canagliflozin). Among SGLT2i initiators, the mean within-group SU change was -1.8 (95% CI, -2.4 to -1.1) mg/dL, including -1.2 (-1.8 to -0.6) mg/dL and -2.5 (-3.6 to -1.3) mg/dL among patients with and without diabetes, respectively, with an adjusted difference between those with and without diabetes of -1.4 (-2.4 to -0.5) mg/dL ( Figure 1 ). The SU did not change after initiating sulfonylurea (+0.3 [-0.3 to 1.0] mg/dL) ( Figure 1 ). The adjusted SU change difference between SGLT2i vs. sulfonylurea initiation was -1.8 (-2.7 to -0.9) mg/dL in all patients. The SU reduction persisted regardless of urate-lowering therapy or diuretic use and the presence of diabetes, chronic kidney disease, or heart failure ( Figure 2 ).
Conclusion: Among gout patients, SGLT2i was associated with a notable reduction in SU compared with sulfonylurea, with a larger reduction among non-diabetic gout patients. With their proven cardiovascular-kidney-metabolic benefits, adding SGLT2i to current gout management could provide streamlined benefits for gout and its comorbidities.
REFERENCES: [1] Doehner et al., PMID: 35788657
Acknowledgements: NIL.
Disclosure of Interests: Chio Yokose: None declared, Gregory Challener: None declared, Bohang Jiang: None declared, Baijun Zhou: None declared, Natalie McCormick: None declared, Sruthi Tanikella: None declared, Kila Panchot: None declared, Minna J. Kohler: None declared, Janeth Yinh: None declared, Yuqing Zhang: None declared, David Bates: None declared, James Januzzi Imbria Pharmaceuticals (board member), Abbott Diagnostics, AstraZeneca, Beckman Coulter, Jana Care, Janssen, Novartis, Prevencio, Quidel, Roche Diagnostics, Abbott Diagnostics, Applied Therapeutics, HeartFlow, Innolife and Novartis, Meghan Sise: None declared, Deborah Wexler: None declared, Hyon Choi Ani, LG, Horizon, Shanton, and Protalix, Horizon